Abstract
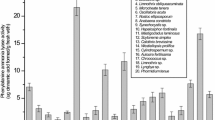
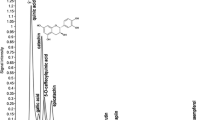
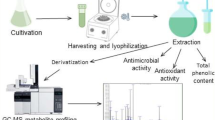
The present study aims to investigate antioxidant activity and the phenolic content in five terrestrial cyanobacterial strains isolated from the Fruška Gora mountain forest ecosystem (Serbia). The antioxidant activity of crude ethanolic extracts was assessed using 2,2-diphenyl-1-picrylhydrazyl (DPPH) and ferric reducing ability of plasma (FRAP) assay. All the tested cyanobacterial strains belonging to Phormidium, Nostoc, Oscillatoria, Anabaena, and Calothrix genera exhibited antioxidant activity. The ethanolic extracts from Calothrix M2 showed strong inhibition against 2,2-diphenyl-1-picrylhydrazyl (DPPH) radicals (half maximal inhibitory concentration (IC50) = 30.72 ± 3.31 μg mL−1) whereby Phormidium M1 expressed the highest radical potential in FRAP assay (22.48 ± 2.18 mg ascorbic acid equivalents (AAE) g−1). Furthermore, the presence and content of 15 phenolics in ethanolic extracts of the tested cyanobacterial strains were identified using liquid chromatography-tandem mass spectrometry (LC-MS/MS). The richest strain in polyphenolics was Phormidium M1 which contained 12 compounds, mostly phenolic acids and flavonoid glycosides, followed by Nostoc M1 and Oscillatoria M2. The noticeable amount of phenolic acids (quinic acid 502 μg g−1; gallic acid 84.9 μg g−1; vanillic acid 50 μg g−1) was observed in Phormidium M1 indicating that phenolic compounds could contribute to the antioxidant activity observed in FRAP assay. LC-MS/MS analysis showed that the tested terrestrial cyanobacterial species could be regarded as a promising new source of bioactive phenolic compounds. Since no significant correlation was observed between phycocyanin content and antioxidant activity, other compounds present in the crude extracts contributed to antioxidant capacities in DPPH and FRAP assay. This study highlights the potential of terrestrial cyanobacterial strains inhabiting forest ecosystems as an interesting source of compounds with antioxidant potential for further researches and exploitation.


Similar content being viewed by others
References
Abd El-Baky HH, El Baz FK, El-Baroty GS (2009) Production of phenolic compounds from Spirulina maxima microalgae and its protective effects. Afr J Biotechnol 8:7059–7067
Babaoğlu Aydaş S, Ozturk S, Aslım B (2013) Phenylalanine ammonia lyase (PAL) enzyme activity and antioxidant properties of some cyanobacteria isolates. Food Chem 136:164–169
Beara I, Lesjak M, Četojević-Simin D, Orčić D, Janković T, Anačkov G, Mimica-Dukić N (2012) Phenolic profile, antioxidant, anti-inflammatory and cytotoxic activities of endemic Plantago reniformis G. Beck. Food Res Int 49:501–507
Benedetti S, Benvenuti F, Pagliarani S, Francogli S, Scoglio S, Canestrari F (2004) Antioxidant properties of a novel phycocyanin extract from the blue-green alga Aphanizomenon flos-aquae. Life Sci 75:2353–2362
Bennett A, Bogorad L (1973) Complementary chromatic adaptation in a filamentous blue-green alga. J Cell Biol 58:419–435
Benzie IFF, Strain JJ (1996) The ferric reducing ability of plasma (FRAP) as a measure of antioxidant power: the FRAP assay. Anal Biochem 239:70–76
Bhat VB, Madyastha KM (2000) C-phycocyanin: a potent peroxyl radical scavenger in vivo and in vitro. Biochem Biophis Res Commun 275:20–25
Böhm V, Puspitasari-Nienaber NL, Ferruzzi MG, Schwartz SJ (2002) Trolox equivalent antioxidant capacity of different geometrical isomers of α-carotene, β-carotene, lycopene, and zeaxanthin. J Agric Food Chem 50:221–226
