Abstract
Caveolin-1 (CAV1) is commonly considered to function as a cell surface protein, for instance in the genesis of caveolae. Nonetheless, it is also present in many intracellular organelles and compartments. The contributions of these intracellular pools to CAV1 function are generally less well understood, and this is also the case in the context of cancer. This review will summarize literature available on the role of CAV1 in cancer, highlighting particularly our understanding of the canonical (CAV1 in the plasma membrane) and non-canonical pathways (CAV1 in organelles and exosomes) linked to the dual role of the protein as a tumor suppressor and promoter of metastasis. With this in mind, we will focus on recently emerging concepts linking CAV1 function to the regulation of intracellular organelle communication within the same cell where CAV1 is expressed. However, we now know that CAV1 can be released from cells in exosomes and generate systemic effects. Thus, we will also elaborate on how CAV1 participates in intracellular communication between organelles as well as signaling between cells (non-canonical pathways) in cancer.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Caveolin-1 (CAV1) is a small, oligomeric scaffolding protein, typically required to generate membrane curvature in structures, such as caveolae [123, 174]. Moreover, CAV1 binds to many other proteins, controls cholesterol homeostasis, and regulates a variety of cell functions, such as endocytosis, receptor internalization, cholesterol accumulation, and cell signaling, proliferation, and death [29, 123]. CAV1-mediated control of signaling events is relevant in cancer. As an example, may it suffice to mention the interaction between CAV1 and Rho GTPases, such as RhoC, which favors the development of metastasis by stimulating α5-integrin expression and Src kinase-dependent activation of the p130Cas/Rac1, FAK/Pyk2, and Ras/Erk1/2 pathways [6, 115]. Importantly, however, CAV1 also functions as a tumor suppressor by aiding E-cadherin in the sequestration of β-catenin, thereby impeding activation of the β-catenin/Tcf-Lef-dependent transcription of genes, like survivin, cyclooxygenase-2, cyclin D1, and many others that favor cancer development [140, 165, 166]. Thus, CAV1 participates both as a tumor suppressor and promoter in cancer (reviewed in [52, 123, 132]).
CAV1 is synthesized, then oligomerizes, and is inserted into the ER membrane through the classical membrane/secretory protein translocation pathway. The hydrophobic domain serves as an ER membrane anchor and adopts a loop configuration exposing the N- and C-terminal domains to the cytoplasm [109]. In addition, the N-terminal domain has a DXE (Asp-X-Glu) sequence that allows CAV1 to concentrate in ER exit sites and then be transported to the Golgi apparatus with the help of the coat protein II (COPII) machinery. Once in the Golgi apparatus, CAV1 undergoes conformational changes and assembles (in a cholesterol-dependent process) into larger and more stable complexes of about 160 caveolin molecules, lipids, and membrane raft-associated cargos [59]. These are transported to the plasma membrane in vesicles and inserted as planar caveolar domains, into which PTRF/cavin1 can be recruited, as well as other cavins, so as to generate the caveola structures [59, 161]. Thus, because the protein traffics to the cell surface and can then be internalized, CAV1 is also detectable at many intracellular sites. While subcellular location is often considered fortuitous, it is becoming increasingly clear that functionally important organelle-associated pools exist, although their precise role remains largely obscure [44].
In caveolae or membrane rafts at the plasma membrane, CAV1 may serve to generate signaling hubs that control many downstream events. As mentioned, CAV1 aids, together with E-cadherin, in sequestering β-catenin to the plasma membrane and in doing so regulates β-catenin/Tcf-Lef-dependent signaling [45, 165, 166]. In addition, CAV1 is also implicated in the internalization and turnover of the transforming growth factor beta (TGF-β) receptor [36]. Some of these roles are related to CAV1 function as a scaffolding protein; nevertheless, alternative possibilities exist. For instance, Raf-1 is a proto-oncogene and serine/threonine protein kinase, which is recruited to caveolae after epidermal growth factor (EGF) stimulation and its presence there is necessary for the activation of the MAP kinase pathway [107]. However, others suggest that caveolae are depleted of membrane proteins [155] and participate indirectly in, for instance, Ras-dependent signaling by changing lipid organization and cholesterol content in domains of the plasma membrane [4, 144]. Thus, CAV1 plays a significant role in modulating cell signaling events at the cell surface but can do so in different ways.
