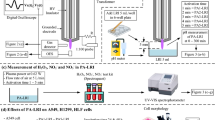
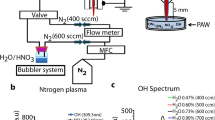
Abstract
The present study investigated the effect of plasma-produced reactive oxygen (ROS) and nitrogen (RNS) species on cancer cell viability. Reactive species were generated in deionized water by using an atmospheric pressure Ar plasma jet within the controlled ambient gases (air, O2 or N2), which allowed the production of plasma-activated water containing only ROS (e.g. O3, H2O2) or both ROS and RNS (e.g. H2O2, NO2–, NO3–). A considerable amount of H2O2 was produced in all ambient gases, and its generation rate was highest in N2 and lowest in O2. The latter was connected with a H2O2 precursor, OH, efficient quenching in O2 ambient gas. Small quantities of NO2– were generated during short (< 5 min) plasma treatments in ambient air and N2. The highest amount of NO3– was produced in N2 ambient gas. Ozone was detected only in the case of O2 environment. Cell viability studies were carried out by utilizing two cancer cell lines: 4T1 (breast cancer) and PPC-1 (prostate cancer). The results of the colorimetric succinate dehydrogenase activity assay showed that the studied cell lines had a similar sensitivity to the plasma activated medium. The impact of medium produced in the O2 ambient environment was determined by H2O2 content. The equivalent amount of H2O2 in the plasma activated medium produced in the N2 ambient environment caused an almost two-fold higher viability than in the case of the O2 ambient gas. It is proposed that this was due to the cellular proliferation enhancing effect of NH3.







Similar content being viewed by others
References
Hirst AM, Frame FM, Arya M et al (2016) Low temperature plasmas as emerging cancer therapeutics: the state of play and thoughts for the future. Tumor Biology 37:7021–7031. https://doi.org/10.1007/s13277-016-4911-7
Semmler ML, Bekeschus S, Schäfer M et al (2020) Molecular mechanisms of the efficacy of Cold Atmospheric Pressure Plasma (CAP) in cancer treatment. Cancers 12:269
Canal C, Fontelo R, Hamouda I et al (2017) Plasma-induced selectivity in bone cancer cells death. Free Radic Biol Med 110:72–80. https://doi.org/10.1016/j.freeradbiomed.2017.05.023
Vandamme M, Robert E, Pesnel S et al (2010) Antitumor effect of plasma treatment on u87 glioma xenografts: preliminary results. Plasma Processes Polym 7:264–273. https://doi.org/10.1002/ppap.200900080
Kumar N, Attri P, Dewilde S, Bogaerts A (2018) Inactivation of human pancreatic ductal adenocarcinoma with atmospheric plasma treated media and water: a comparative study. J Phys D Appl Phys. https://doi.org/10.1088/1361-6463/aac571
Wang M, Holmes B, Cheng X et al (2013) Cold atmospheric plasma for selectively ablating metastatic breast cancer cells. PLoS ONE 8:e73741. https://doi.org/10.1371/journal.pone.0073741
Gerber IC, Mihai CT, Gorgan L et al (2017) Viability and cell biology for HeLa and vero cells after exposure to low-temperature air dielectric barrier discharge plasma. Plasma Med 7:159–173. https://doi.org/10.1615/PlasmaMed.2017019487
Van Der Paal J, Verheyen C, Neyts EC, Bogaerts A (2017) Hampering effect of cholesterol on the permeation of reactive oxygen species through phospholipids bilayer: possible explanation for plasma cancer selectivity. Sci Rep 7:1–11. https://doi.org/10.1038/srep39526
Yusupov M, Razzokov J, Cordeiro RM, Bogaerts A (2019) Transport of reactive oxygen and nitrogen species across aquaporin: a molecular level picture. Oxid Med Cell Longev. https://doi.org/10.1155/2019/2930504
