Abstract
Purpose
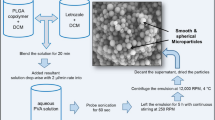
Biodegradable poly(lactide-co-glycolide) (PLGA) microparticles loaded with either risperidone or naltrexone were prepared from an emulsification homogenization process. The objective of this study was to determine the impact the post-treatment temperature has on the properties and subsequent performance of the microparticles.
Methods
The post-treatment temperature of an ethanolic solution was characterized from 10 ~ 35ºC for the naltrexone and risperidone micropartilces.
Results
The wash temperature resulted in a typical triphasic in vitro release pattern at low wash temperatures or a biphasic pattern consisting of an elevated release rate at higher post-treatment temperatures. The post-treatment temperature largely influences the particle morphology, residual solvent levels, glass transition temperature, and drug loading and is molecule dependent, whereby these characteristics subsequently influence the drug release rate.
Conclusion
The study highlights the importance of both the post-treatment process and control during manufacturing to obtain a formulation within the desired product profile.





Similar content being viewed by others
Data Availability
The datasets used and/or analyzed during the present study are available from the corresponding author on reasonable request.
References
Park K, Otte A, Sharifi F, Garner J, Skidmore S, Park H, Jhon YK, Qin B, Wang Y. Potential roles of the glass transition temperature of PLGA microparticles in drug release kinetics. Mol Pharm. 2021;18:18–32.
Park K, Skidmore S, Hadar J, Garner J, Park H, Otte A, Soh BK, Yoon G, Yu D, Yun Y, Lee BK, Jiang X, Wang Y. Injectable, long-acting PLGA formulations: Analyzing PLGA and understanding microparticle formation. J Control Release. 2019;304:125–34.
Park K, Otte A, Sharifi F, Garner J, Skidmore S, Park H, Jhon YK, Qin B, Wang Y. Formulation composition, manufacturing process, and characterization of poly(lactide-co-glycolide) microparticles. J Control Release. 2021;329:1150–61.
Garner J, Skidmore S, Park H, Park K, Choi S, Wang Y. Beyond Q1/Q2: The impact of manufacturing conditions and test methods on drug release from PLGA-based microparticle depot formulations. J Pharm Sci. 2018;107:353–61.
Ochi M, Wan B, Bao Q, Burgess DJ. Influence of PLGA molecular weight distribution on leuprolide release from microspheres. Int J Pharm. 2021;599: 120450.
Wan B, Bao Q, Zou Y, Wang Y, Burgess DJ. Effect of polymer source variation on the properties and performance of risperidone microspheres. Int J Pharm. 2021;610: 121265.
Chen W, Palazzo A, Hennink WE, Kok RJ. Effect of particle size on drug loading and release kinetics of gefitinib-loaded plga microspheres. Mol Pharm. 2017;14:459–67.
Acharya G, Shin CS, Vedantham K, McDermott M, Rish T, Hansen K, Fu Y, Park K. A study of drug release from homogeneous PLGA microstructures. J Control Release. 2010;146:201–6.
Bodmeier R, McGinity JW. The preparation and evaluation of drug-containing poly(dl-lactide) microspheres formed by the solvent evaporation method. Pharm Res. 1987;4:465–71.
Vay K, Frieß W, Scheler S. A detailed view of microparticle formation by in-process monitoring of the glass transition temperature. Eur J Pharm Biopharm. 2012;81:399–408.
Andhariya JV, Shen J, Choi S, Wang Y, Zou Y, Burgess DJ. Development of in vitro-in vivo correlation of parenteral naltrexone loaded polymeric microspheres. J Control Release. 2017;255:27–35.
Shen J, Choi S, Qu W, Wang Y, Burgess DJ. In vitro-in vivo correlation of parenteral risperidone polymeric microspheres. J Control Release. 2015;218:2–12.
Otte A, Turasan H, Park K. Implications of particle size on the respective solid-state properties of naltrexone in PLGA microparticles. Int J Pharm. 2022;626:122170.
