Abstract
Aims
We evaluated the efficacy of biochar application for suppressing bacterial wilt of tomato and identified the potential underlying mechanisms involved in the disease control.
Methods
We measured the impact of two different sized biochar (53–120 μm and 380–830 μm) on bacterial wilt incidence in a greenhouse experiment. The efficiency of different sized biochar for the adsorption of tomato root exudates and the pathogen was further examined in vitro. We also quantified the effects of biochar and tomato root exudates on two pathogen virulence factors, chemotaxis, swarming motility and examined the effect of biochar on pathogen root colonization.
Results
Fine biochar application (3%; w:w) significantly decreased the bacterial wilt incidence by 19.9%. Biochar with different particle size had similar adsorption capacity for root exudates, while fine biochar was efficient (91%) in pathogen adsorption. Root exudates and fine biochar increased the chemotaxis ability of pathogen, while fine biochar reduced pathogen swarming motility and rhizosphere colonization.
Conclusions
Application of fine biochar can significantly decreased bacterial wilt incidence. This was mechanistically explained by biochar ability to 1) adsorb pathogen directly and indirectly via adsorption of root exudates (based on pathogen chemotaxis) and to 2) directly suppress pathogen swarming motility and subsequent root colonization.





Similar content being viewed by others
References
Abit SM, Bolster CH, Cai P, Walker SL (2012) Influence of feedstock and pyrolysis temperature of biochar amendments on transport of Escherichia coli in saturated and unsaturated soil. Environ Sci Technol 46:8097–8105. doi:10.1021/es300797z
Addy HS, Askora A, Kawasaki T, Fujie M, Yamada T (2012) Loss of virulence of the phytopathogen Ralstonia solanacearum through infection by phiRSM filamentous phages. Phytopathology 102:469–477. doi:10.1094/PHYTO-11-11-0319-R
Ahmad M, Rajapaksha AU, Lim JE, Zhang M, Bolan N, Mohan D, Vithanage M, Lee SS, Ok YS (2014) Biochar as a sorbent for contaminant management in soil and water: a review. Chemosphere 99:19–33. doi:10.1016/j.chemosphere.2013.10.071
Atkinson CJ, Fitzgerald JD, Hipps NA (2010) Potential mechanisms for achieving agricultural benefits from biochar application to temperate soils: a review. Plant Soil 337:1–18. doi:10.1007/s11104-010-0464-5
Badri DV, Chaparro JM, Zhang R, Shen Q, Vivanco JM (2013) Application of natural blends of phytochemicals derived from the root exudates of Arabidopsis to the soil reveal that phenolic-related compounds predominantly modulate the soil microbiome. J Biol Chem 288:4502–4512. doi:10.1074/jbc.M112.433300
Bais HP, Prithiviraj B, Jha AK, Ausubel FM, Vivanco JM (2005) Mediation of pathogen resistance by exudation of antimicrobials from roots. Nature 434:217–221. doi:10.1038/nature09809
Bais HP, Weir TL, Perry LG, Gilroy S, Vivanco JM (2006) The role of root exudates in rhizosphere interactions with plants and other organisms. Annu Rev Plant Biol 57:233–266. doi:10.1146/annurev.arplant.57.032905.105159
Boyer F-D, de Saint GA, Pouvreau J-B, Clavé G, Pillot J-P, Roux A, Rasmussen A, Depuydt S, Lauressergues D, Frei dit Frey N, Heugebaert TSA, Stevens CV, Geelen D, Goormachtig S, Rameau C (2014) New strigolactone analogs as plant hormones with low activities in the rhizosphere. Mol Plant 7:675–690. doi:10.1093/mp/sst163
