Abstract
Purpose
The rates of cardiovascular implantable electronic device (CIED) implantations and cardiac ablation procedures are increasing worldwide. To date, the management of CIED lead thrombi in the peri-ablation period remains undefined and key clinical management questions remained unanswered. We sought to describe the clinical course and management strategies of patients with a CIED lead thrombus detected in the peri-ablative setting.
Methods
We performed a retrospective analysis of all patients who underwent a cardiac ablation procedure at Mayo Clinic Rochester from 2000 to 2014. Patients were included in our study cohort if they had documented CIED lead thrombus noted on peri-ablation imaging studies. Electronic medical records were reviewed to determine the overall management strategy, outcomes, and embolic complications in these patients.
Results
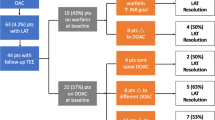
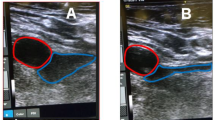
Our overall cohort included 1833 patients, with 27 (1.4 %) having both cardiac ablation procedures as well as CIED lead thrombus detected on imaging. Of these 27 patients, 21 were male (77 %), and the mean age was 59.2 years. The mean duration of follow-up was 16.5 months (range 3 days–48.3 months). Anticoagulation was an effective therapeutic strategy, with 11/14 (78.6 %) patients experiencing either resolution of the thrombus or reduction in size on re-imaging. For atrial fibrillation ablation, the most common management strategy was a deferment in ablation with initiation/intensification of anticoagulation medication. For ventricular tachycardia ablations, most procedures involved a modified approach with the use of a retrograde aortic approach to access the left ventricle. No patient had any documented embolic complications.
Conclusions
The incidence of lead thrombi in patients undergoing an ablation was small in our study cohort (1.4 %). Anticoagulation and deferral of ablation represented successful management strategies for atrial fibrillation ablation. For patients undergoing ventricular tachycardia ablation, a modified approach using retrograde aortic access to the ventricle was successful. In patients who are not on warfarin anticoagulation at the time of thrombus detection, we recommend initiation of this medication, with a goal INR of 2–3. For patients on warfarin at the time of thrombus detection, we recommend an intensification of anticoagulation with a goal INR of 3.0.


Similar content being viewed by others
References
Gadler, F., Valzania, C., & Linde, C. (2015). Current use of implantable electrical devices in Sweden: data from the Swedish pacemaker and implantable cardioverter-defibrillator registry. Europace, 17(1), 69–77.
Voigt, A., Shalaby, A., & Saba, S. (2010). Continued rise in rates of cardiovascular implantable electronic device infections in the United States: temporal trends and causative insights. Pacing and Clinical Electrophysiology, 33(4), 414–9.
Uslan, D. Z., et al. (2008). Temporal trends in permanent pacemaker implantation: a population-based study. American Heart Journal, 155(5), 896–903.
Zhan, C., et al. (2008). Cardiac device implantation in the United States from 1997 through 2004: a population-based analysis. Journal of General Internal Medicine, 23(Suppl 1), 13–9.
Boriani, G., et al. (2010). Implantable electrical devices for prevention of sudden cardiac death: data on implant rates from a ‘real world’ regional registry. Europace, 12(9), 1224–30.
van Veldhuisen, D. J., et al. (2009). Implementation of device therapy (cardiac resynchronization therapy and implantable cardioverter defibrillator) for patients with heart failure in Europe: changes from 2004 to 2008. European Journal of Heart Failure, 11(12), 1143–51.
Raatikainen, M. J., et al. (2015). Statistics on the use of cardiac electronic devices and electrophysiological procedures in the European Society of Cardiology countries: 2014 report from the European Heart Rhythm Association. Europace, 17(Suppl 1), i1–i75.
Epstein, A. E., et al. (2013). 2012 ACCF/AHA/HRS focused update incorporated into the ACCF/AHA/HRS 2008 guidelines for device-based therapy of cardiac rhythm abnormalities: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines and the Heart Rhythm Society. Journal of the American College of Cardiology, 61(3), e6–75.
