Abstract
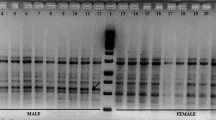
There is an urgent need for early sex identification to support field planting in Ginkgo biloba L., due to the different economic and medicinal values between male and female trees. An easy, rapid and reliable molecular method for sex type determination of G. biloba was reported in the paper. Random amplification of polymorphic DNA (RAPD) and sequence-characterized amplified region (SCAR) were used to search for specific molecular markers linked to the sex locus. A total of 48 primers were used for screening of specific RAPD markers in six male and three female samples. Only one primer, S10, showed different amplification band patterns associated with sex types. Then the sex-specific bands, S10-BandA and S10-BandB, were cloned and sequenced. Based on the sequences two pairs of SCAR primers, GBA and GBB, were designed. The GBA primers amplify a single 571 bp band in male samples but not in female samples, and DNA amplification using GBB primers could generate a 688 bp band only in the female individuals. Finally, the SCAR primers were used to test 16 sex-unknown samples. SCAR primers developed in this paper can be used as effective, convenient and reliable molecular markers for sex identification in G. biloba.



Similar content being viewed by others
References
Agrawal V, Sharma K, Gupta S, Kumar R, Prasad M (2007) Identification of sex in Simmondsia chinensis (Jojoba) using RAPD markers. Plant Biotechnol Rep 1:207–210. doi:10.1007/s11816-007-0031-6
Alstrom RC, Lascoux M, Wang YC, Roberts G, Tuskan GA (1998) Identification of a RAPD marker linked to sex determination in the basket willow (Salix viminalis L.). J Hered 89:44–49. doi:10.1093/jhered/89.1.44
Ćulafić L, Samofalova A, Nešković M (1987) In vitro organogenesis in two dioecious species, Rumex acetosella L. and R. acetosa L. (Polygonaceae). Plant Cell Tissue Organ Cult 11:125–131. doi:10.1007/BF00041845
Dellaporta SL, Calderon UA (1993) Sex determination in flowering plants. Plant Cell 5:1241–1251
Gill GP, Harvey CF, Gardner RC, Fraser LG (1998) Development of sex-linked PCR markers for gender identification in Actinidia. Theor Appl Genet 97:439–445. doi:10.1007/s001220050914
Hardrys H, Balick M, Schierwater B (1992) Applications of random amplified polymorphic DNA (RAPD) in molecular ecology. Mol Ecol 1:55–63. doi:10.1111/j.1365-294X.1992.tb00155.x
Hiroki I, Shigehiro K, Tamami T, Yoshimasa S (1998) Ginkgolide production in relation to organogenesis in Ginkgo biloba. J Wood Sci 44:375–378. doi:10.1007/BF01130450
Hisatsune H, Matsumura K, Ogawa M, Uemura A, Kondo N, Yamashita JK, Katsuta H, Nishikawa S, Chiba T, Nishikawa SI (2005) A high level of endo-thelial cell-specific gene expression by a combination of 5′flanking region and 5′half of the first intron of VE-cadherin gene. Blood 105(12):4657–4663. doi:10.1182/blood-2004-09-3554
Hormaza JI, Dollo L, Polito VS (1994) Identification of a RAPD marker linked to sex determination in Pistacia vera using bulked segregant analysis. Theor Appl Genet 89:9–13. doi:10.1007/BF00226975
Jiang C, Sink KC (1997) RAPD and SCAR markers linked to the sex expression locus M in asparagus. Euphytica 94:329–333. doi:10.1023/A:1002958007407
Jones CJ, Edwards KJ, Castaglione S, Winfield MO, Sala F, Wiel C, Bredemeijer G, Vosman B, Matthes M, Daly A, Brettschneider R, Bettini P, Buiatti M, Maestri E, Malcevschi A, Marmiroli N, Aert R, Volckaert G, Rueda J, Linacero R, Vazquez A, Karp A (1997) Reproducibility testing of RAPD, AFLP and SSR markers in plants by a network of European laboratories. Mol Breed 3:381–390. doi:10.1023/A:1009612517139
Lemos EGM, Silva CLSP, Zaidan HA (2002) Identification of sex in Carica papaya L. using RAPD markers. Euphytica 127:179–184. doi:10.1023/A:1020269727772