Brewer CE, Chuang VJ, Masiello CA, Gonnermann H, Gao X, Dugan B, Driver LE, Panzacchi P, Zygourakis K, Davies CA (2014) New approaches to measuring biochar density and porosity. Biomass Bioenergy 66:176–185. doi:10.1016/j.biombioe.2014.03.059
Broek AV, Vanderleyden J (1995) The role of bacterial motility, chemotaxis, and attachment in bacteria-plant interactions. Mol Plant-Microbe Interact 8:800–810. doi:10.1094/MPMI-8-0800
Callaway RM (2000) Invasive plants versus their new and old neighbors: a mechanism for exotic invasion. Science 290:521–523. doi:10.1126/science.290.5491.521
Chen Y, Yan F, Chai Y, Liu H, Kolter R, Losick R, Guo J (2013) Biocontrol of tomato wilt disease by Bacillus subtilis isolates from natural environments depends on conserved genes mediating biofilm formation. Environ Microbiol 15:848–864. doi:10.1111/j.1462-2920.2012.02860.x
Colburn-Clifford JM, Scherf JM, Allen C (2010) Ralstonia solanacearum Dps contributes to oxidative stress tolerance and to colonization of and virulence on tomato plants. Appl Environ Microbiol 76:7392–7399. doi:10.1128/AEM.01742-10
Digonnet C, Martinez Y, Denance N, Chasseray M, Dabos P, Ranocha P, Marco Y, Jauneau A, Goffner D (2012) Deciphering the route of Ralstonia solanacearum colonization in Arabidopsis thaliana roots during a compatible interaction: focus at the plant cell wall. Planta 236:1419–1431. doi:10.1007/s00425-012-1694-y
Elad Y, David DR, Harel YM, Borenshtein M, Kalifa HB, Silber A, Graber ER (2010) Induction of systemic resistance in plants by biochar, a soil-applied carbon sequestering agent. Phytopathology 100:913–921. doi:10.1094/PHYTO-100-9-0913
Elmer WH, Pignatello JJ (2011) Effect of biochar amendments on mycorrhizal associations and fusarium crown and root rot of asparagus in replant soils. Plant Dis 95:960–966. doi:10.1094/pdis-10-10-0741
Elphinstone J, Hennessy J, Wilson J, Stead D (1996) Sensitivity of different methods for the detection of Ralstonia solanacearum in potato tuber extracts. EPPO Bull 26:663–678. doi:10.1111/j.1365-2338.1996.tb01511.x
Fujiwara A, Fujisawa M, Hamasaki R, Kawasaki T, Fujie M, Yamada T (2011) Biocontrol of Ralstonia solanacearum by treatment with lytic bacteriophages. Appl Environ Microbiol 77:4155–4162. doi:10.1128/AEM.02847-10
Genin S (2010) Molecular traits controlling host range and adaptation to plants in Ralstonia solanacearum. New Phytol 187:920–928. doi:10.1111/j.1469-8137.2010.03397.x
Ghosh U, Luthy RG, Cornelissen G, Werner D, Menzie CA (2011) In-situ sorbent amendments: a new direction in contaminated sediment management. Environ Sci Technol 45:1163–1168. doi:10.1021/es102694h
Gibert O, Lefevre B, Fernandez M, Bernat X, Paraira M, Calderer M, Martinez-Llado X (2013) Characterising biofilm development on granular activated carbon used for drinking water production. Water Res 47:1101–1110. doi:10.1016/j.watres.2012.11.026
Hale L, Luth M, Kenney R, Crowley D (2014) Evaluation of pinewood biochar as a carrier of bacterial strain Enterobacter cloacae UW5 for soil inoculation. Appl Soil Ecol 84:192–199. doi:10.1016/j.apsoil.2014.08.001
Hayward AC (1991) Biology and epidemiology of bacterial wilt caused by Pseudomonas solanacearum. Annu Rev Phytopathol 29:65–87. doi:10.1146/annurev.py.29.090191.000433
Inyang M, Dickenson E (2015) The potential role of biochar in the removal of organic and microbial contaminants from potable and reuse water: a review. Chemosphere 134:232–240. doi:10.1016/j.chemosphere.2015.03.072