Sharifi F, Otte A, Yoon G, Park K. Continuous in-line homogenization process for scale-up production of naltrexone-loaded PLGA microparticles. J Control Release. 2020;325:347–58.
Hua Y, Wang Z, Wang D, Lin X, Liu B, Zhang H, Gao J, Zheng AA-O. Key factor study for generic long-acting PLGA microspheres based on a reverse engineering of vivitrol(®). Molecules. 2021;26:1247.
Andhariya JV, Choi SH, Wang Y, Zou Y, Burgess DJ, Shen J. Accelerated in vitro release testing method for naltrexone loaded PLGA microspheres. Int J Pharm. 2017;520(1–2):79–85.
D’Souza S, Faraj JA, Giovagnoli S, Deluca PP. Development of Risperidone PLGA Microspheres. J Drug Deliv. 2014;2014: 620464.
Kohno M, Andhariya JV, Wan B, Bao Q, Rothstein S, Hezel M, Wang Y, Burgess DJ. The effect of PLGA molecular weight differences on risperidone release from microspheres. Int J Pharm. 2020;582: 119339.
Shang Q, Zhang A, Wu Z, Huang S, Tian R. In vitro evaluation of sustained release of risperidone-loaded microspheres fabricated from different viscosity of PLGA polymers. Polym Adv Technol. 2018;29:384–93.
Amann LC, Gandal MJ, Lin R, Liang Y, Siegel SJ. In Vitro–In Vivo correlations of scalable PLGA-risperidone implants for the treatment of schizophrenia. Pharm Res. 2010;27:1730–7.
Su Z-X, Shi Y-N, Teng L-S, Li X, Wang L-X, Meng Q-F, Teng L-R, Li Y-X. Biodegradable poly(D, L-lactide-co-glycolide) (PLGA) microspheres for sustained release of risperidone: Zero-order release formulation. Pharm Dev Technol. 2011;16:377–84.
Lagreca E, Onesto V, Di Natale C, La Manna S, Netti PA, Vecchione R. Recent advances in the formulation of PLGA microparticles for controlled drug delivery. Prog Biomater. 2020;9:153–74.
Brittain HG, Dickason DA, Hotz J, Lyons SL, Ramstack JM, Wright SG. Polymorphic forms of naltrexone. In: Alkermes Pharma Ireland Ltd, 2007.
Li W-I, Anderson KW, Mehta RC, Deluca PP. Prediction of solvent removal profile and effect on properties for peptide-loaded PLGA microspheres prepared by solvent extraction/ evaporation method. J Control Release. 1995;37:199–214.
Yoshioka T, Kawazoe N, Tateishi T, Chen G. Effects of structural change induced by physical aging on the biodegradation behavior of PLGA films at physiological temperature. Macromol Mater Eng. 2011;296:1028–34.
Rawat A, Burgess DJ. Effect of physical ageing on the performance of dexamethasone loaded PLGA microspheres. Int J Pharm. 2011;415:164–8.
Marquette S, Peerboom C, Yates A, Denis L, Langer I, Amighi K, Goole J. Stability study of full-length antibody (anti-TNF alpha) loaded PLGA microspheres. Int J Pharm. 2014;470:41–50.
Blasi P, D’Souza SS, Selmin F, DeLuca PP. Plasticizing effect of water on poly(lactide-co-glycolide). J Control Release. 2005;108:1–9.
D’Souza S, Dorati R, DeLuca PP. Effect of hydration on physicochemical properties of end-capped PLGA. Adv Biomater. 2014;2014:834942.
Acknowledgements
This study was supported by UH3 DA048774 from the National Institute on Drug Abuse (NIDA) and the Showalter Research Trust Fund.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interests/Competing Interests
The authors declare no competing financial or conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Otte, A., Soh, B.K. & Park, K. The Impact of Post-Processing Temperature on PLGA Microparticle Properties. Pharm Res 40, 2677–2685 (2023). https://doi.org/10.1007/s11095-023-03568-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-023-03568-z