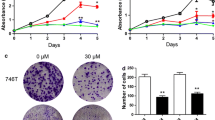
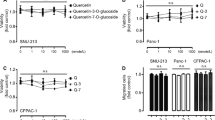
Abstract
Pancreatic ductal adenocarcinoma (PDA) is an aggressive type of malignant tumor with a poor prognosis and high mortality. Aberrant activation of hedgehog signaling plays a crucial role in the maintenance and progression of PDA. Here, we report that the dietary bioflavonoid quercetin has therapeutic potential for PDA by targeting sonic hedgehog (SHH) signaling. The effects of quercetin on the proliferation, apoptosis, migration, and invasion of pancreatic cancer cells (PCCs) and tumor growth and metastasis in PDA xenograft mouse models were evaluated. Additionally, SHH signaling activity was determined. Quercetin significantly inhibited PCC proliferation by downregulating c-Myc expression. In addition, quercetin suppressed epithelial-mesenchymal transition (EMT) by reducing TGF-β1 level, which resulted in inhibition of PCC migration and invasion. Moreover, quercetin induced PCC apoptosis through mitochondrial and death receptor pathways. In nude mouse models, PDA growth and metastasis were reduced by quercetin treatment. Mechanically, quercetin exerts its therapeutic effects on PDA by decreasing SHH activity. Interestingly, quercetin-induced SHH inactivation is mainly dependent on Gli2, but not Gli1. Enhance SHH activity by recombinant Shh protein abolished the quercetin-mediated inhibition of PCC proliferation, migration, and invasion. Furthermore, Shh activated TGF-β1/Smad2/3 signaling and promoted EMT by inducing the expression of Zeb2 and Snail1 that eventually resulted in a partial reversal of quercetin-mediated inhibition of PCC migration and invasion. We conclude that quercetin inhibited the growth, migration, and invasion and induced apoptosis of PCCs by antagonizing SHH and TGF-β/Smad signaling pathways. Thus, quercetin may be a potential candidate for PDA treatment.








Similar content being viewed by others
Abbreviations
- Bax:
-
Bcl-2-associated X protein
- Bcl-2:
-
B cell lymphoma 2
- CCK-8:
-
Cell counting kit 8
- DMEM:
-
Dulbecco’s modified Eagle’s medium
- ELISA:
-
Enzyme-linked immunosorbent assay
- EMT:
-
Epithelial-to-mesenchymal transition
- EMT-TFs:
-
EMT-inducing transcription factors
- FBS:
-
Fetal bovine serum
- GAPDH:
-
Glyceraldehyde 3-phosphate dehydrogenase
- IHC:
-
Immunohistochemical
- PBS:
-
Phosphate buffer saline
- PCCs:
-
Pancreatic cancer cells
- PDA:
-
Pancreatic ductal adenocarcinoma
- qRT-PCR:
-
Quantitative reverse transcriptase-PCR
- RTCA:
-
Real-time cell analysis
- Shh:
-
Sonic hedgehog
- Smo:
-
Smoothened
- TGF-β1:
-
Transforming growth factor-β1
References
Aiello NM, Brabletz T, Kang Y, Nieto MA, Weinberg RA, Stanger BZ. Upholding a role for EMT in pancreatic cancer metastasis. Nature. 2017;547:E7–8.
Amakye D, Jagani Z, Dorsch M. Unraveling the therapeutic potential of the Hedgehog pathway in cancer. Nat Med. 2013;19:1410–22.
Bai CB, Auerbach W, Lee JS, Stephen D, Joyner AL. Gli2, but not Gli1, is required for initial Shh signaling and ectopic activation of the Shh pathway. Development. 2002;129:4753–61.
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424.
Cannac F, Qi C, Falschlunger J, Hausmann G, Basler K, Korkhov VM. Cryo-EM structure of the Hedgehog release protein Dispatched. Sci Adv. 2020;6:eaay7928.
Chen S, Jiang H, Wu X, Fang J. Therapeutic effects of quercetin on inflammation, obesity, and type 2 diabetes. Mediators Inflamm. 2016;2016:9340637.
Christenson ES, Jaffee E, Azad NS. Current and emerging therapies for patients with advanced pancreatic ductal adenocarcinoma: a bright future. Lancet Oncol. 2020;21:e135–45.
Collisson EA, Bailey P, Chang DK, Biankin AV. Molecular subtypes of pancreatic cancer. Nat Rev Gastroenterol Hepatol. 2019;16:207–20.
David CJ, Huang YH, Chen M, Su J, Zou Y, Bardeesy N, et al. TGF-beta tumor suppression through a lethal EMT. Cell. 2016;164:1015–30.
