Summary
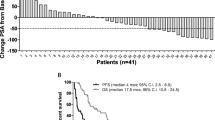
Based on the activity of menadione (M) in the human tumor stem cell assay, we conducted a phase I trial of M in patients with advanced cancer. Forty patients (19 men, 21 women) were treated with 90 courses of M; 82 treatment courses are evaluable for toxicity. The median patient age, Karnofsky performance status, and number of prior chemotherapy regimens were 61 years (range 32–74 years), 80% (range 50–100%), and two, respectively. M was given by a short (1–5 h) intravenous infusion every 3 weeks, starting at 40 mg/m2 and escalating by modified Fibonacci scheme to 1360 mg/m2. Toxicity was graded according to the Southwest Oncology Group toxicity scale with defined hypersensitivity reaction (HSR) scales. No grade ≧2 hematologic toxicity was observed. Non-hematologic toxicity consisted of a HSR syndrome of paresthesiae of the extremities, facial flushing, burning of the eyes and mucous membranes, chest pain and dyspnea. HSR was defined as Grade I toxicity by the presence of facial numbness, flushing, and/or a tingling sensation or burning of the eyes and mucous membranes. Grade II toxicity was defined as the presence of the same above symptoms plus chest tightness, paresthesiae of extremities and/or dyspnea and chest pain. These toxicities were grade 1 in 3 of 4 patients at a dose of 840 mg/m2. At 1360 mg/m2, 2 of 13 patients suffered grade 1 HSR and 7 of 13 grade 2 HSR No objective partial or complete responses were observed. Plasma menadione concentrations peaked at 1.9–7.4 μ M during the infusion in 3 patients receiving 1360 mg/m2. Further phase 1 and 2 combination trials using longer infusion durations have resulted from this trial.
Similar content being viewed by others
References
Thor H, Smith MT, Hartszell P, Bellomo G, Jewell SA, Orrenius S: The metabolism of menadione (2-methyl-1,4-naphthoquinone) by isolated hepatocytes. A study of the implications of oxidative stress in intact cells. J Biol Chem 257: 12419–12425, 1982
Lind C, Hochstein P, Ernster L: DT-diaphorase as a quinone reductase: A cellular control device against semiquinone and superoxide radical formation. Archives Biochem & Biophy 216: 178–185, 1982
Akman SA, Doroshow JH, Dietrich MF, Chlebowski RT, Block JS: Synergistic cytotoxicity between menadione and dicumarol vs. murine leukemia L1210. J Pharm Exper Therapeut 240: 486–491, 1987
Chlebowski RT, Dietrich M, Akman S, Block JB: Vitamin K3 inhibition of malignant murine cell growth and human tumor colony formation. Cancer Treatment Reports 69: 527–532, 1985
Prasad KN, Edwards-Prasad J, Sakamoto A: Vitamin K3 (menadione) inhibits the growth of mammalian tumor cells in culture. Life Sciences 29: 1387–1392, 1981
Akman SA, Dietrich, M, Chlebowski, RT, Limberg P, Block JB: Modulation of cytotoxicity of menadione sodium bisulfite versus leukemia L1210 by the acid-soluble thiol pool. Cancer Res 45: 5257–5262, 1985
Miller AB, Hoogstraten B, Staquet M: Reporting results of cancer treatment. Cancer 47: 207–214, 1981
Yang WH, Purchase EC, Rivingston RN: Positive skin tests and Prausnitz-Kustner reactions in metabisulfite-sensitive subjects. J Allergy Clin Immuno 79: 15, 1987
Riggs BS, Harcherlroad FP Jr, Poole C: Allergic reaction to sulfiting agents. Annals Emergency Medicine. 15: 77–79, 1986
Sprenger JD, Altman LC, Marshall SG, Pierson WE, Koenig JQ: Studies of neutrophil chemotactic factor of anaphylaxis in metabisulfite sensitivity. Annals Allergy 62: 117–121, 1989
Margolin KA, Akman SA, Leong LA, Morgan RJ, Somlo G, Raschko JW, Ahn C, Doroshow JH: Phase I study of mitomycin C and menadione in advanced solid tumors. Cancer Chemother Pharmacol 36: 293–298, 1995
Su YZ, Duarte TE, Dill PE, Weisenthal LM: Selective enhancement by Menadiol of in vitro drug activity in human lymphatic neoplasms. Cancer Treat Rep 71: 619, 1987
Durse L, Rajagopalan S, Eliot HM, Covey JM, Sinha BK: DNA interstrand crosslink and free radical formation in a human multidrug-resistant cell line from mitomycin C and its analogues. Cancer Res 50: 648, 1990
Keizer H, De Leeuw SJ, Van Rijn J, Pinedo HM, Joenje H: Effect of artificial electron acceptors on the cytotoxicity of mitomycin C and doxorubicin in human lung tumor cells. Eur J Cancer Clin Oncol 25: 1113, 1989
Nishikawa Y, Carr B, Wang M, Kar S, Finn F, Dowd P, Zheng Z, Kerns J, Naganathan S: Growth Inhibition of hepatoma cells induced by vitamin K and its analogs. J Biol Chem 270: 28304–28310, 1995
Tetef M, Margolin K, Ahn C, Akman S, Chow W, Coluzzi P, Leong L, Morgan Jr RJ, Raschko J, Shibata S, Somlo G, Doroshow JH: Mitomycin C and menadione for the treatment of advanced gastrointestinal cancer: A phase II trial. J Cancer Res Clin Oncol 121: 103–106, 1995
Tetef M, Margolin K, Ahn C, Akman S, Chow W, Leong L, Morgan RJ Jr, Raschko J, Somlo G, Doroshow JH: Mitomycin C and menadione for the treatment of lung cancer: A phase II trial. Invest New Drugs 13: 157–162, 1995
Siderova YA, Grishanova AY: Dose- and time-dependent effects of menadione on enzymes of xenobiotic metabolism in rat liver. Bull Exp Biol Med 137: 231–234, 2004
Taper HS, Jamison JM, Gilloteaux J, Summers JL, Calderon PB: Inhibition of the development of metastases by dietary vitamin C combination. Life Sci 75: 955–967, 2004
von Gruenigen VE, Jamison JM, Gilloteaux J, Lorimer HE, Summers M, Pollard RR, Gwin CA, Summers JL: The in vitro antitumor activity of vitamins C and K3 against ovarian carcinoma. Anticancer Res 23: 3279–3287, 2003
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lim, D., Morgan, R.J., Akman, S. et al. Phase I trial of menadiol diphosphate (vitamin K3) in advanced malignancy. Invest New Drugs 23, 235–239 (2005). https://doi.org/10.1007/s10637-005-6731-2
Issue Date:
DOI: https://doi.org/10.1007/s10637-005-6731-2