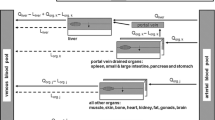
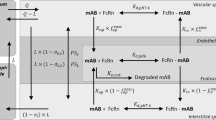
Abstract
Monoclonal antibodies, endogenous IgG, and serum albumin bind to FcRn in the endosome for salvaging and recycling after pinocytotic uptake, which prolongs their half-life. This mechanism has been broadly recognized and is incorporated in currently available PBPK models. Newer types of large molecules have been designed and developed, which also bind to FcRn in the plasma space for various mechanistic reasons. To incorporate FcRn binding affinity in PBPK models, binding in the plasma space and subsequent internalisation into the endosome needs to be explicitly represented. This study investigates the large molecules model in PK-Sim® and its applicability to molecules with FcRn binding affinity in plasma. With this purpose, simulations of biologicals with and without plasma binding to FcRn were performed with the large molecule model in PK-Sim®. Subsequently, this model was extended to ensure a more mechanistic description of the internalisation of FcRn and the FcRn-drug complexes. Finally, the newly developed model was used in simulations to explore the sensitivity for FcRn binding in the plasma space, and it was fitted to an in vivo dataset of wild-type IgG and FcRn inhibitor plasma concentrations in Tg32 mice. The extended model demonstrated a strongly increased sensitivity of the terminal half-life towards the plasma FcRn binding affinity and could successfully fit the in vivo dataset in Tg32 mice with meaningful parameter estimates.








Similar content being viewed by others
References
Chaudhury C, Brooks CL, Carter DC et al (2006) Albumin binding to FcRn: distinct from the FcRn-IgG interaction. Biochemistry 45:4983–4990. https://doi.org/10.1021/bi052628y
Abdiche YN, Yeung YA, Chaparro-Riggers J et al (2015) The neonatal Fc receptor (FcRn) binds independently to both sites of the IgG homodimer with identical affinity. MAbs 7:331–343. https://doi.org/10.1080/19420862.2015.1008353
Igawa T, Maeda A, Haraya K et al (2013) Engineered monoclonal antibody with novel antigen-swee** activity in vivo. PLOS ONE 8:e63236. https://doi.org/10.1371/journal.pone.0063236
Niederalt C, Kuepfer L, Solodenko J et al (2018) A generic whole body physiologically based pharmacokinetic model for therapeutic proteins in PK-Sim. J Pharmacokinet Pharmacodyn 45:235–257. https://doi.org/10.1007/s10928-017-9559-4
Lee C-H, Kang TH, Godon O et al (2019) An engineered human Fc domain that behaves like a pH-toggle switch for ultra-long circulation persistence. Nat Commun 10:5031. https://doi.org/10.1038/s41467-019-13108-2
Mackness B, Qiu H (2021) Methods of treating antibody-mediated disorders with FcRn antagonists. https://patents.google.com/patent/US20210024620A1/en
Mackness BC, Jaworski JA, Boudanova E et al (2019) Antibody Fc engineering for enhanced neonatal Fc receptor binding and prolonged circulation half-life. MAbs 11:1276–1288. https://doi.org/10.1080/19420862.2019.1633883
Andersen JT, Daba MB, Berntzen G et al (2010) Cross-species binding analyses of mouse and human neonatal Fc receptor show dramatic differences in immunoglobulin G and albumin binding. J Biol Chem 285:4826–4836. https://doi.org/10.1074/jbc.M109.081828
Li T, Balthasar JP (2018) FcRn expression in wildtype mice, transgenic mice, and in human tissues. Biomolecules 8:115. https://doi.org/10.3390/biom8040115
Fan Y-Y, Avery LB, Wang M et al (2016) Tissue expression profile of human neonatal Fc receptor (FcRn) in Tg32 transgenic mice. MAbs 8:848–853. https://doi.org/10.1080/19420862.2016.1178436
Roopenian DC, Low BE, Christianson GJ et al (2015) Albumin-deficient mouse models for studying metabolism of human albumin and pharmacokinetics of albumin-based drugs. mAbs 7:344–351. https://doi.org/10.1080/19420862.2015.1008345
Wong H, Chow TW (2017) Physiologically based pharmacokinetic modeling of therapeutic proteins. JPharmSci 106:2270–2275. https://doi.org/10.1016/j.xphs.2017.03.038