Chandrasekar D, Madhusudhana K, Ramakrishna S, Diwan PV (2006) Determination of DPPH free radical scavenging activity by reversed-phase HPLC: a sensitive screening method for polyherbal formulations. J Pharm Biomed 40:460–464
Chen F (1996) High cell density culture of microalgae in heterotrophic growth. Trends Biotechnol 14:421–426
Chen F, Li HB, Wong RNS, Ji B, Jiang Y (2005) Isolation and purification of the bioactive carotenoid zeaxanthin from the microalga Microcystis aeruginosa by high-speed counter-current chromatography. J Chromatogr A 1064:183–186
Colla LM, Reinehr CO, Reichert C, Costa JAV (2007) Production of biomass and nutraceutical compounds by Spirulina platensis under different temperature and nitrogen regimes. Bioresource Technol 98:1489–1493
Dixon RA, Achnine L, Kota P, Liu CJ, Reddy MS, Wang L (2002) The phenylpropanoid pathway and plant defence—a genomics perspective. Mol Plant Pathol 3:371–390
Duval B, Shetty K, Thomas WH (1999) Phenolic compounds and antioxidant properties in the snow alga Chlamydomonas nivalis after exposure to UV light. J Appl Phycol 11:559–566
Espin JC, Soler-Rivas C, Wichers HJ (2000) Characterisation of the total free radical scavenger capacity of vegetable oils and oil fractions using 2,2-diphenyl-1-picrylhydrazyl radical. J Agric Food Chem 48:648–656
Gantar M, Simović D, Djilas S, Gonzalez WW, Miksovska J (2012) Isolation, characterization and antioxidative activity of C-phycocyanin from Limnothrix sp. strain 37-2-1. J Biotechnol 159:21–26
Goiris K, Muylaert K, Fraeye I, Foubert I, de Brabanter J, de Cooman L (2012) Antioxidant potential of microalgae in relation to their phenolic and carotenoid content. J Appl Phycol 24:1477–1486
Goiris K, Muylaert K, Voorspoels S, Noten B, De Paepe D, Baart GJE, De Cooman L (2014) Detection of flavonoids in microalgae from different evolutionary lineages. J Phycol 50:483–492
Gouveia L, Raymundo A, Batista AP, Sousa I, Empis J (2006) Chlorella vulgaris and Haematococcus pluvialis biomass as colouring and antioxidant in food emulsions. Eur Food Res Technol 222:362–367
Guedes AC, Amaro HM, Gião MS, Malcata FX (2013) Optimization of ABTS radical cation assay specifically for determination of antioxidant capacity of intracellular extracts of microalgae and cyanobacteria. Food Chem 138:638–643
Hajimahmoodi M, Faramarzi MA, Mohammadi N, Soltani N, Oveisi MR, Nafissi-Varcheh N (2010) Evaluation of antioxidant properties and total phenolic contents of some strains of microalgae. J Appl Phycol 22:43–50
Hirata T, Tanaka M, Ooike M, Tsunomura T, Sakaguchi M (2000) Antioxidant activities of phycocyanobilin prepared from Spirulina platensis. J Appl Phycol 12:435–439
Huang Z, Guo BJ, Wong RNS, Jiang Y (2007) Characterization and antioxidant activity of selenium-containing phycocyanin isolated from Spirulina platensis. Food Chem 100:1137–1143
Kepekçi RA, Saygideger SD (2012) Enhancement of phenolic compound production in Spirulina platensis by two-step batch mode cultivation. J Appl Phycol 24:897–905
Kepekçi RA, Polat S, Çelik A, Bayat N, Saygideger SD (2013) Protective effect of Spirulina platensis enriched in phenolic compounds against hepatotoxicity induced by CCl4. Food Chem 141:1972–1979
Klejdus B, Kopecký J, Benešová L, Vacek J (2009) Solid-phase/supercritical-fluid extraction for liquid chromatography of phenolic compounds in freshwater microalgae and selected cyanobacterial species. J Chromatogr A 1216:763–771
Liu RH (2003) Health benefits of fruit and vegetables are from additive and synergistic combinations of phytochemicals. Am J Clin Nutr 78:517S–520S