Yan D, **ao H, Zhu W et al (2017) The role of aquaporins in the anti-glioblastoma capacity of the cold plasma-stimulated medium. J Phys D Appl Phys. https://doi.org/10.1088/1361-6463/aa53d6
Bekeschus S, Lin A, Fridman A et al (2018) A comparison of floating-electrode DBD and kINPen jet: plasma parameters to achieve similar growth reduction in colon cancer cells under standardized conditions. Plasma Chem Plasma Process 38:1–12. https://doi.org/10.1007/s11090-017-9845-3
Dezest M, Chavatte L, Bourdens M et al (2017) Mechanistic insights into the impact of cold atmospheric pressure plasma on human epithelial cell lines. Sci Rep 7:1–17. https://doi.org/10.1038/srep41163
Gülden M, Jess A, Kammann J et al (2010) Free radical biology & medicine cytotoxic potency of H 2 O 2 in cell cultures : impact of cell concentration and exposure time. Free Radical Biol Med 49:1298–1305. https://doi.org/10.1016/j.freeradbiomed.2010.07.015
Bienert GP, Chaumont F (2014) Aquaporin-facilitated transmembrane diffusion of hydrogen peroxide. Biochim Biophys Acta Gen Subj 1840:1596–1604. https://doi.org/10.1016/j.bbagen.2013.09.017
Direito I, Madeira A, Brito MA, Soveral G (2016) Aquaporin-5: from structure to function and dysfunction in cancer. Cell Mol Life Sci 73:1623–1640. https://doi.org/10.1007/s00018-016-2142-0
Razzokov J, Yusupov M, Cordeiro RM, Bogaerts A (2018) Atomic scale understanding of the permeation of plasma species across native and oxidized membranes. J Phys D Appl Phys. https://doi.org/10.1088/1361-6463/aad524
Girard P-M, Arbabian A, Fleury M et al (2016) Synergistic effect of H2O2 and NO2 in cell death induced by cold atmospheric He plasma. Sci Rep 6:29098. https://doi.org/10.1038/srep29098
Bauer G, Sersenová D, Graves DB, Machala Z (2019) Dynamics of singlet oxygen-triggered, RONS-based apoptosis induction after treatment of tumor cells with cold atmospheric plasma or plasma-activated medium. Sci Rep 9:1–34. https://doi.org/10.1038/s41598-019-50329-3
Xu D, Cui Q, Xu Y et al (2018) NO 2 - and NO 3 - enhance cold atmospheric plasma induced cancer cell death by generation of ONOO -. AIP Adv 8:105219. https://doi.org/10.1063/1.5046353
Cheng X, Sherman J, Murphy W et al (2014) The effect of tuning cold plasma composition on glioblastoma cell viability. PLoS ONE 9:1–9. https://doi.org/10.1371/journal.pone.0098652
Vermeylen S, De Waele J, Vanuytsel S et al (2016) Cold atmospheric plasma treatment of melanoma and glioblastoma cancer cells. Plasma Processes Polym 13:1195–1205. https://doi.org/10.1002/ppap.201600116
Uchida G, Nakajima A, Ito T et al (2016) Effects of nonthermal plasma jet irradiation on the selective production of H 2 O 2 and NO 2 − in liquid water. J Appl Phys 120:203302. https://doi.org/10.1063/1.4968568
Jõgi I, Talviste R, Raud S et al (2020) Comparison of two cold atmospheric pressure plasma jet configurations in argon. Contrib Plasma Phys. https://doi.org/10.1002/ctpp.201900127
Talviste R, Raud S, Jogi I et al (2019) Investigation of a He micro plasma-jet utilized for treatment of prostate cancer cells. Plasma Res Express. https://doi.org/10.1088/2516-1067/ab4ea9
Kaushik NK, Ghimire B, Li Y et al (2019) Biological and medical applications of plasma-activated media, water and solutions. Biol Chem 400:39–62. https://doi.org/10.1515/hsz-2018-0226