Chetty M, Li L, Rose R et al (2015) Prediction of the pharmacokinetics, pharmacodynamics, and efficacy of a monoclonal antibody, using a physiologically based pharmacokinetic FcRn model. Front Immunol. https://doi.org/10.3389/fimmu.2014.00670
Shah DK, Betts AM (2012) Towards a platform PBPK model to characterize the plasma and tissue disposition of monoclonal antibodies in preclinical species and human. J Pharmacokinet Pharmacodyn 39:67–86. https://doi.org/10.1007/s10928-011-9232-2
Li T, Balthasar JP (2019) Application of physiologically based pharmacokinetic modeling to predict the effects of FcRn inhibitors in mice, rats, and monkeys. J Pharm Sci 108:701–713. https://doi.org/10.1016/j.xphs.2018.10.065
Robbie GJ, Criste R, Dall’Acqua WF, et al (2013) A novel investigational Fc-modified humanized monoclonal antibody, motavizumab-YTE, has an extended half-life in healthy adults. Antimicrob Agents Chemother 57:6147–6153. https://doi.org/10.1128/AAC.01285-13
Valente D, Mauriac C, Schmidt T et al (2020) Pharmacokinetics of novel Fc-engineered monoclonal and multispecific antibodies in cynomolgus monkeys and humanized FcRn transgenic mouse models. MAbs 12:1829337. https://doi.org/10.1080/19420862.2020.1829337
Avery LB, Wang M, Kavosi MS et al (2016) Utility of a human FcRn transgenic mouse model in drug discovery for early assessment and prediction of human pharmacokinetics of monoclonal antibodies. mAbs 8:1064–1078. https://doi.org/10.1080/19420862.2016.1193660
Malik PRV, Hamadeh A, Edginton AN (2022) Model-based assessment of the contribution of monocytes and macrophages to the pharmacokinetics of monoclonal antibodies. Pharm Res 39:239–250. https://doi.org/10.1007/s11095-022-03177-2
Salerno SN, Deng R, Kakkar T (2022) Physiologically-based pharmacokinetic modeling of immunoglobulin and antibody coadministration in patients with primary human immunodeficiency. CPT: Pharmacometrics Syst Pharmacol 11:1316–1327. https://doi.org/10.1002/psp4.12847
Yang D, Giragossian C, Castellano S et al (2017) Maximizing in vivo target clearance by design of pH-dependent target binding antibodies with altered affinity to FcRn. MAbs 9:1105–1117. https://doi.org/10.1080/19420862.2017.1359455
Hardiansyah D, Ng CM (2018) Minimal physiologically-based pharmacokinetic model to investigate the effect of pH dependent FcRn affinity and the endothelial endocytosis on the pharmacokinetics of anti-VEGF humanized IgG1 antibody in cynomolgus monkey. Eur J Pharm Sci 125:130–141. https://doi.org/10.1016/j.ejps.2018.09.015
Li T, Balthasar JP (2019) Development and evaluation of a physiologically based pharmacokinetic model for predicting the effects of anti-FcRn therapy on the disposition of endogenous IgG in humans. J Pharm Sci 108:714–724. https://doi.org/10.1016/j.xphs.2018.10.067
Acknowledgements
All authors are employees of Sanofi Research & Development. The authors are grateful to the following individuals for their contributions to this work: Liduo Shen, Sarah Nzerko, Wei Sun, Jennifer Fretland, Renee Bodinizzo, Megan Pike, Erik Zarazinski, Ekaterina Boudanova, Julie A. Jaworski and Veronique De Brabandere.
Funding
The authors did not receive support from any organization for the submitted work.
Author information
Authors and Affiliations
Contributions
WdW developed the model. WdW and MS wrote and supervised the main manuscript and prepared the figures. TvB advised on the model development and manuscript preparation. BM, LA, and AP prepared, designed and supervised the in vitro and in vivo experiments. All authors reviewed the manuscript
Corresponding author
Ethics declarations
Competing interest
All authors were employed at Sanofi at the time of this study. WdW is currently employed at esqLABS GmbH.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
de Witte, W.E.A., Avery, L.B., Mackness, B.C. et al. Mechanistic incorporation of FcRn binding in plasma and endosomes in a whole body PBPK model for large molecules. J Pharmacokinet Pharmacodyn 50, 229–241 (2023). https://doi.org/10.1007/s10928-023-09849-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10928-023-09849-9