Ding Q, Motoyama J, Gasca S, Mo R, Sasaki H, Rossant J, et al. Diminished Sonic hedgehog signaling and lack of floor plate differentiation in Gli2 mutant mice. Development. 1998;125:2533–43.
Drenkhahn SK, Jackson GA, Slusarz A, Starkey NJ, Lubahn DB. Inhibition of hedgehog/Gli signaling by botanicals: a review of compounds with potential hedgehog pathway inhibitory activities. Curr Cancer Drug Targets. 2013;13:580–95.
Foltyn W, Zajecki W, Marek B, Kajdaniuk D, Sieminska L, Zemczak A, et al. The value of the Ki-67 proliferation marker as a prognostic factor in gastroenteropancreatic neuroendocrine tumours. Endokrynol Pol. 2012;63:362–6.
Fu J, Rodova M, Roy SK, Sharma J, Singh KP, Srivastava RK, et al. GANT-61 inhibits pancreatic cancer stem cell growth in vitro and in NOD/SCID/IL2R gamma null mice xenograft. Cancer Lett. 2012;330:22–32.
Gaarenstroom T, Hill CS. TGF-beta signaling to chromatin: how Smads regulate transcription during self-renewal and differentiation. Semin Cell Dev Biol. 2014;32:107–18.
Guo X, Wang XF. Signaling cross-talk between TGF-beta/BMP and other pathways. Cell Res. 2009;19:71–88.
Haque I, De A, Majumder M, Mehta S, McGregor D, Banerjee SK, et al. The matricellular protein CCN1/Cyr61 is a critical regulator of Sonic Hedgehog in pancreatic carcinogenesis. J Biol Chem. 2012;287:38569–79.
Heldin CH, Vanlandewijck M, Moustakas A. Regulation of EMT by TGFbeta in cancer. FEBS Lett. 2012;586:1959–70.
Hidalgo M, Maitra A. The hedgehog pathway and pancreatic cancer. N Engl J Med. 2009;361:2094–6.
Kasai K. GLI1, a master regulator of the hallmark of pancreatic cancer. Pathol Int. 2016;66:653–60.
Kawabata K, Mukai R, Ishisaka A. Quercetin and related polyphenols: new insights and implications for their bioactivity and bioavailability. Food Funct. 2015;6:1399–417.
Khaksary Mahabady M, Gholami MR, Najafzadeh Varzi H, Zendedel A, Doostizadeh M. Protective effect of quercetin on skeletal and neural tube teratogenicity induced by cyclophosphamide in rat fetuses. Vet Res Forum. 2016;7:133–8.
Kim JH, Kim MJ, Choi KC, Son J. Quercetin sensitizes pancreatic cancer cells to TRAIL-induced apoptosis through JNK-mediated cFLIP turnover. Int J Biochem Cell Biol. 2016;78:327–34.
Kuspert M, Weider M, Muller J, Hermans-Borgmeyer I, Meijer D, Wegner M. Desert hedgehog links transcription factor Sox10 to perineurial development. J Neurosci. 2012;32:5472–80.
Lan CY, Chen SY, Kuo CW, Lu CC, Yen GC. Quercetin facilitates cell death and chemosensitivity through RAGE/PI3K/AKT/mTOR axis in human pancreatic cancer cells. J Food Drug Anal. 2019;27:887–96.
Liu X, Sun N, Mo N, Lu S, Song E, Ren C, et al. Quercetin inhibits kidney fibrosis and the epithelial to mesenchymal transition of the renal tubular system involving suppression of the Sonic Hedgehog signaling pathway. Food Funct. 2019;10:3782–97.
Lu H, Chen B, Hong W, Liang Y, Bai Y. Transforming growth factor-beta1 stimulates hedgehog signaling to promote epithelial-mesenchymal transition after kidney injury. FEBS J. 2016;283:3771–90.
Lu H, Wu L, Liu L, Ruan Q, Zhang X, Hong W, et al. Quercetin ameliorates kidney injury and fibrosis by modulating M1/M2 macrophage polarization. Biochem Pharmacol. 2018;154:203–12.
Massague J. TGFbeta signalling in context. Nat Rev Mol Cell Biol. 2012;13:616–30.
McMahon SB. MYC and the control of apoptosis. Cold Spring Harb Perspect Med. 2014;4:a014407.
Moutasim KA, Mellows T, Mellone M, Lopez MA, Tod J, Kiely PC, et al. Suppression of Hedgehog signalling promotes pro-tumourigenic integrin expression and function. J Pathol. 2014;233:196–208.