As indicated, CAV1 has been reported to function both as a tumor suppressor and as a promoter of tumor progression and metastasis (reviewed in [24, 133, 147]). A large number of reports support the role of CAV1 as a tumor suppressor, associating the reduced expression of CAV1 with cell transformation [12, 173, 178]. On the other hand, the re-expression of CAV1, often observed in later stages of cancer, has been linked to tumor progression, multi-drug resistance, and metastasis [38, 73, 89, 95]. To what extent such variability may be linked to the different ways in which CAV1 modulates signaling at the cell surface or elsewhere is currently an area of great interest.
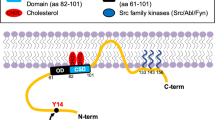
The central region of CAV1 (residues 82–101), referred to as the caveolin scaffolding domain (CSD), is proposed to bind to many other proteins (containing a region enriched in aromatic residues termed caveolin-binding motif, CBM) and prevent downstream signaling events [118]. For instance, CAV1 reportedly interacts with G proteins and suppresses their basal activity by inhibiting GDP/GTP exchange [85, 124]. Moreover, this domain is implicated in regulating cell proliferation and survival by inhibiting cell signaling proteins, such as eNOS, Gi2α, and PKCα [136]. Furthermore, the CSD also regulates Ca2+ influx into cells by interacting with and modulating the activity of certain ion channels, such as TRPC1 [117, 160], thereby leading to the alteration of cellular processes, including cell proliferation [25] and tumor invasion [117]. Moreover, this domain is suggested to be crucial in controlling cell migration, possibly via STAT3, and also cell cycle progression in several cancer cell lines [117].
In striking contrast, other reports describe how this domain reduces the activity of the serine/threonine protein phosphatases PP1 and PP2A in order to sustain Akt activation and thereby promotes cell survival in prostate [83] and pancreatic cancer [61]. Others implicate the CSD in mediating interactions between CAV1 and Rho GTPases. For instance, Rufini and colleagues showed that the disruption of this interaction by overexpressing CSD peptides in metastatic melanoma cells led to diminished survival and extravasation of these cells [6]. Additionally, Hordijk and collaborators showed that CAV1 promotes directional cell migration via CSD-mediated interaction with the C-terminal domain of Rac1 [114].
However, several reports suggest that CAV1 effects are not due to the interaction between a possible CBM in target proteins and the CSD in CAV1. In general, proteins associated with caveolae are not enriched in CBM. Moreover, CBM are hydrophobic segments enriched in aromatic residues that are buried within the protein structure, making it difficult to envision their participation in the interaction with other proteins [20, 33]. In addition, the CSD is buried in the membrane and inaccessible for the interaction with cytoplasmic proteins [5]. This data points towards the relevance of other mechanisms through which CAV1 regulates signaling events, such as those previously mentioned involving alterations in the lipid domain organization or via interactions mediated by segments of the protein that are phosphorylated.
In this respect, CAV1 phosphorylation on tyrosine 14 (pY14-CAV1) [105] has emerged as being important. Phosphorylation at this site is associated with modulation of focal adhesion dynamics, cell migration, invasion, and tumor metastasis as will be detailed later on [48, 71, 115, 119]. Interestingly, Nabi and colleagues showed that CAV1 phosphorylation on tyrosine 14 stabilizes focal adhesion proteins and promotes cell motility in a CSD-dependent manner in prostate cancer cells [105]. The authors suggest that CAV1 phosphorylation favors the interaction between the CSD and several focal adhesion proteins, which in turn promotes focal adhesion tension and cell migration, thus identifying pY14-CAV1 as a molecular regulator of such processes (see also review in this same special edition by Nabi and co-workers). Other reports also mention the existence of a link between pY14-CAV1 and the CSD, whereby CAV1 phosphorylation on tyrosine 14 is suggested to trigger conformational changes within the oligomeric structure of CAV1 molecules that increase CSD accessibility for interactions with other proteins [150, 186].