Lubbers EL, Arthur L, Hanna WW, Ozias AP (1994) Molecular markers shared by diverse apomictic Pennisetum species. Theor Appl Genet 89:636–642. doi:10.1007/BF00222459
Major RT (1967) The ginkgo, the most ancient living tree. Science 157:1270–1273. doi:10.1126/science.157.3794.1270
Mandolino G, Carboni A, Forapani S, Faeti V, Ranalli P (1999) Identification of DNA markers linked to the male sex in dioecious hemp (Cannabis sativa L.). Theor Appl Genet 98:86–92. doi:10.1007/s001220051043
Paran I, Michelmore RW (1993) Development of reliable PCR-based markers linked to downy mildew resistance genes in lettuce. Theor Appl Genet 85:985–993. doi:10.1007/BF00215038
Pessino SC, Ortiz JPA, Leblanc O, Valle CB, Evans C, Hayward MD (1997) Identification of a maize linkage group related to apomixis in Brachiaria. Theor Appl Genet 94:439–444. doi:10.1007/s001220050434
Prakash S, Staden JV (2006) Sex identification in Encephalartos natalensis (Dyer and Verdoorn) using RAPD markers. Euphytica 152:197–200. doi:10.1007/s10681-006-9198-0
Salgueiro S, Pignocchi C, Parry MAJ (2000) Intron-mediated gusA ex-pression in tritordeum and wheat resulting from particle bombardment. Plant Mol Biol 42:615–622. doi:10.1023/A:1006331831858
Sheriar GH, Risto P, Rudolf J, Paul B (1998) A gene-targeting ap-proach identifies a function for the first intron in expression of the α1(I) collagen gene. Mol Cell Biol 18:3368–3375
Stehlik I, Blantner FR (2004) Sex-specific SCAR markers in the dioecious plant Rumex nivalis (Polygonaceae) and implications for the evolution of sex chromosomes. Theor Appl Genet 108:238–242. doi:10.1007/s00122-003-1425-7
Tredici PD, Ling H, Yang G (1992) The Ginkgos of Tian Mu Shan. Conserv Biol 6:202–209. doi:10.1046/j.1523-1739.1992.620202.x
Wang SM (2006) Analyses on the leaf physiological and biochemical characteristics of different sexual ginkgo plant. Chin J Eco-Agri 14(1):49–50
Wang XM, Song WQ, Liu S, Li XL, Chen RY (2001) AFLP markers related to sex in a dioecious plant, Ginkgo biloba L. Acta Sci Natur Univ Nankaiensis 34(1):5–9
Welsh J, McClelland M (1990) Fingerprinting genomes using PCR with arbitrary primers. Nucleic Acids Res 18:7213–7218. doi:10.1093/nar/18.24.7213
Williams J, Kublik A, Livak K, Rafalski J, Tingey S (1990) DNA polymorphisms amplified by arbitrary primers are useful as genetics markers. Nucleic Acids Res 18:6531–6535. doi:10.1093/nar/18.22.6531
Xu WJ, Wang BW, Cui KM (2004) RAPD and SCAR markers linked to sex determination in Eucommia ulmoides Oliv. Euphytica 136:233–238. doi:10.1023/B:EUPH.0000032741.99885.c4
Yakubov B, Barazani O, Golan-Goldhirsh A (2005) Combination of SCAR primers and Touchdown-PCR for sex identification in Pistacia vera L. Sci Hortic (Amsterdam) 103:473–478. doi:10.1016/j.scienta.2004.06.008
Yan FS, Evans KA, Stevens LH, Beek TAV, Schoonhoven LM (1990) Deterrents extracted from the leaves of Ginkgo biloba: effects on feeding and contact chemoreceptors. Entomol Exp Appl 54:57–64. doi:10.1007/BF00353987
Acknowledgments
This work was supported by the grant from National Facilities and Information Infrastructure for Science and Technology of China (No. 2005DKA21404) and National Natural Science Foundation of China (No. 50621063). We wish to thank Dr. Diqiang Li, Yuguang Zhang and Daigui Zhang, and are also grateful to the staff in the Hunan Hu**shan National Nature Reserve and Hunan Badagongshan National Nature Reserve for assistance in specimen collection.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liao, L., Liu, J., Dai, Y. et al. Development and application of SCAR markers for sex identification in the dioecious species Ginkgo biloba L.. Euphytica 169, 49–55 (2009). https://doi.org/10.1007/s10681-009-9913-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10681-009-9913-8