Machu L, Misurcova L, Ambrozova JV, Orsavova J, Mlcek J, Sochor J, Jurikova T (2015) Phenolic content and antioxidant capacity in algal food products. Molecules 20:1118–1133
Manach C, Scalbert A, Morand C, Rémésy C, Jiménez L (2004) Polyphenols: food sources and bioavailability. Am J Clin Nutr 79:727–747
Onofrejová L, Vašíčková J, Klejdus B, Stratil P, Mišurcová L, Kráčmar S, Kopecký J, Vacek J (2010) Bioactive phenols in algae: the application of pressurized-liquid and solid-phase extraction techniques. J Pharm Biomed 51:464–470
Orčić DZ, Francišković MM, Bekvalac KJ, Svričev EĐ, Beara IN, Lesjak MM, Mimica-Dukić NM (2014) Quantitative determination of plant phenolics in Urtica dioica extracts by high-performance liquid chromatography coupled with tandem mass spectrometric detection. Food Chem 143:48–53
Pandey U, Pandey J (2008) Enhanced production of biomass, pigments and antioxidant capacity of a nutritionally important cyanobacterium Nostochopsis lobatus. Bioresource Technol 99:4520–4523
Parwani L, Bhatnagar M, Bhatnagar A, Sharma V (2014) Antioxidant and iron-chelating activities of cyanobacterial exopolymers with potential for wound healing. J Appl Phycol 26:1473–1482
Piñero Estrada JE, Bermejo Bescós P, Villar del Fresno AM (2001) Antioxidant activity of different fractions of Spirulina platensis protean extract. Il Farmaco 56:497–500
Plaza M, Santoyo S, Jaime L, Reina GGB, Herrero M, Señoráns FJ, Ibáñez E (2010) Screening for bioactive compounds from algae. J Pharm Biomed 51:450–455
Pumas C, Vacharapiyasophon P, Peerapornpisal Y, Leelapornpisid P, Boonchum W, Ishii M, Khanongnuch C (2011) Thermostablility of phycobiliproteins and antioxidant activity from four thermotolerant cyanobacteria. Phycol Res 59:166–174
Rastogi RP, Sinha RP (2009) Biotechnological and industrial significance of cyanobacterial secondary metabolites. Biotechnol Adv 27:521–539
Rippka R, Deruelles J, Waterbury JB, Herdman M, Stanier RY (1979) Generic assignments, strain histories and properties of pure cultures of cyanobacteria. J Gen Microbiol 111:1–61
Santoyo S, Herrero M, Javier F, Cifuentes A, Ibanez E, Jaime L (2006) Functional characterization of pressurized liquid extracts of Spirulina platensis. Eur Food Res Technol 224:75–81
Simeunović J, Bešlin K, Svirčev Z, Kovač D, Babić O (2013) Impact of nitrogen and drought on phycobiliprotein content in terrestrial cyanobacterial strains. J Appl Phycol 25:597–607
Singh S, Singh RP (2008) In vitro methods of assay of antioxidants: an overview. Food Rev Int 24:392–415
Soni B, Trivedi U, Madamwar D (2008) A novel method of single step hydrophobic interaction chromatography for the purification of phycocyanin from Phormidium fragile and its characterization for antioxidant property. Bioresource Technol 99:188–194
Srivastava AK, Bhargava P, Rai LC (2005) Salinity and copper-induced oxidative damage and changes in the antioxidative defence systems of Anabaena doliolum. World J Microbiol Biotechnol 21:1291–1298
Tsao R, Deng Z (2004) Separation procedures for naturally occurring antioxidant phytochemicals. J Chromatogr B 812:85–99
Woese CR (1987) Bacterial evolution. Microbiol Rev 51:221–271
Acknowledgments
This study has been supported by the funding of the Ministry of Education and Science of the Serbian Government (project number: III 43002) which is greatly acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Babić, O., Kovač, D., Rašeta, M. et al. Evaluation of antioxidant activity and phenolic profile of filamentous terrestrial cyanobacterial strains isolated from forest ecosystem. J Appl Phycol 28, 2333–2342 (2016). https://doi.org/10.1007/s10811-015-0773-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-015-0773-4