Abstract
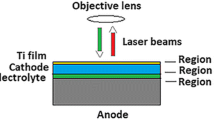
A thin-film calorimeter has been developed to investigate the thermodynamic properties of thin films including battery layer sequences. A new approach, i.e., the application of high-temperature stable piezoelectric resonators as highly sensitive planar temperature sensor, is chosen. Thin films with a thickness of several micrometers of the material of interest are deposited on the resonators. The production or consumption of latent heat by the active layer(s) results in temperature fluctuations with respect to surroundings, in our case the furnace in which the sensor is placed. The temperature fluctuations can be easily monitored in situ via changes of the resonance frequency of the resonator. This enables us to extract the temperature and time dependence of phase transformations as well as the associated enthalpies. To cover a temperature range from −20 to 1000 °C, high-temperature stable piezoelectric langasite (La3Ga5SiO14) resonators are applied. Initially, aluminum and tin layers are used to test the calorimeter. The temperature and enthalpy of the solid–liquid phase transformation agree well with the literature data. Further, the thermodynamic data of the battery materials to be used as cathode, solid electrolyte, and anode in lithium ion batteries are investigated by the newly developed method. The cathode materials Li(Ni0.8Co0.15Al0.05)O2-δ (NCA) and LiMn2O4-δ (LMO) are amorphous after deposition and crystallize during heating. NCA shows this transformation at 455 °C with an enthalpy of −4.8 J/g. LMO undergoes three phase transformations at 330, 410 and 600 °C. They require initially an activation which is followed by an exothermic enthalpy. The associated energies (activation; enthalpy) are (+67.2; −50.2) J/g, (+29.3; −29.3) J/g, and (+20.4; −26.2) J/g, respectively. The solid electrolyte Li3.4V0.6Si0.4O4-δ (LVSO) shows no phase transformation up to its decomposition at about 220 °C. The anode material molybdenum disulfide (MoS2) exhibits a phase transformation at 480 °C with an enthalpy of −183.2 J/g.



















Similar content being viewed by others
References
Sun YK, Myung ST, Park BC, Prakash J, Belharouak I, Amine K (2009) Nat Mater 8:320
Joho F, Novák P, Spahr ME (2002) J Electrochem Soc 149(8):A1020
Maleki H, Al Hallaj S, Selman JR, Dinwiddie RB, Wang H (1999) J Electrochem Soc 146(3):947
Balaya P (2008) Energy Environ Sci 1:645
Chen X, Li C, Grätzel M, Kostecki R, Mao SS (2012) Chem Soc Rev 41:7909
Zhang Z, Fouchard D, Rea JR (1998) J Power Sources 70:16
Wakihara M (2001) Mater Sci Eng R33:109
Armand M, Tarascon JM (2008) Nature 451:652
Vetter J, Novák P, Wagner MR, Veit C, Möller KC, Besenhard JO, Winter M, Wohlfahrt-Mehrens M, Vogler C, Hammouche A (2005) J Power Sources 147:269
Vetter J, Winter M, Wohlfahrt-Mehrens M (2009) In: Garche J, Dyer CK, Moseley PT, Ogumi Z, Rand DAJ, Scrosati B (eds) Encyclopedia of Electrochemical Power Sources. Elsevier, Amsterdam
Amatucci GG, Pereira N, Zheng T, Tarascon JM (2001) J Electrochem Soc 148:A171
Palomares V, Rojo T (2012) In: Ilias Belharouak (Ed), Lithium Ion Batteries—New Developments, InTech, ISBN: 978-953-51-0077-5
Fritze H (2011) Meas Sci Technol 22:012002
Ohtsuka H, Yamaki J (1989) Jpn J Appl Phys 28:2264
