Abstract
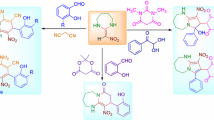
A series of novel and diverse diaryl[d,f][1,3]diazepines were designed and synthesized to expand the pharmaceutical utility of the [6,7]bicyclic molecular skeletons. The facile synthesis involved two key steps: a one-pot Suzuki coupling to construct the bi-aryl intermediates from corresponding halides, and a ring closure by direct condensation with carboxylic acids.
Similar content being viewed by others
References
Antonow D, Thurston DE (2011) Synthesis of DNA-interactive pyrrolo[2,1-c][1,4]benzodiazepines (PBDs). Chem Rev 111: 2815–2864. doi:10.1021/cr100120f
PDR Staff (2011) Physicians’ desk reference, 65th edn. PDR Network, Montvale
Cipolla L, Araujo AC, Airoldi C, Bini D (2009) Pyrrolo[2,1-c][1,4]benzodiazepine as a scaffold for the design and synthesis of anti-tumour drugs. Anti-Cancer Agents Med Chem 9: 1–31. doi:10.2174/187152009787047743
Geyer HM, Martin LL, Crichlow CA, Dekow FW, Ellis DB, Kruse H, Setescak LL, Worms M (1982) (±)-4-Ary1-4,5-dihydro-3H-l,3-benzodiazepines. 1. Synthesis and evaluation of (±)-4,5-dihydro-2,3-dimethyl-4-phenyl-3H-1,3-benzodiazepine and analogues as potential antidepressant agents. J Med Chem 25: 340–346. doi:10.1021/jm00346a003
Nichols PL, Skidmore J, Ward RW, Wilson DM (2008) 5-Phenyl-1,3,4-oxadiazol-2-yl-acetyl-4-piperidinyl derivatives as CGRP receptor antagonists. Patent WO2009000819
Zhu Z, Sun ZY, Ye Y, McKittrick B, Greenlee W, Czarniecki M, Fawzi A, Zhang H, Lachowicz JE (2009) Design and discovery of 1,3-benzodiazepines as novel dopamine antagonists. Bioorg Med Chem Lett 19: 5218–5221. doi:10.1016/j.bmcl.2009.07.012
Takayama T, Umemiya H, Amada H, Yabuuchi T, Koami T, Shiozawa F, Oka Y, Takaoka A, Yamaguchi A, Endo M, Sato M (2010) Ring-fused pyrazole derivatives as potent inhibitors of lymphocyte-specific kinase (Lck): structure, synthesis, and SAR. Bioorg Med Chem Lett 20: 112–116. doi:10.1016/j.bmcl.2009.11.013
Rotas G, Natchkebia K, Natsvlishvil N, Kekelidze M, Kimbaris A, Varvounis G, Mikeladze D (2005) Action of a novel pyrrolo[1,2-c][1.3]benzodiazepine on the viability of Jurkat and neuronal/glial cells. Bioorg Med Chem Lett 20: 3220–3223. doi:10.1016/j.bmcl.2005.05.006
Rotas G, Kimbaris A, Varvounis G (2011) Synthesis of a novel pyrrolo[1,2-c][1.3] benzodiazepine analogue of VPA-985. Tetrahedron 67: 7805–7810. doi:10.1016/j.tet.2011.07.083
Fukamachi S, Kobayashi A, Konishi H, Kobayashi K (2010) A convenient synthesis of new types of benzodiazepine derivatives: 2-alkylsulfanyl-3H-4,5-dihydro-1,3-benzodiazepin-4-ones and 2-alkylsulfanyl-3H-4,5-dihydro-1,3-benzodiazepine-4-thiones. Synthesis 288–292. doi:10.1055/s-0029-1217100
Dengiz C, Ozcan S, Sahin E, Balci M (2010) New synthetic methodology for construction of the 1,3,4,5-tetrahydro-2H-1,3-benzodiazepin-2-one skeleton. Synthesis 1365–1370. doi:10.1055/s-0029-1218673
Ried W, Sinharay A (1964) Note for the ring closure reaction of o,o-diamino-biphenyl. Chem Ber 97: 1214–1215
Ried W, Sinharay A (1965) More for the ring closure reaction of o,o-diamino-biphenyl. Chem Ber 98: 3523–3531
Kaczmarek L, Nantka-Namirski P (1990) Synthesis of N-monosubstituted-3,3′diamines. Monatsh Chem 121: 821–828. doi:10.1007/BF00808375
Matsuda K, Yanagisawa I, Isomura Y, Mase T, Shibanuma T (1997) Alternative synthesis of dibenzo- and dipyrido-[1,3]diazepines from thioamides and o,o′-diaminobiaryls. Synth Commun 27: 2393–2402. doi:10.1080/00397919708004101
Trzewik B, Ciez D, Hodorowicz M, Stadnicka K (2008) Newα-amido-α-aminonitrones as building blocks for constructing heterocyclic systems. Synthesis 18: 2977–2985. doi:10.1055/s-2008-1067236
Zaleska B, Karelus M, Trzewik B, Serda P (2007) A new way to quinazolines, perimidines and dibenzo[d,f][1,3]diazepines. J Chem Res 4: 195–199. doi:10.3184/030823407X203387
Maddaford SP, Keay BA (1994) Scope and limitations of the palladium-catalyzed cross-coupling reaction of in situ generated organoboranes with aryl and vinyl halides. J Org Chem 59: 6501–6503. doi:10.1021/jo00101a001
Baudoin O, Guenard D, Gueritte F (2000) Palladium-catalyzed borylation of ortho-substituted phenyl halides and application to the one-pot synthesis of 2,2′-disubstituted biphenyls. J Org Chem 65: 9268–9271. doi:10.1021/jo005663d
Broutin PE, Cerna I, Campaniello M, Leroux F, Colobert F (2004) Palladium-catalyzed borylation of phenyl bromides and application in one-pot Suzuki–Miyaura biphenyl synthesis. Org Lett 6: 4419–4422. doi:10.1021/ol048303b
Brown SD, Armstrong RW (1996) Synthesis of tetrasubstituted ethylenes on solid support via resin capture. J Am Chem Soc 118: 6331–6332. doi:10.1021/ja961203j
Akkaoui AE, Berteina-Raboin S, Mouaddib A, Guillaumet G (2010) Direct arylation of imidazo[1,2-b]pyridazines: microwave-assisted one-pot Suzuki coupling/Pd-catalysed arylation. Eur J Org Chem 862–871 doi:10.1002/ejoc.200900849
Lam KC, Marder TB, Lin Z (2010) Mechanism of the palladium-catalyzed borylation of aryl halides with pinacolborane. Organometallics 29: 1849–1857. doi:10.1021/om9010802
Yamamoto T, Morita T, Takagi J, Yamakawa T (2011) NiCl2(PMe3)2-catalyzed borylation of aryl chlorides. Org Lett 13: 5766–5769. doi:10.1021/ol202267t
Slevin A, Koolmeister T, Scobie M (2007) A versatile synthesis of diverse 3,4-fused cinnolines via the base catalysed condensation of 2-amino-2′-nitrobiaryls. Chem Commun 24: 2506–2508. doi:10.1039/B618318B
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
The Below is the Electronic Supplementary Material.
Rights and permissions
About this article
Cite this article
Yan, L., Che, X., Bai, X. et al. Syntheses of novel diaryl[d,f ][1,3]diazepines via one-pot Suzuki coupling followed by direct ring closure with carboxylic acids. Mol Divers 16, 489–501 (2012). https://doi.org/10.1007/s11030-012-9382-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11030-012-9382-1