Abstract
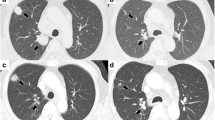
We report the case of a 75-year old woman who received sorafenib (Nexavar®, Bayer Pharmaceuticals Corporation, West Haven, CT) for a CNS relapse of clear cell renal cell carcinoma. After four months of sorafenib treatment, a brain magnetic resonance imaging showed 95%-volumetric regression of cerebral metastasis. To the best of our knowledge, this is the first almost complete resolution of brain metastases in renal cell carcinoma treated with sorafenib that has been described.


Similar content being viewed by others
References
Cohen H, McGovern F (2005) Renal Cell carcinoma. N Engl J Med 353:3477–3490
Flanigan RC, Mickisch G, Sylvester R et al (2004) Cytoreductive nephrectomy in patients with metastatic renal cancer: a combined analysis. J Urol 171:1071–1076. doi:10.1097/01.ju.0000110610.61545.ae
Gerstner ER, Fine RL (2007) Increased permeability of the blood–brain barrier to chemotherapy in metastatic brain tumors: establishing a treatment paradigm. J Clin Oncol 25(16):2306–2312. doi:10.1200/JCO.2006.10.0677
Costa LJ, Drabkin HA (2007) Renal cell carcinoma: new developments in molecular biology and potential for targeted therapies. Oncologist 12:1404–1415. doi:10.1634/theoncologist.12-12-1404
Chang YS, Henderson A, Xue D et al (2005) BAY 43-9006 (Sorafenib) inhibits ectopic and orthotopic growth of a murine model of renal adenocarcinoma (Renca) predominantly through inhibition of tumor angiogenesis. Proc Am Assoc Cancer Res: 46. Abstract
Strumberg D, Clark JW, Awada A et al (2007) Safety, pharmacokinetics, and preliminary antitumor activity of sorafenib: a review of four phase I trials in patients with advanced refractory solid tumors. Oncologist 12:426–437. doi:10.1634/theoncologist.12-4-426
Ratain MJ, Eisen T, Stadler WM et al (2006) Phase II placebo-controlled randomized discontinuation trial of sorafenib in patients with metastatic renal cell carcinoma. J Clin Oncol 24:2505–2512. doi:10.1200/JCO.2005.03.6723
Motzer RJ, Bacik J, Schwartz LH et al (2004) Prognostic factors for survival in previously treated patients with metastatic renal cell carcinoma. J Clin Oncol 22:454–463. doi:10.1200/JCO.2004.06.132
Bracarda S, Caserta C, Sordini L et al (2007) Protein kinase inhibitors in the treatment of renal cell carcinoma: sorafenib. Ann Oncol 18(Suppl 6):vi22–vi25. doi:10.1093/annonc/mdm219
Wilhelm SM, Carter C, Tang L et al (2004) BAY 43–9006 exhibits broad spectrum oral antitumor activity and targets the RAF/MEK/ERK pathway and receptor tyrosine kinases involved in tumor progression and angiogenesis. Cancer Res 64:7099–7109. doi:10.1158/0008-5472.CAN-04-1443
Motzer RJ, Bander NH, Nanus DM (1996) Renal-cell carcinoma. N Engl J Med 335:865–875. doi:10.1056/NEJM199609193351207
Ratain MJ, Eisen T, Stadler WM et al (2006) Phase II placebo-controlled randomised discontinuation trial of sorafenib in patients with metastatic renal cell carcinoma. J Clin Oncol 24:2505–2512. doi:10.1200/JCO.2005.03.6723
Escudier B, Eisen T, Stadler WM et al (2007) Sorafenib in advanced clear-cell renal-cell carcinoma. N Engl J Med 356:125–134. doi:10.1056/NEJMoa060655
Massard C, Zonierek J, Laplanche A et al (2006) Incidence of brain metastasis in advanced renal cell carcinoma among patients randomised in a phase III trial of sorafenib, an oral multi-kinase inhibitor. Ann Oncol 17:148–149 (suppl 9, abstr 454P)
Medioni J, Cojocarasu O, Belcaceres JL et al (2007) Complete cerebral response with sunitinib for metastatic renal cell carcinoma. Ann Oncol 18(7):1282–1283. doi:10.1093/annonc/mdm275
Trinh QD, Cardinal E, Gallina A et al (2006) Sunitinib relieves renal cell carcinoma spinal cord compression. Eur Urol 51(6):1741–1743. doi:10.1016/j.eururo.2006.10.050
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Valcamonico, F., Ferrari, V., Amoroso, V. et al. Long-lasting successful cerebral response with sorafenib in advanced renal cell carcinoma. J Neurooncol 91, 47–50 (2009). https://doi.org/10.1007/s11060-008-9676-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-008-9676-4