Ishihara T, Mitsuhara I, Takahashi H, Nakaho K (2012) Transcriptome analysis of quantitative resistance-specific response upon Ralstonia solanacearum infection in tomato. PLoS One 7:e46763. doi:10.1371/journal.pone.0046763
Jacobs JM, Babujee L, Meng F, Milling A, Allen C (2012) The in planta transcriptome of Ralstonia solanacearum: conserved physiological and virulence strategies during bacterial wilt of tomato. MBio 3:e00114–e00112. doi:10.1128/mBio.00114-12
Jiang TY, Jiang J, Xu RK, Li Z (2012) Adsorption of Pb(II) on variable charge soils amended with rice-straw derived biochar. Chemosphere 89:249–256. doi:10.1016/j.chemosphere.2012.04.028
Kempe J, Sequeira L (1983) Biological control of bacterial wilt of potatoes: attempts to induce resistance by treating tubers with bacteria. Plant Dis 67:499–503. doi:10.1094/PD-67-499
Kinney TJ, Masiello CA, Dugan B, Hockaday WC, Dean MR, Zygourakis K, Barnes RT (2012) Hydrologic properties of biochars produced at different temperatures. Biomass Bioenergy 41:34–43. doi:10.1016/j.biombioe.2012.01.033
Lehmann J, Rillig MC, Thies J, Masiello CA, Hockaday WC, Crowley D (2011) Biochar effects on soil biota – a review. Soil Biol Biochem 43:1812–1836. doi:10.1016/j.soilbio.2011.04.022
Liu Z, Dugan B, Masiello CA, Barnes RT, Gallagher ME, Gonnermann H (2016) Impacts of biochar concentration and particle size on hydraulic conductivity and DOC leaching of biochar–sand mixtures. J Hydrol 533:461–472. doi:10.1016/j.jhydrol.2015.12.007
Masiello CA, Chen Y, Gao X, Liu S, Cheng H-Y, Bennett MR, Rudgers JA, Wagner DS, Zygourakis K, Silberg JJ (2013) Biochar and microbial signaling: production conditions determine effects on microbial communication. Environ Sci Technol 47:11496–11503. doi:10.1021/es401458s
Mendes R, Kruijt M, de Bruijn I, Dekkers E, van der Voort M, Schneider JH, Piceno YM, DeSantis TZ, Andersen GL, Bakker PA, Raaijmakers JM (2011) Deciphering the rhizosphere microbiome for disease-suppressive bacteria. Science 332:1097–1100. doi:10.1126/science.1203980
Miyake-Nakayama C, Ikatsu H, Kashihara M, Tanaka M, Arita M, Miyoshi S, Shinoda S (2006) Biodegradation of dichloromethane by the polyvinyl alcohol-immobilized methylotrophic bacterium Ralstonia metallidurans PD11. Appl Microbiol Biotechnol 70:625–630. doi:10.1007/s00253-005-0194-4
Mohanty SK, Boehm AB (2014) Escherichia coli Removal in biochar-augmented biofilter: effect of infiltration rate, initial bacterial concentration, biochar particle size, and presence of compost. Environ Sci Technol 48:11535–11542. doi:10.1021/es5033162
Mohanty SK, Cantrell KB, Nelson KL, Boehm AB (2014) Efficacy of biochar to remove Escherichia coli from stormwater under steady and intermittent flow. Water Res 61:288–296. doi:10.1016/j.watres.2014.05.026
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497. doi:10.1111/j.1399-3054.1962.tb08052.x
Naka K, Watarai S, Inoue K, Kodama Y, Oguma K, Yasuda T, Kodama H (2001) Adsorption effect of activated charcoal on enterohemorrhagic Escherichia coli. J Vet Med Sci 63:281–285. doi:10.1292/jvms.63.281
Oleszczuk P, Hale SE, Lehmann J, Cornelissen G (2012) Activated carbon and biochar amendments decrease pore-water concentrations of polycyclic aromatic hydrocarbons (PAHs) in sewage sludge. Bioresour Technol 111:84–91. doi:10.1016/j.biortech.2012.02.030
Panke-Buisse K, Poole AC, Goodrich JK, Ley RE, Kao-Kniffin J (2015) Selection on soil microbiomes reveals reproducible impacts on plant function. ISME J 9:980–989. doi:10.1038/ismej.2014.196