European Society of, C. (2013). ESC guidelines on cardiac pacing and cardiac resynchronization therapy: the task force on cardiac pacing and resynchronization therapy of the European Society of Cardiology (ESC). Developed in collaboration with the European Heart Rhythm Association (EHRA). Europace, 15(8), 1070–118.
Kumar, S., et al. (2013). Ten-year trends in the use of catheter ablation for treatment of atrial fibrillation vs. the use of coronary intervention for the treatment of ischaemic heart disease in Australia. Europace, 15(12), 1702–9.
Kneeland, P. P., & Fang, M. C. (2009). Trends in catheter ablation for atrial fibrillation in the United States. Journal of Hospital Medicine, 4(7), E1–5.
Palaniswamy, C., et al. (2014). Catheter ablation of postinfarction ventricular tachycardia: ten-year trends in utilization, in-hospital complications, and in-hospital mortality in the United States. Heart Rhythm, 11(11), 2056–63.
Katz, D.F. et al. (2015). Safety of ventricular tachycardia ablation in clinical practice: findings from 9,699 hospital discharge records. Circular Arrhythmetic Electrophysiology
Spittell, P. C., & Hayes, D. L. (1992). Venous complications after insertion of a transvenous pacemaker. Mayo Clinic Proceedings, 67(3), 258–65.
Mitrovic, V., et al. (1983). Thrombotic complications with pacemakers. International Journal of Cardiology, 2(3–4), 363–74.
Stoney, W. S., et al. (1976). The incidence of venous thrombosis following long-term transvenous pacing. Annals of Thoracic Surgery, 22(2), 166–70.
Prozan, G. B., et al. (1968). Pulmonary thromboembolism in the presence of an endocardiac pacing catheter. JAMA, 206(7), 1564–5.
Supple, G. E., et al. (2011). Mobile thrombus on device leads in patients undergoing ablation: identification, incidence, location, and association with increased pulmonary artery systolic pressure. Circulation, 124(7), 772–8.
DeSimone, C. V., et al. (2013). Stroke or transient ischemic attack in patients with transvenous pacemaker or defibrillator and echocardiographically detected patent foramen ovale. Circulation, 128(13), 1433–41.
Vaidya, V. R., et al. (2014). Implanted endocardial lead characteristics and risk of stroke or transient ischemic attack. Journal of Interventional Cardiac Electrophysiology, 41(1), 31–8.
Rahbar, A. S., et al. (2013). Risk factors and prognosis for clot formation on cardiac device leads. Pacing and Clinical Electrophysiology, 36(10), 1294–300.
Baker, W. L., et al. (2009). Meta-analysis to assess the quality of warfarin control in atrial fibrillation patients in the United States. Journal of Managed Care Pharmacy, 15(3), 244–52.
van Walraven, C., et al. (2006). Effect of study setting on anticoagulation control: a systematic review and metaregression. Chest, 129(5), 1155–66.
Narducci, M. L., et al. (2013). Usefulness of intracardiac echocardiography for the diagnosis of cardiovascular implantable electronic device-related endocarditis. Journal of the American College of Cardiology, 61(13), 1398–405.
Noheria, A., et al. (2015). Pulmonary embolism in patients with transvenous cardiac implantable electronic device leads. Europace
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Financial support
Dr. DeSimone is supported by a NIH Training Grant T32 #HL0070111.
Conflict of interest
None
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(MP4 18547 kb)
Rights and permissions
About this article
Cite this article
Sugrue, A., DeSimone, C.V., Lenz, C.J. et al. Mobile thrombus on cardiac implantable electronic device leads of patients undergoing cardiac ablation: incidence, management, and outcomes. J Interv Card Electrophysiol 46, 115–120 (2016). https://doi.org/10.1007/s10840-015-0085-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10840-015-0085-2