Abstract
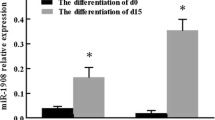
MicroRNAs (miRNAs) are short non-coding RNAs that are involved in post-transcriptional regulation of gene expression. Hsa-miR-26b is an intronic miRNA located in the intron of CTDSP1 (carboxy-terminal domain, RNA polymerase II, polypeptide A, small phosphatase 1). In the present study, the expression of hsa-miR-26b was examined during human preadipocyte differentiation. 15 days after induction of differentiation, mature human adipocytes were treated with adipokines. Hsa-miR-26b was differentially expressed during human preadipocyte differentiation. Tumor necrosis factor-α (TNF-α), leptin and resistin, but not interleukin-6, caused downregulation of hsa-miR-26b expression in adipocytes. These results suggest that the expression of hsa-miR-26b is affected by TNF-α, leptin and resistin and that hsa-miR-26b may be an important mediator in regulating the obesity-related insulin sensitivity and inflammatory responses.





Similar content being viewed by others
References
Jeffery RW, Sherwood NE (2008) Is the obesity epidemic exaggerated? No. BMJ 7638(336):245
Haslam DW, James WP (2005) Obesity. Lancet 9492(366):1197–1209
Galic S, Oakhill JS, Steinberg GR (2010) Adipose tissue as an endocrine organ. Mol Cell Endocrinol 2(316):129–139
Rosen ED, Spiegelman BM (2006) Adipocytes as regulators of energy balance and glucose homeostasis. Nature 7121(444):847–853
Wellen KE, Hotamisligil GS (2003) Obesity-induced inflammatory changes in adipose tissue. J Clin Invest 12(112):1785–1788
Trayhurn P, Wang B, Wood IS (2008) Hypoxia in adipose tissue: a basis for the dysregulation of tissue function in obesity? Br J Nutr 2(100):227–235
Esau C, Kang X, Peralta E, Hanson E, Marcusson EG, Ravichandran LV et al (2004) MicroRNA-143 regulates adipocyte differentiation. J Biol Chem 50(279):52361–52365
Wang Q, Li YC, Wang J, Kong J, Qi Y, Quigg RJ et al (2008) miR-17-92 cluster accelerates adipocyte differentiation by negatively regulating tumor-suppressor Rb2/p130. Proc Natl Acad Sci USA 8(105):2889–2894
Arora H, Qureshi R, Park AK, Park WY (2011) Coordinated regulation of ATF2 by miR-26b in gamma-irradiated lung cancer cells. PLoS ONE 8(6):e23802
Liu XX, Li XJ, Zhang B, Liang YJ, Zhou CX, Cao DX et al (2011) MicroRNA-26b is underexpressed in human breast cancer and induces cell apoptosis by targeting SLC7A11. FEBS Lett 9(585):1363–1367
Wu N, Zhao X, Liu M, Liu H, Yao W, Zhang Y et al (2011) Role of microRNA-26b in glioma development and its mediated regulation on EphA2. PLoS ONE 1(6):e16264
Teleman AA, Maitra S, Cohen SM (2006) Drosophila lacking microRNA miR-278 are defective in energy homeostasis. Genes Dev 4(20):417–422
Baroukh N, Ravier MA, Loder MK, Hill EV, Bounacer A, Scharfmann R et al (2007) MicroRNA-124a regulates Foxa2 expression and intracellular signaling in pancreatic beta-cell lines. J Biol Chem 27(282):19575–19588
Lovis P, Gattesco S, Regazzi R (2008) Regulation of the expression of components of the exocytotic machinery of insulin-secreting cells by microRNAs. Biol Chem 3(389):305–312
**e H, Lim B, Lodish HF (2009) MicroRNAs induced during adipogenesis that accelerate fat cell development are downregulated in obesity. Diabetes 5(58):1050–1057
Dill H, Linder B, Fehr A, Fischer U (2012) Intronic miR-26b controls neuronal differentiation by repressing its host transcript, ctdsp2. Genes Dev 1(26):25–30
Kloting N, Berthold S, Kovacs P, Schon MR, Fasshauer M, Ruschke K et al (2009) MicroRNA expression in human omental and subcutaneous adipose tissue. PLoS ONE 3(4):e4699
Kajimoto K, Naraba H, Iwai N (2006) MicroRNA and 3T3-L1 pre-adipocyte differentiation. RNA 9(12):1626–1632
Neville MJ, Collins JM, Gloyn AL, McCarthy MI, Karpe F (2011) Comprehensive human adipose tissue mRNA and microRNA endogenous control selection for quantitative real-time-PCR normalization. Obesity (Silver Spring) 4(19):888–892