Kuwata N, Kawamura J, Toribami K, Sata N, Hattori T (2004) Electrochem Commun 6(4):417
Kawamura J, Kuwata N, Toribami K, Sata N, Kamishima O, Hattori T (2004) Solid State Ion 175:273
Kuwata N, Kumar R, Toribami K, Suzuki T, Hattori T, Kawamura J (2006) Solid State Ion 177:2827
Kuwata N, Iwagami N, Kawamura J (2009) Solid State Ion 180:644
Whittingham MS (2004) Chem Rev 104:4271
Kostecki R, McLarnon F (2004) Electrochem Solid State Lett 7(10):A380
Fischer J, Adelhelm C, Bergfeldt T, Chang K, Ziebert C, Leiste H, Stüber M, Ulrich S, Music D, Hallstedt B, Seifert HJ (2012) Thin Solid Films 528:217
Ceder G, Chiang YM, Sadoway DR, Aydinol MK, Jang YI, Huang B (1998) Nature 392:694
Yoshio M, Noguchi H (2009) In: Yoshio M, Brodd RJ, Kozawa A (eds) Lithium-ion batteries: science and technologies. Springer, New York
Tarascon JM, Armand M (2001) Nature 414:359
Xu B, Fell CR, Chi M, Meng YS (2011) Energy Environ Sci 4:2223
Hy S, Su WN, Chen JM, Hwang BJ (2012) J Phys Chem C 116(48):25242
Ceder G, Mishra SK (1999) Electrochem Solid-State Lett 2(11):550
Feng C, Ma J, Li H, Zeng R, Guo Z, Liu H (2009) Mater Res Bull 44(9):1811
Li A, Liu H, Zhu Z, Huang M, Yang Y (2006) J Mater Sci Technol 22(1):40
Dominko R, Arcon D, Mrzel A, Zorko A, Cevc P, Venturini P, Gaberscek M, Remskar M, Mihailovic D (2002) Adv Mater 14(21):1531
Huggins RA (1999) J Power Sources 18–19:13
Scrosati B (2000) Electrochim Acta 45(5–16):2461
Hassoun J, Scrosati B (2010) Angew Chem 122:2421
Au M, McWhorter S, Ajo H, Adams T, Zhao Y, Gibbs J (2010) J Power Sources 195(10):3333
Hamon Y, Brousse T, Jousse F, Topart P, Buvat P, Schleich DM (2001) J Power Sources 97–98:185
Schneider T, Richter D, Doerner S, Fritze H, Hauptmann P (2005) Sens Actuators B 111–112:187
Albrecht D, Wulfmeier H, Ivanov S, Bund A, Fritze H (2013) MRS Proceedings 1496. doi:10.1557/opl.2013.126
Kong H, Wang J, Zhang H, Yin X, Zhang S, Liu Y, Cheng X, Gao L, Hu X, Jiang M (2003) J Cryst Growth 254:360
Lide DR (2003) CRC Handbook of Chemistry and Physics, 84th edn. CRC Press, Boca Raton
Fritze H, Tuller HL (2002) High-temperature balance. US Patent No 6 370 955
Wilthan B (2013) Status of round robin tests. Workshop of the GEFTA thermophysics working group, Dresden, March 18–19
Wojtczak L (1967) Phys Stat Sol 23:K163
Sauerbrey G (1959) Zeitschrift für Physik A 155:206
Yoon WS, Chung KY, McBreen J, Yang XQ (2006) Electrochem Comm 8:1257
JCPDS database, pdf card number 000271252
JCPDS database, pdf card number 000350782
Julien CM, Massot M (2003) Mater Sci Eng B97:217
Julien CM, Massot M (2003) Mater Sci Eng B100:69
Kuchling H (1978) Taschenbuch der Physik. Verlag Harri Deutsch, Thun
Hollemann AF, Wiberg N (2007) Lehrbuch der Anorganischen Chemie, 102nd edn. Walter de Gruyter, Berlin
Ravelo R, Baskes M (1997) Phys Rev Lett 79(13):2482
Wolf G, Schmidt H-G, Bohmhammel K (1994) Thermochim Acta 235:23
Acknowledgements
The authors gratefully thank the German Research Foundation (DFG) for the financial support within the priority program 1473 “WeNDeLIB”.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wulfmeier, H., Albrecht, D., Ivanov, S. et al. High-temperature thin-film calorimetry: a newly developed method applied to lithium ion battery materials. J Mater Sci 48, 6585–6596 (2013). https://doi.org/10.1007/s10853-013-7455-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-013-7455-x