Park SY, Kim R, Ryu CM, Choi SK, Lee CH, Kim JG, Park SH (2008) Citrinin, a mycotoxin from Penicillium citrinum, plays a role in inducing motility of Paenibacillus polymyxa. FEMS Microbiol Ecol 65:229–237. doi:10.1111/j.1574-6941.2008.00492.x
Raaijmakers JM, Paulitz TC, Steinberg C, Alabouvette C, Moënne-Loccoz Y (2008) The rhizosphere: a playground and battlefield for soilborne pathogens and beneficial microorganisms. Plant Soil 321:341–361. doi:10.1007/s11104-008-9568-6
Rivera-Utrilla J, Bautista-Toledo I, Ferro-García MA, Moreno-Castilla C (2001) Activated carbon surface modifications by adsorption of bacteria and their effect on aqueous lead adsorption. J Chem Technol Biotechnol 76:1209–1215. doi:10.1002/jctb.506
Rudrappa T, Czymmek KJ, Pare PW, Bais HP (2008) Root-secreted malic acid recruits beneficial soil bacteria. Plant Physiol 148:1547–1556. doi:10.1104/pp.108.127613
Salanoubat M, Genin S, Artiguenave F, Gouzy J, Mangenot S, Arlat M, Billault A, Brottier P, Camus JC, Cattolico L, Chandler M, Choisne N, Claudel-Renard C, Cunnac S, Demange N, Gaspin C, Lavie M, Moisan A, Robert C, Saurin W, Schiex T, Siguier P, Thebault P, Whalen M, Wincker P, Levy M, Weissenbach J, Boucher CA (2002) Genome sequence of the plant pathogen Ralstonia solanacearum. Nature 415:497–502. doi:10.1038/415497a
Schreiner K, Hagn A, Kyselkova M, Moenne-Loccoz Y, Welzl G, Munch JC, Schloter M (2010) Comparison of barley succession and take-all disease as environmental factors sha** the rhizobacterial community during take-all decline. Appl Environ Microbiol 76:4703–4712. doi:10.1128/AEM.00481-10
Shi S, Nuccio E, Herman DJ, Rijkers R, Estera K, Li J, da Rocha UN, He Z, Pett-Ridge J, Brodie EL, Zhou J, Firestone M (2015) Successional trajectories of rhizosphere bacterial communities over consecutive seasons. MBio 6:e00746. doi:10.1128/mBio.00746-15
Sohi S, Krull E, Lopez-Capel E, Bol R (2010) A review of biochar and its use and function in soil. Adv Argon 105:47–82. doi:10.1016/S0065-2113 (10)05002-9
Sun H, Hockaday WC, Masiello CA, Zygourakis K (2012) Multiple controls on the chemical and physical structure of biochars. Ind Eng Chem Res 51:3587–3597. doi:10.1021/ie201309r
Taghizadeh-Toosi A, Clough TJ, Sherlock RR, Condron LM (2011) Biochar adsorbed ammonia is bioavailable. Plant Soil 350:57–69. doi:10.1007/s11104-011-0870-3
Tan S, Gu Y, Yang C, Dong Y, Mei X, Shen Q, Xu Y (2015) Bacillus amyloliquefaciens T-5 may prevent Ralstonia solanacearum infection through competitive exclusion. Biol Fertil Soils 52:341–351. doi:10.1007/s00374-015-1079-z
Tans-Kersten J, Huang H, Allen C (2001) Ralstonia solanacearum needs motility for invasive virulence on tomato. J Bacteriol 183:3597–3605. doi:10.1128/JB.183.12.3597-3605.2001
Tans-Kersten J, Brown D, Allen C (2004) Swimming motility, a virulence trait of Ralstonia solanacearum, is regulated by FlhDC and the plant host environment. Mol Plant-Microbe Interact 17:686–695. doi:10.1094/MPMI.2004.17.6.686
Tong X-j, Li J-y, Yuan J-h, Xu R-k (2011) Adsorption of Cu(II) by biochars generated from three crop straws. Chem Eng J 172:828–834. doi:10.1016/j.cej.2011.06.069
van Overbeek LS, Bergervoet JH, Jacobs FH, van Elsas JD (2004) The low-temperature-induced viable-but-nonculturable state affects the virulence of Ralstonia solanacearum biovar 2. Phytopathology 94:463–469. doi:10.1094/PHYTO.2004.94.5.463
Wardle DA, Zackrisson O, Nilsson M-C (1998) The charcoal effect in boreal forests: mechanisms and ecological consequences. Oecologia 115:419–426. doi:10.1007/s004420050536