Nevala-Plagemann C, Hidalgo M, Garrido-Laguna I. From state-of-the-art treatments to novel therapies for advanced-stage pancreatic cancer. Nat Rev Clin Oncol. 2019;17:108–23.
Olive KP, Jacobetz MA, Davidson CJ, Gopinathan A, McIntyre D, Honess D, et al. Inhibition of Hedgehog signaling enhances delivery of chemotherapy in a mouse model of pancreatic cancer. Science. 2009;324:1457–61.
Panzuto F, Boninsegna L, Fazio N, Campana D, Pia Brizzi M, Capurso G, et al. Metastatic and locally advanced pancreatic endocrine carcinomas: analysis of factors associated with disease progression. J Clin Oncol. 2011;29:2372–7.
Park HL, Bai C, Platt KA, Matise MP, Beeghly A, Hui CC, et al. Mouse Gli1 mutants are viable but have defects in SHH signaling in combination with a Gli2 mutation. Development. 2000;127:1593–605.
Probst S, Zeller R, Zuniga A. The hedgehog target Vlk genetically interacts with Gli3 to regulate chondrocyte differentiation during mouse long bone development. Differentiation. 2013;85:121–30.
Rhim AD, Mirek ET, Aiello NM, Maitra A, Bailey JM, McAllister F, et al. EMT and dissemination precede pancreatic tumor formation. Cell. 2012;148:349–61.
Rhodes DR, Yu J, Shanker K, Deshpande N, Varambally R, Ghosh D, et al. ONCOMINE: a cancer microarray database and integrated data-mining platform. Neoplasia. 2004;6:1–6.
Saini F, Argent RH, Grabowska AM. Sonic Hedgehog Ligand: a role in formation of a mesenchymal niche in human pancreatic ductal adenocarcinoma. Cells. 2019;8:424.
Salama YA, El-Karef A, El Gayyar AM, Abdel-Rahman N. Beyond its antioxidant properties: quercetin targets multiple signalling pathways in hepatocellular carcinoma in rats. Life Sci. 2019;236:116933.
Sistani Karampour N, Arzi A, Najafzadeh Varzi H, Mohammadian B, Rezaei M. Quercetin preventive effects on theophylline-induced anomalies in rat embryo. Jundishapur J Nat Pharm Prod. 2014;9:e17834.
Tang SN, Fu J, Nall D, Rodova M, Shankar S, Srivastava RK. Inhibition of sonic hedgehog pathway and pluripotency maintaining factors regulate human pancreatic cancer stem cell characteristics. Int J Cancer. 2011;131:30–40.
Tang YA, Chen YF, Bao Y, Mahara S, Yatim S, Oguz G, et al. Hypoxic tumor microenvironment activates GLI2 via HIF-1alpha and TGF-beta2 to promote chemoresistance in colorectal cancer. Proc Natl Acad Sci U S A. 2018;115:E5990–9.
Thiery JP, Acloque H, Huang RY, Nieto MA. Epithelial-mesenchymal transitions in development and disease. Cell. 2009;139:871–90.
Xu FG, Ma QY, Wang Z. Blockade of hedgehog signaling pathway as a therapeutic strategy for pancreatic cancer. Cancer Lett. 2009;283:119–24.
Xu Y, Chang R, Peng Z, Wang Y, Ji W, Guo J, et al. Loss of polarity protein AF6 promotes pancreatic cancer metastasis by inducing Snail expression. Nat Commun. 2015;6:7184.
Xu L, Deng S, **ong H, Shi W, Luo S, Chen L. GATA-6 transcriptionally inhibits Shh to repress cell proliferation and migration in lung squamous cell carcinoma. Int J Biochem Cell Biol. 2019;115:105591.
Zhang J, Tian XJ, **ng J. Signal transduction pathways of EMT induced by TGF-beta, SHH, and WNT and their crosstalks. J Clin Med. 2016;5:28.
Zhang X, Lu H, **e S, Wu C, Guo Y, **ao Y, et al. Resveratrol suppresses the myofibroblastic phenotype and fibrosis formation in kidneys via proliferation-related signalling pathways. Br J Pharmacol. 2019;176:4745–59.
Zheng X, Carstens JL, Kim J, Scheible M, Kaye J, Sugimoto H, et al. Epithelial-to-mesenchymal transition is dispensable for metastasis but induces chemoresistance in pancreatic cancer. Nature. 2015;527:525–30.
Zhou P, Li B, Liu F, Zhang M, Wang Q, Liu Y, et al. The epithelial to mesenchymal transition (EMT) and cancer stem cells: implication for treatment resistance in pancreatic cancer. Mol Cancer. 2017;16:52.