Yan D, Talbot A, Nourmohammadi N et al (2015) Principles of using cold atmospheric plasma stimulated media for cancer treatment. Sci Rep 5:1–17. https://doi.org/10.1038/srep18339
Jablonowski H, Hänsch MACh, Dünnbier M et al (2015) Plasma jet’s shielding gas impact on bacterial inactivation. Biointerphases 10:029506. https://doi.org/10.1116/1.4916533
Liu Z, Xu D, Liu D et al (2017) Production of simplex RNS and ROS by nanosecond pulse N2/O2 plasma jets with homogeneous shielding gas for inducing myeloma cell apoptosis. J Phys D Appl Phys. https://doi.org/10.1088/1361-6463/aa66f0
Azzariti A, Iacobazzi RM, Di Fonte R et al (2019) Plasma-activated medium triggers cell death and the presentation of immune activating danger signals in melanoma and pancreatic cancer cells. Sci Rep 9:1–13. https://doi.org/10.1038/s41598-019-40637-z
Lietz AM, Barnat EV, Foster JE, Kushner MJ (2020) Ionization wave propagation in a He plasma jet in a controlled gas environment. J Appl Phys. https://doi.org/10.1063/5.0020264
Noori H, Raud J, Talviste R, Jõgi I (2020) Water dissolution of nitrogen oxides produced by ozone oxidation of nitric oxide. Ozone: Sci Eng. https://doi.org/10.1080/01919512.2020.1839379
Lukes P, Dolezalova E, Sisrova I, Clupek M (2014) Aqueous-phase chemistry and bactericidal effects from an air discharge plasma in contact with water: Evidence for the formation of peroxynitrite through a pseudo-second-order post-discharge reaction of H2O2and HNO2. Plasma Sources Sci Technol. https://doi.org/10.1088/0963-0252/23/1/015019
Roti JL (2008) Cellular responses to hyperthermia (40–46°C): cell killing and molecular events. Int J Hyperth 24:3–15. https://doi.org/10.1080/02656730701769841
Jõgi I, Raud J, Hein K, Laan M (2014) Spectral characterization of medium-pressure RF discharge in argon-oxygen mixture. J Phys D Appl Phys. https://doi.org/10.1088/0022-3727/47/33/335206
Raud J, Jogi I, Matisen L et al (2017) Characterization of Ar/N2/H2 middle-pressure RF discharge and application of the afterglow region for nitridation of GaAs. J Phys D Appl Phys. https://doi.org/10.1088/1361-6463/aa9635
Yubero C, García MC, Calzada MD (2006) On the use of the Hα spectral line to determine the electron density in a microwave (2.45 GHz) plasma torch at atmospheric pressure. Spectrochim Acta - Part B Atomic Spectrosc 61:540–544. https://doi.org/10.1016/j.sab.2006.03.011
Park S, Choe W, Kim H, Park JY (2015) Continuum emission-based electron diagnostics for atmospheric pressure plasmas and characteristics of nanosecond-pulsed argon plasma jets. Plasma Sources Sci Technol. https://doi.org/10.1088/0963-0252/24/3/034003
Raud J, Laan M, Jgi I (2011) Rotational temperatures of N<inf>2</inf>(C,0) and OH(A,0) as gas temperature estimates in the middle pressure Ar/O<inf>2</inf> discharge. J Phys D Appl Phys. https://doi.org/10.1088/0022-3727/44/34/345201
Tachibana K, Nakamura T (2019) Comparative study of discharge schemes for production rates and ratios of reactive oxygen and nitrogen species in plasma activated water. J Phys D: Appl Phys. https://doi.org/10.1088/1361-6463/ab2529
Julák J, Hujacová A, Scholtz V et al (2018) Contribution to the chemistry of plasma-activated water. Plasma Phys Rep 44:125–136. https://doi.org/10.1134/S1063780X18010075
Kubota Y, Koga K, Ohno M, Hara T (2010) Synthesis of ammonia through direct chemical reactions between an atmospheric nitrogen plasma jet and a liquid. Plasma Fus Res 5:042–042. https://doi.org/10.1585/pfr.5.042