The regulation of all the signaling pathways described above is attributed to CAV1 presence at the plasma membrane (canonical and predominantly studied roles). However, an analysis of information available in data bases reveals that CAV1 localizes to many cell organelles and subcellular compartments (Table 1 and Fig. 1), suggesting that CAV1 function is not likely to be limited to the plasma membrane, and moreover, that these CAV1 pools modulate important cellular functions that in general are less characterized (non-canonical roles).
Subcellular localizations of CAV1. Representative scheme that summarizes the subcellular compartments where CAV1 is suggested to be localized as supported by the UniProt, PSORT II, MGI, and LocDB databases and select references (see Table 1). According to the data, CAV1 is present in the plasma membrane, Golgi apparatus, ER, nucleus, endocytic as well as exocytic vesicles, multivesicular bodies (MVB), and lipid droplets. Additional evidence places CAV1 at mitochondria-ER interphase sites (localizing CAV1 indirectly to mitochondria), referred to as mitochondria-associated ER membranes (MAMs), as well as lysosomal, peroxisomal, and exosomal membranes, as will be discussed in this review
Thus, here, we will review in detail the literature available on the role of CAV1 in cancer and specifically the canonical and non-canonical functions of the proteins associated with CAV1 at the plasma membrane and in other subcellular localizations, respectively.
2 Canonical role of CAV1 at the plasma membrane
2.1 CAV1 as a tumor suppressor or promoter
At early stages, CAV1 has been ascribed roles as a tumor suppressor in colorectal, breast, lung, and liver cancers. For instance, the reduction of stromal CAV1 is associated with poor patient survival in breast cancer [182]. CAV1 downregulation promotes proliferation, while the overexpression induces apoptosis in lung cancer cell lines [49]. In addition, CAV1 downregulation inhibits senescence, while promoting lung tumor development and increasing mortality in mice [173].
The regulation of CAV1 protein levels is poorly understood. Low and Nicholson reviewed the effect of epigenetic regulation of CAV1, concluding that hypermethylation of the CAV1 promoter decreases protein levels in breast and prostate cancer [98]. This result is also observed in gastric cardia adenocarcinoma and alveolar rhabdomyosarcoma, where mRNA and protein levels are downregulated by hypermethylation of CpG islands, which is associated with a decrease in patient survival [51, 67]. Treatment with DNA-hypomethylating agents, such as 5-aza-2′-deoxycytidine, restores the expression of CAV1, not only in breast and prostate but also in ovarian, colon, and liver cancer cells [98]. In addition, treatment of colon cancer cells with histone deacetylase inhibitors, such as trichostatin A, increases CAV1 expression and prevents cell proliferation [35]. Besides, epigenetic regulation of CAV1-associated proteins is involved in cancer development. For instance, hypermethylation of the PTRF/cavin1 promoter leads to a reduction in caveola formation and Ewing sarcoma development. The re-introduction of PTRF/cavin1 and CAV1 increases caveola number, promotes cell death, and decreases tumor size [68].
On the other hand, the analysis of CAV1 expression in samples of patients at early stages of colorectal cancer reveals that protein levels are downregulated, but there is no correlation with a reduction in mRNA levels, probably due to post-transcriptional regulation involving miRNA 124 [164].
