Abstract
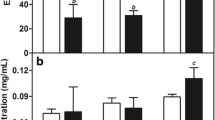
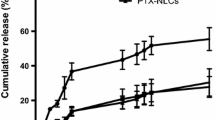
The N-(butanoyloxyethyl)-4-(chloromethyl)-3-nitrobenzamide (BNB) is a nitroaromatic derivative with significant antitumor activity. Preformulation, forced degradation (distilled water, acid and base hydrolysis, oxidation, and light), and formulation studies were performed to investigate the chemical behavior of the molecule, the physicochemical properties, and the impact of formulation variables. Pharmacokinetic properties for BNB were estimated in silico. Solid lipid nanoparticles (SLN) and nanostructured lipid carriers (NLC) containing BNB were developed by a hot melt homogenization method for parenteral administration. Degradation studies demonstrated that this compound is sensitive to hydrolysis. BNB was predicted to have a favorable absorption, distribution, metabolism, and excretion profile. The nanocarriers developed were characterized for particle size (PS = 61 to 85 nm), polydispersity index (PI < 0.3), zeta potential (ZP = − 22 to − 34 mV), and encapsulation efficiency (EE = 66 to 90%) and remained stable for 30 days of storage. These studies indicated that BNB (inhibitory concentration (IC50) 21.8 μM) and BNB-loaded NLC (IC50 33.7 μM) showed moderate cytotoxicity against breast cancer cell line. Blank formulations did not induce cytotoxicity and BNB-loaded SLN was able to potentiate the action of BNB (lC50 12.4 μM). BNB is a promising antitumor agent and it is possible to modulate its activity based on the particle size of the formulation.






Similar content being viewed by others
References
Anvisa. Farmacopeia Brasileira (2010) Agência Nacional de Vigilância Sanitária, 5th edn. http://www.anvisa.gov.br/hotsite/cd_farmacopeia/index.htm
Baertschi SW, Alsante KM, Reed RA (2011) Pharmaceutical stress testing: predicting drug degradation. Informa Health, London
Bao H, Zhang Q, Xu H, Yan Z (2016) Effects of nanoparticle size on antitumor activity of 10-hydroxycamptothecin-conjugated gold nanoparticles: in vitro and in vivo studies. Int J Nanomedicine 11:929–940. https://doi.org/10.2147/IJN.S96422
Beloqui A, Solinís MA, Rodríguez-Gascón A, Almeida AJ, Préat V (2016) Nanostructured lipid carriers: promising drug delivery systems for future clinics. Nanomedicine: NBM 12(1):143–161. https://doi.org/10.1016/j.nano.2015.09.004
Brioschi A, Zara GP, Calderoni S, Gasco MR, Mauro A (2008) Cholesterylbutyrate solid lipid nanoparticles as a butyric acid prodrug. Molecules 13(2):230–254. https://doi.org/10.3390/molecules13020230
Castro GA, Coelho AL, Oliveira CA, Machecha GA, Oréfice RL, Ferreira LA (2009) Formation of ion pairing as an alternative to improve encapsulation and stability and to reduce skin irritation of retinoic acid loaded in solid lipid nanoparticles. Int J Pharm 381(1):77–83. https://doi.org/10.1016/j.ijpharm.2009.07.025
Cancerrxgene (2017) Genomics of drug sensitivity in cancer. http://www.cancerrxgene.org/. Accessed 26 June 2017
ClinicalTrials.gov (2017) Study of HT-302 monotherapy as second-line treatment in advanced biliary tract cancer. https://clinicaltrials.gov/ct2/show/NCT02433639. Accessed 17 April 2017
Fang J, Nakamura H, Maeda H (2011) The EPR effect: unique features of tumor blood vessels for drug delivery, factors involved, and limitations and augmentation of the effect. Adv Drug Deliv Rev 63(3):136–151. https://doi.org/10.1016/j.addr.2010.04.009
Doktorovova S, Souto EB, Silva AM (2014) Nanotoxicology applied to solid lipid nanoparticles and nanostructured lipid carriers—a systematic review of in vitro data. Eur J Pharm Biopharm 87(1):871–818. https://doi.org/10.1016/j.ejpb.2014.02.005
Guise CP, Mowday AM, Ashoorzadeh A, Yuan R, Lin WH, DH W et al (2014) Bioreductive prodrugs as cancer therapeutics: targeting tumor hypoxia. Chin J Cancer 33(2):80–86. https://doi.org/10.5732/cjc.012.10285
International Conference on Harmonization (ICH) (1996) Guidance for industry Q1B. Photostability testing of new drug substances and products. Geneva, pp 1–14
Jencks WP, Carriuolo J (1960) Reactivity of nucleophilic reagents toward esters. J Am Chem Soc 82(7):1778–1786. https://doi.org/10.1021/ja01492a058