Royintarat T, Seesuriyachan P, Boonyawan D et al (2019) Mechanism and optimization of non-thermal plasma-activated water for bacterial inactivation by underwater plasma jet and delivery of reactive species underwater by cylindrical DBD plasma. Curr Appl Phys 19:1006–1014. https://doi.org/10.1016/j.cap.2019.05.020
Schmidt A, Bekeschus S, Jablonowski H et al (2017) Role of ambient gas composition on cold physical plasma-elicited cell signaling in keratinocytes. Biophys J 112:2397–2407. https://doi.org/10.1016/j.bpj.2017.04.030
He X, Lin J, Bangbang H et al (2018) The formation pathways of aqueous hydrogen peroxide in a plasma-liquid system with liquid as the cathode. Plasma Sources Sci Technol. https://doi.org/10.1140/epjd/e2020-100371-2
Gorbanev Y, O’Connell D, Chechik V (2016) Non-thermal plasma in contact with water: the origin of species. Chem Eur J 22:3496–3505. https://doi.org/10.1002/chem.201503771
Brisset JL, Hnatiuc E (2012) Peroxynitrite: a re-examination of the chemical properties of non-thermal discharges burning in air over aqueous solutions. Plasma Chem Plasma Process 32:655–674. https://doi.org/10.1007/s11090-012-9384-x
Brisset JL, Pawlat J (2016) Chemical effects of air plasma species on aqueous solutes in direct and delayed exposure modes: discharge, post-discharge and plasma activated water. Plasma Chem Plasma Process 36:355–381. https://doi.org/10.1007/s11090-015-9653-6
Tachibana K, Nakamura T (2019) Characterization of dielectric barrier discharges with water in correlation to productions of OH and H2O2 in gas and liquid phases. Jpn J Appl Phys. https://doi.org/10.7567/1347-4065/aafe73
Machala L, Tarabova B, Sersenova J, Hensel K (2019) Chemical and antibacterial effects of plasma activated water : correlation with gaseous and aqueous reactive oxygen and nitrogen species. J Phys D: Appl Phys 52:17
Sellevag SR, Georgievskii Y, Miller JA (2009) Kinetics of the gas-phase recombination reaction of hydroxyl radicals to form hydrogen peroxide. J Phys Chem A 113:4457–4467. https://doi.org/10.1021/jp8110524
Atkinson R, Baulch DL, Cox RA et al (1989) Evaluated kinetic and photochemical data for atmospheric chemistry: supplement III. IUPAC subcommittee on gas kinetic data evaluation for atmospheric chemistry. J Phys Chem Ref Data. https://doi.org/10.1063/1.555832
Atkinson R, Baulch DL, Cox RA et al (2004) Evaluated kinetic and photochemical data for atmospheric chemistry: part 1 – gas phase reactions of Ox, HOx, NOx and SOx species. Atmos Chem Phys Discuss 3:6179–6699. https://doi.org/10.5194/acpd-3-6179-2003
Sasaki S, Honda R, Hokari Y et al (2016) Characterization of plasma-induced cell membrane permeabilization: Focus on OH radical distribution. J Phys D Appl Phys. https://doi.org/10.1088/0022-3727/49/33/334002
Liu Y, Tan Z, Chen X et al (2019) A Numerical investigation on the effects of water vapor on electron energy and oh production in atmospheric-pressure he/h2o and ar/h2o plasma jets. IEEE Trans Plasma Sci 47:1593–1604. https://doi.org/10.1109/TPS.2019.2896060
Srivastava N, Wang C (2011) Effects of water addition on OH radical generation and plasma properties in an atmospheric argon microwave plasma jet. J Appl Phys. https://doi.org/10.1063/1.3632970
Staehelin J, Buehler RE, Hoigne J (1984) Ozone decomposition in water studied by pulse radiolysis. 2. Hydroxyl and hydrogen tetroxide (HO4) as chain intermediates. J Phys Chem 88:5999–6004. https://doi.org/10.1021/j150668a051
