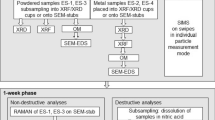
Abstract
The Israeli National Nuclear Forensics Laboratory (INNFL) investigated four unknown samples during the Collaborative Material Exercise 7 (CMX-7). The samples, identified as depleted uranium, were marked ES-1 to ES-4. ES-2 isotopic composition (0.200 ± 0.004% U-235) was different from the other samples (0.300 ± 0.003% U-235). ES-2 phase exhibited characteristics similar to metallic crystalline uranium with its appearance and assay. However, its density (16.9 ± 1.7 g/cm3) deviated significantly from metallic uranium (19.1 g/cm3). A series of analyses indicated that vanadium was present (app. 3000 ppm) in porosive areas on ES-2 surface. The oxygen isotope ratios suggested thermal processing under environmental conditions.










Similar content being viewed by others
References
Brandis M, Gershinsky G, Bolker A et al (2021) Morphological and chemical characterization of uranium and cerium nuclear forensics samples. J Nucl Mater 555:153109. https://doi.org/10.1016/j.jnucmat.2021.153109
Schwantes JM, Marsden O, Pellegrini KL (2017) State of practice and emerging application of analytical techniques of nuclear forensic analysis: highlights from the 4th collaborative materials exercise of the nuclear forensics international technical working group (ITWG). J Radioanal Nucl Chem 311:1441–1452. https://doi.org/10.1007/s10967-016-5037-5
Jovanovic SV, Weber PK, Pidduck AJ et al (2020) Uncovering uranium isotopic heterogeneity of fuel pellets from the fifth collaborative materials exercise of the nuclear forensics international technical working group. J Radioanal Nucl Chem 326:1853–1866. https://doi.org/10.1007/s10967-020-07470-5
Taylor F, Higginson M, Marsden O, Schwantes J (2020) State of practice and emerging application of analytical techniques of nuclear forensic analysis: highlights from the 5th collaborative materials exercise of the nuclear forensics international technical working group (ITWG). J Radioanal Nucl Chem 323:415–430. https://doi.org/10.1007/s10967-019-06950-7
Schwantes JM, Corbey JF, Marsden O (2022) Exercise Celestial Skónis: Part 2 – emerging technologies and State of Practice of nuclear forensic analyses demonstrated during the 6th collaborative materials exercise of the nuclear forensics International Technical Working Group. Forensic Chem 29:100423. https://doi.org/10.1016/j.forc.2022.100423
Assulin M, Yam R, Brandis M et al (2021) Oxygen isotopes of fuel pellets from the fifth collaborative materials exercise and uranium oxides reference materials determined by continuous flow laser fluorination mass spectrometry for nuclear forensic applications. J Radioanal Nucl Chem 329:757–768. https://doi.org/10.1007/s10967-021-07858-x
Lander GH, Mueller MH (1970) Neutron diffraction study of α-uranium at low temperatures. Acta Cryst B 26:129–136. https://doi.org/10.1107/S0567740870002066
Barrett CS, Mueller MH, Hitterman RL (1963) Crystal structure variations in alpha uranium at low temperatures. Phys Rev 129:625–629. https://doi.org/10.1103/PhysRev.129.625
Choi CS, Prask HJ (1985) Neutron powder diffraction studies of two uranium–0.75 wt% titanium alloys. J Appl Cryst 18:141–144. https://doi.org/10.1107/S0021889885009992
Assulin M, Yam R, Kossoy A et al (2022) Equilibrium fractionation of oxygen isotopes in the U3O8–atmospheric oxygen system. ACS Omega 7:47040–47047. https://doi.org/10.1021/acsomega.2c06148
Assulin M, Yam R, Elish E, Shemesh A (2022) Oxygen isotopic composition of U3O8 synthesized from U metal, uranyl nitrate hydrate, and UO3 as a signature for nuclear forensics. ACS Omega 7:7973–7980. https://doi.org/10.1021/acsomega.1c07042
Oerter EJ, Singleton M, Pili E et al (2022) The oxygen stable isotope composition of CRM 125-A UO2 standard reference material. Appl Geochem 146:105470. https://doi.org/10.1016/j.apgeochem.2022.105470
Bowman AL, Arnold GP, Witteman WG et al (1966) The crystal structure of UC2. Acta Cryst 21:670–671. https://doi.org/10.1107/S0365110X66003670
Rundle RE, Baenziger NC, Wilson AS, McDonald RA (1948) The Structures of the carbides, nitrides and oxides of uranium1. J Am Chem Soc 70:99–105
Bourgeois L, Nadal M-H, Clément F, Ravel-Chapuis G (2007) Determination of elastic moduli at high temperatures foruranium–vanadium alloy and pure plutonium by an ultrasonic method. J Alloy Compd 444–445:261–264. https://doi.org/10.1016/j.jallcom.2006.10.092
Berche A, Alpettaz T, Chatain S et al (2011) Thermodynamic study of theuranium–vanadium system. J Chem Thermodyn 43:458–466. https://doi.org/10.1016/j.jct.2010.10.023
Sunwoo A (1997) Weldability of cast and heat treated uranium-0.25% vanadium alloy. Scripta Mater 37:691–697. https://doi.org/10.1016/S1359-6462(97)00151-6
Staker MR (1998) Theuranium–vanadium equilibrium phase diagram. J Alloy Compd 266:167–179. https://doi.org/10.1016/S0925-8388(97)00454-4
Staker MR (2004) Stakalloy: a uranium-vanadium-niobium alloy, US patent US76726876B1
Acknowledgements
The authors would like to thank the ITWG organization, specifically Olivia Marsden and Jon Schwantes, for their efforts in creating the CMX7 exercise.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No funds, grants, or other support was received for the submitted work or preparation of the manuscript. The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Brandis, M., Gershinsky, G., Rubinshtein, A. et al. Phase investigation of uranium–vanadium alloy using multiple techniques. J Radioanal Nucl Chem (2023). https://doi.org/10.1007/s10967-023-09200-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10967-023-09200-z