Konopleva M, Thall PF, Yi CA, Borthakur G, Coveler A, Bueso-Ramos C, Benito J, Konoplev S, Gu Y, Ravandi F, Jabbour E, Faderl S, Thomas D, Cortes J, Kadia T, Kornblau S, Daver N, Pemmaraju N, Nguyen HQ, Feliu J, Lu H, Wei C, Wilson WR, Melink TJ, Gutheil JC, Andreeff M, Estey EH, Kantarjian H (2015) Phase I/II study of the hypoxia-activated prodrug PR104 in refractrory/relapsed acute myeloid leukemia and acute lymphoblastic leukemia. Haematologica 100(7):927–934. https://doi.org/10.3324/haematol.2014.118455
Li X, Sloat BR, Yanasarn N, Cui Z (2011) Relationship between the size of nanoparticles and their adjuvant activity: data from a study with an improved experimental design. Eur J Pharm Biopharm 78(1):107–116. https://doi.org/10.1016/j.ejpb.2010.12.017
Lopes MS, Sena CFA, Silva BL, de Souza CM, Ramos JP, Cassali GD et al (2015) Synthesis of nitroaromatic compounds as potential anticancer agents. Anti Cancer Agents Med Chem 15(2):206–216. https://doi.org/10.2174/1871520614666141114201749
Mehenert W, Mader K (2001) Solid lipid nanoparticles, production, characterization and applications. Adv Drug Deliv 47(2-3):165–196. https://doi.org/10.1016/S0169-409X(01)00105-3
Muller RH, Mader K, Gohla S (2000) Solid lipid nanoparticles (SLN) for controlled drug delivery—a review of the state of the art. Eur J Pharm Biopharm 50(1):161–177. https://doi.org/10.1016/S0939-6411(00)00087-4
Nudelman A, Gnizia E, Katza Y, Azulaia R, Cohen-Ohana M, Zhuka Z et al (2001) Prodrugs of butyric acid. Novel derivatives possessing increased aqueous solubility and potential for treating cancer and blood diseases. Eur J Med Chem 36(1):63–74. https://doi.org/10.1016/S0223-5234(00)01199-5
Pardeike J, Hommoss A, Müller RH (2009) Lipid nanoparticles (SLN, NLC) in cosmetic and pharmaceutical dermal products. Int J Pharm 366(1-2):170–184. https://doi.org/10.1016/j.ijpharm.2008.10.003
Prabhakaran P, Hassiotou F, Blancafort P, Filgueira L (2013) Cisplatin induces differentiation of breast cancer cells. Front Oncol 3:1–10. https://doi.org/10.3389/fonc.2013.00134
QikProp 4.6 User Manual (2016) Schrödinger. LCC, New York
Santini V, Gozzini A, Scarppini B, Grossi A, Ferrini PR (2001) Searching for the magic bullet against cancer: the butyrate saga. Leuk Lymphoma 42(3):275–289. https://doi.org/10.3109/10428190109064584
Selvamuthukumar S, Velmurugan R (2012) Nanostructured lipid carriers: a potential drug carrier for cancer chemotherapy. Lipids Health Dis 11(1):1–8. https://doi.org/10.1186/1476-511X-11-159
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A (2015) Global cancer statistics, 2012. CA Cancer J Clin 65(2):87–108. https://doi.org/10.3322/caac.21262
USP 36-NF 34 (2016) The United States Pharmacopeia and National Formulary. United States Pharmacopeial Convention, Rockville
Üner M, Yener G (2007) Importance of solid lipid nanoparticles (SLN) in various administration routes and future perspectives. Int J Nanomedicine 2(3):289–300
Vichai V, Kirtikara K (2006) Sulforhodamine B colorimetric assay for cytotoxicity screening. Nat Protoc 1(3):1112−1116–1112−1116. https://doi.org/10.1038/nprot.2006.179
Waite CL, Roth CM (2012) Nanoscale drug delivery systems for enhanced drug penetration into solid tumors: current progress and opportunities. Crit Rev Biomed Eng 40(1):21–41. https://doi.org/10.1615/CritRevBiomedEng.v40.i1.20
Zhao S, Yang X, Garamus VM, Handge UA, Berengere L, Zhao L et al (2014) Mixture of nonionic/ionic surfactants for the formulation of nanostructured lipid carriers: effects on physical properties. Langmuir 30(23):6920–6928. https://doi.org/10.1021/la501141m
Acknowledgments
The authors are grateful to Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for the financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Sena, C.F.A., Apolinário, L.S., Duarte, J.A. et al. Preformulation and formulation development of a bioactive nitroaromatic compound. J Nanopart Res 19, 379 (2017). https://doi.org/10.1007/s11051-017-4073-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-017-4073-6