Ju LP, Han KL, Varandas AJC (2007) Variational transition-state theory study of the atmospheric reaction OH + O3 → HO2 + O2. Int J Chem Kinet 39:148–153. https://doi.org/10.1002/kin.20226
Sander R (2015) Compilation of Henry’s law constants (version 4.0) for water as solvent. Atmos Chem Phys 15:4399–4981
Schmidt-Bleker A, Bansemer R, Reuter S, Weltmann KD (2016) How to produce an NOx- instead of Ox-based chemistry with a cold atmospheric plasma jet. Plasma Processes Polym 13:1118–1125. https://doi.org/10.1002/ppap.201600062
Baulch DL, Cobos CJ, Cox RA et al (1992) Evaluated kinetic data for combustion modelling. J Phys Chem Ref Data. https://doi.org/10.1063/1.555908
Verlackt CCW, van Boxem W, Bogaerts A (2018) Transport and accumulation of plasma generated species in aqueous solution. Phys Chem Chem Phys 20:6845–6859. https://doi.org/10.1039/c7cp07593f
Shin KS, Michael JV (1991) Rate constants for the reactions H+O2→OH+O and D+O 2→OD+O over the temperature range 1085–2278 K by the laser photolysis-shock tube technique. J Chem Phys 95:262–273. https://doi.org/10.1063/1.461483
Simonetti V, Quagliariello V, Giustetto P et al (2017) Association of ozone with 5-fluorouracil and cisplatin in regulation of human colon cancer cell viability: in vitro anti-inflammatory properties of ozone in colon cancer cells exposed to lipopolysaccharides (Evidence-Based Complementary an. Evid-Based Complemen Altern Med. https://doi.org/10.1155/2017/7414083
Bekeschus S, Mueller A, Miller V et al (2017) Physical plasma elicits immunogenic cancer cell death and mitochondrial singlet oxygen. IEEE Trans Radiat Plasma Med Sci 2:138–146. https://doi.org/10.1109/trpms.2017.2766027
Zhu Z, Jiao L, Li T et al (2018) Expression of AQP3 and AQP5 as a prognostic marker in triple-negative breast cancer. Oncol Lett 16:2661–2667. https://doi.org/10.3892/ol.2018.8955
Bründl J, Wallinger S, Breyer J et al (2018) Expression, localisation and potential significance of aquaporins in benign and malignant human prostate tissue. BMC Urol 18:1–9. https://doi.org/10.1186/s12894-018-0391-y
Emerson K, Russo RC, Lund RE, Thurston RV (1975) Aqueous ammonia equilibrium calculations: effect of pH and temperature. J Fish Res Board Can 32:2379–2383. https://doi.org/10.1139/f75-274
Lie S, Wang T, Forbes B et al (2019) The ability to utilise ammonia as nitrogen source is cell type specific and intricately linked to GDH, AMPK and mTORC1. Sci Rep 9:1–15. https://doi.org/10.1038/s41598-018-37509-3
Acknowledgements
The authors would like to thank Dr. Indrek Renge for valuable discussions on chemistry in the liquid phase. The present study was supported by Estonian Science Agency Grant no PUT1432.
Funding
The study was supported by Estonian Science Agency Grant no PUT1432.
Author information
Authors and Affiliations
Contributions
S.R., J.R. and I.J. contributed to the study conception and design. Experiments and data analysis were conducted by S.R., J.R. and C-T. P. S.R. wrote the manuscript with input from all authors.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Data material
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Raud, S., Raud, J., Jõgi, I. et al. The Production of Plasma Activated Water in Controlled Ambient Gases and its Impact on Cancer Cell Viability. Plasma Chem Plasma Process 41, 1381–1395 (2021). https://doi.org/10.1007/s11090-021-10183-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11090-021-10183-6