Olefsky JM, Glass CK (2010) Macrophages, inflammation, and insulin resistance. Annu Rev Physiol 72:219–246
Jellema A, Plat J, Mensink RP (2004) Weight reduction, but not a moderate intake of fish oil, lowers concentrations of inflammatory markers and PAI-1 antigen in obese men during the fasting and postprandial state. Eur J Clin Invest 11(34):766–773
Rui L, Aguirre V, Kim JK, Shulman GI, Lee A, Corbould A et al (2001) Insulin/IGF-1 and TNF-alpha stimulate phosphorylation of IRS-1 at inhibitory Ser307 via distinct pathways. J Clin Invest 2(107):181–189
Bastard JP, Maachi M, Lagathu C, Kim MJ, Caron M, Vidal H et al (2006) Recent advances in the relationship between obesity, inflammation, and insulin resistance. Eur Cytokine Netw 1(17):4–12
Pickup JC, Mattock MB, Chusney GD, Burt D (1997) NIDDM as a disease of the innate immune system: association of acute-phase reactants and interleukin-6 with metabolic syndrome X. Diabetologia 11(40):1286–1292
Kern PA, Ranganathan S, Li C, Wood L, Ranganathan G (2001) Adipose tissue tumor necrosis factor and interleukin-6 expression in human obesity and insulin resistance. Am J Physiol Endocrinol Metab 5(280):E745–E751
Pedersen BK, Febbraio MA (2007) Point: interleukin-6 does have a beneficial role in insulin sensitivity and glucose homeostasis. J Appl Physiol 2(102):814–816
Mooney RA (2007) Counterpoint: interleukin-6 does not have a beneficial role in insulin sensitivity and glucose homeostasis. J Appl Physiol 102(2):816–818; discussion 818–819
Stefan N, Fritsche A, Haring H, Stumvoll M (2001) Acute stimulation of leptin concentrations in humans during hyperglycemic hyperinsulinemia. Influence of free fatty acids and fasting. Int J Obes Relat Metab Disord 1(25):138–142
Ahima RS, Saper CB, Flier JS, Elmquist JK (2000) Leptin regulation of neuroendocrine systems. Front Neuroendocrinol 3(21):263–307
Loffreda S, Yang SQ, Lin HZ, Karp CL, Brengman ML, Wang DJ et al (1998) Leptin regulates proinflammatory immune responses. Faseb J 1(12):57–65
Steppan CM, Bailey ST, Bhat S, Brown EJ, Banerjee RR, Wright CM et al (2001) The hormone resistin links obesity to diabetes. Nature 6818(409):307–312
Burnett MS, Devaney JM, Adenika RJ, Lindsay R, Howard BV (2006) Cross-sectional associations of resistin, coronary heart disease, and insulin resistance. J Clin Endocrinol Metab 1(91):64–68
Sentinelli F, Romeo S, Arca M, Filippi E, Leonetti F, Banchieri M et al (2002) Human resistin gene, obesity, and type 2 diabetes: mutation analysis and population study. Diabetes 3(51):860–862
Nagaev I, Bokarewa M, Tarkowski A, Smith U (2006) Human resistin is a systemic immune-derived proinflammatory cytokine targeting both leukocytes and adipocytes. PLoS ONE 1:e31
Lehrke M, Reilly MP, Millington SC, Iqbal N, Rader DJ, Lazar MA (2004) An inflammatory cascade leading to hyperresistinemia in humans. PLoS Med 2(1):e45
Acknowledgments
This project was supported by grants from the National Key Basic Research Program of China (2013CB530604), the National Natural Science Foundation of China (No. 81170797), the Natural Science Foundation of Jiangsu Province China (BK2011107), the Program for Innovative Research Teams of Jiangsu Province (LJ201108), and Nan**g Technological Development Program (201104013).
Disclosures
No competing financial interests exist.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Guangfeng Xu and Chenbo Ji contributed equally to this study.
Rights and permissions
About this article
Cite this article
Xu, G., Ji, C., Shi, C. et al. Modulation of hsa-miR-26b levels following adipokine stimulation. Mol Biol Rep 40, 3577–3582 (2013). https://doi.org/10.1007/s11033-012-2431-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-012-2431-0