Acknowledgments
This study was sponsored by Wenzhou Science and Technology Plan Project, China (Grant No. Y20180100) and Key Laboratory of Diagnosis and Treatment of Severe Hepato-Pancreatic Diseases of Zhejiang Province (2018E10008).
Author information
Authors and Affiliations
Contributions
YB and XL designed the research. YG, HZ, and YX performed the experiments, analyzed the data, and drafted the manuscript. YG, HG, SM, LS, and WZ performed the experiments and collected data for the revision. YT and YB edited the manuscript. YB, SM, and XL contributed to the discussion and review of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Declaration of transparency and scientific rigor
This Declaration acknowledges that this paper adheres to the principles for transparent reporting and scientific rigor of preclinical research as stated in the BJP guidelines for Design & Analysis, Immunoblotting and Immunochemistry, and Animal Experimentation, and as recommended by funding agencies, publishers, and other organizations engaged with supporting research.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Bullet point summary
What is already known?
Quercetin inhibits the growth, migration, and invasion and induced apoptosis of pancreatic cancer cells.
What does this study add?
Gli2-dependent sonic hedgehog signaling is critical for quercetin-mediated anticancer effects in pancreatic ductal adenocarcinoma.
Clinical significance
Quercetin has therapeutic potential for the treatment of pancreatic ductal adenocarcinoma by targeting SHH and TGF-β/Smad signaling pathways.
Highlights
1. Quercetin inhibited growth, migration, and invasion and induced apoptosis of pancreatic cancer cells.
2. Gli2-dependent SHH signaling pathway was involved in quercetin-mediated anticancer effects.
3. Quercetin reduced SHH activity, thereby inhibiting TGF-β1/Smad2/3 signaling and blocking the EMT process by regulating the expression of EMT-TFs Zeb2 and Snail1.
Pharmacological nomenclature
TARGETS
Smo
LIGANDS
Quercetin
Transforming growth factor beta-1
Electronic supplementary material
Figure S1
Effect of quercetin on the activities of JAK2, β-catenin and mTOR. (A) Western blot analysis showing the expression and phosphorylation of JAK2 in quercetin-treated PANC-1 and Patu8988 cells. (B) Western blot analysis showing the expression and phosphorylation of β-catenin in quercetin-treated PANC-1 and Patu8988 cells. (C) Western blot analysis for the expression and phosphorylation of mTOR in quercetin-treated PANC-1 and Patu8988 cells. Data were presented as the mean ± standard deviation in quintuplicate for the cell line experiment. (PNG 583 kb)
Figure S2
Shh expression in human pancreatic ductal adenocarcinoma tissues. (A) Immunohistochemical (IHC) staining for Shh in human pancreatic ductal adenocarcinoma (PDA) and adjacent normal tissues. Bar = 100 μm and 50 μm. (B) ELISA for Shh level in pancreatic ductal epithelial cells (HPDE6-C7 and hTERT-HPNE) and tumor cells (PANC-1 and Patu8988). c In silico analyses of three other independent data sets obtained from Oncomine. *P < 0.05; ***P < 0.001. (PNG 1560 kb)
Figure S3
Gli2 expression in pancreatic ductal adenocarcinoma data sets. (A) In silico analyses of two other independent data sets obtained from Oncomine. (B) Correlation between Gli2 expression level and survival curve of pancreatic ductal adenocarcinoma (PDA). (PNG 399 kb)
Figure S4
Effect of TGF-β1 on SHH signaling in pancreatic cancer cells. Western blot analysis showing the expression of Smo, Gli1, and Gli2 in TGF-β1-treated PANC-1 and Patu8988 cells. Data were presented as the mean ± standard deviation in quintuplicate for the cell line experiment.*P < 0.05. (PNG 392 kb)
Figure S5
Effect of Gli2 siRNA on TGF-β1 expression in pancreatic cancer cells. (A) Western blot analysis showing the expression of Gli2 and TGF-β1 in PANC-1 cells. (B) qRT-PCR analysis showing the mRNA expression of Gli2 and TGF-β1 in PANC-1 cells. Data were presented as the mean ± standard deviation in quintuplicate for the cell line experiment.***P < 0.001. (PNG 241 kb)
ESM.6
(DOCX 16 kb)
ESM 7
(DOCX 14 kb)
Rights and permissions
About this article
Cite this article
Guo, Y., Tong, Y., Zhu, H. et al. Quercetin suppresses pancreatic ductal adenocarcinoma progression via inhibition of SHH and TGF-β/Smad signaling pathways. Cell Biol Toxicol 37, 479–496 (2021). https://doi.org/10.1007/s10565-020-09562-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-020-09562-0