Warnock DD, Lehmann J, Kuyper TW, Rillig MC (2007) Mycorrhizal responses to biochar in soil – concepts and mechanisms. Plant Soil 300:9–20. doi:10.1007/s11104-007-9391-5
Wei Z, Yang X, Yin S, Shen Q, Ran W, Xu Y (2011) Efficacy of bacillus-fortified organic fertiliser in controlling bacterial wilt of tomato in the field. Appl Soil Ecol 48:152–159. doi:10.1016/j.apsoil.2011.03.013
Wei Z, Huang J-F, Hu J, Gu Y-A, Yang C-L, Mei X-L, Shen Q-R, Xu Y-C, Friman V-P (2015a) Altering transplantation time to avoid periods of high temperature can efficiently reduce bacterial wilt disease incidence with tomato. PLoS One 10:e0139313. doi:10.1371/journal.pone.0139313
Wei Z, Yang T, Friman V-P, Xu Y, Shen Q, Jousset A (2015b) Trophic network architecture of root-associated bacterial communities determines pathogen invasion and plant health. Nat Commun 6:8413. doi:10.1038/ncomms9413
**e X, Yoneyama K, Kisugi T, Uchida K, Ito S, Akiyama K, Hayashi H, Yokota T, Nomura T (2013) Confirming stereochemical structures of strigolactones produced by rice and tobacco. Mol Plant 6:153–163. doi:10.1093/mp/sss139
Xue QY, Yin YN, Yang W, Heuer H, Prior P, Guo JH, Smalla K (2011) Genetic diversity of Ralstonia solanacearum strains from China assessed by PCR-based fingerprints to unravel host plant- and site-dependent distribution patterns. FEMS Microbiol Ecol 75:507–519. doi:10.1111/j.1574-6941.2010.01026.x
Yao J, Allen C (2006) Chemotaxis is required for virulence and competitive fitness of the bacterial wilt pathogen Ralstonia solanacearum. J Bacteriol 188:3697–3708. doi:10.1128/JB.188.10.3697
Zhang G, Zhang Q, Sun K, Liu X, Zheng W, Zhao Y (2011) Sorption of simazine to corn straw biochars prepared at different pyrolytic temperatures. Environ Pollut 159:2594–2601. doi:10.1016/j.envpol.2011.06.012
Zheng W, Guo M, Chow T, Bennett DN, Rajagopalan N (2010) Sorption properties of greenwaste biochar for two triazine pesticides. J Hazard Mater 181:121–126. doi:10.1016/j.jhazmat.2010.04.103
Zygourakis K, Sun H, Markenscoff P (2013) A nanoscale model for characterizing the complex pore structure of biochars. AIChE J 59:3412–3420. doi:10.1002/aic.14134
Acknowledgements
This research was supported by the National Key Basic Research Program of China (2015CB150503), the National Natural Science Foundation of China (41471213, 41671248), the 111 project (B12009), Jiangsu Key Technology R&D Program (BE2014340), the Young Elite Scientist Sponsorship Program by CAST (2015QNRC001), the Priority Academic Program Development (PAPD) of Jiangsu Higher Education Institutions (Qirong Shen), and the Qing Lan Project (Yangchun Xu).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Responsible Editor: Birgit Mitter.
Electronic supplementary material
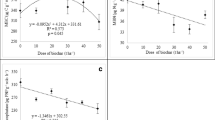
Fig. S1
Dose-response for the adsorption of R. solanacearum QL-Rs1115 by biochar with different particle size. Error bars indicate the standard error of the mean. Different letters indicate significant differences between different treatments. (GIF 40 kb)
Rights and permissions
About this article
Cite this article
Gu, Y., Hou, Y., Huang, D. et al. Application of biochar reduces Ralstonia solanacearum infection via effects on pathogen chemotaxis, swarming motility, and root exudate adsorption. Plant Soil 415, 269–281 (2017). https://doi.org/10.1007/s11104-016-3159-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-016-3159-8