In terms of the signaling pathways implicated in CAV1-associated tumor suppression, there are several studies on its role as a plasma membrane-bounded protein (see Section 1). Furthermore, CAV1 has been shown to control proliferation and apoptosis of human lung carcinoma cell lines. In these cells, CAV1 reduces cadherin-11/Stat3/Rac1 signaling by interactions at the plasma membrane involving the scaffolding domain [49]. Also, CAV1 stabilizes cell–cell contacts and inhibits the spread of tumor cells during metastasis by favoring the plasma membrane localization of E-cadherin and p120-Catenin and the development of adherent junctions in ovarian carcinoma cell lines [108]. Alternatively, EGF treatment induces caveola-dependent endocytosis of plasma membrane E-cadherin/β-catenin complex, disrupts cell–cell contacts, downregulates E-cadherin as well as CAV1, and thereby promotes invasion of cancer cells [99].
Moreover, CAV1 has been proposed to inhibit TGF-β signaling, via caveolae/membrane raft-mediated endocytosis and lysosomal degradation of TGF-β receptors [28, 175]. On the one hand, CAV1 interacts with TGF-β receptors, abolishing their interaction with SMAD proteins and downstream events [28]. On the other hand, caveolae and membrane rafts create a favorable environment for the recruitment of ubiquitin ligases which promote the ubiquitination and degradation of CAV1 and TGF-β receptors [28]. In this case, CAV1 recruits ubiquitinated TGF-β receptors in the plasma membrane and aids in transport to lysosomes, thereby inhibiting TGF-β signaling. The reduction of the ubiquitination of CAV1 and TGF-β receptors by the deubiquitinase POH1 reduces lysosomal degradation of the TGF-β receptors in liver cancer cells. In doing so, POH1 promotes migration and invasion via TGF-β signaling [175].
Quite to the contrary, CAV1 promotes metastasis and multi-drug resistance in late stages of cancer. The expression of CAV1 is higher in cancer compared to benign tissue and correlates with poor prognosis [127]. The overexpression of CAV1 is associated with increased cell survival, anchorage-independent growth, and epithelial-mesenchymal transition, migration, invasion, and resistance to anti-neoplastic drugs [38, 94, 111, 127, 176].
An important step during the metastatic process is the epithelial-mesenchymal transition (EMT), where CAV1, E-cadherin, and the TGF-β receptor at the plasma membrane are implicated [94, 127]. Looi and collaborators reviewed the role of E-cadherin in EMT, where the protein is replaced by N-cadherin, which disrupts adherent junctions and promotes cancer cell spread [97]. Interestingly, during tumor progression, the switching of cadherins at the plasma membrane is associated with augmented CAV1 expression. In this sense, CAV1 increases migration of melanoma metastatic cells, but the co-expression of CAV1 and E-cadherin inhibits tumor formation and lung metastasis [94]. In addition, CAV1 reprograms the TGF-β signaling pathway from suppressing tumor formation to being oncogenic. The lack of CAV1 increases the expression of epithelial, but reduces the expression of mesenchymal TGF-β target genes in metastatic prostate cancer cells. Moreover, CAV1 silencing increases the expression of E-cadherin, integrin β4, and desmoplakin, thereby favoring epithelial integrity and inhibiting the tumor spread [127].
Recent studies from our laboratory indicate that not only the expression of CAV1 at the plasma membrane but also the phosphorylation of CAV1 on tyrosine 14 is necessary to increase the metastatic potential of cancer cells in vitro and in vivo [119]. Src family kinases are responsible for CAV1 phosphorylation on tyrosine 14 and alternatively, phosphorylated CAV1 aids in the recruitment of particularly Src to the plasma membrane, leading to accumulation and activation of Src in focal adhesions [50]. In metastatic gastric cancer cells, CAV1 promotes resistance to anoikis in a Src-dependent manner by activation of the EGFR/integrin βI, PI3K/pAkt, and MEK/ERK signaling pathways [176]. In addition, in hepatocellular cancer cell lines, CAV1 is required for the induction of survival via the TGF-β/EGFR/pAkt signaling pathway [111].
2.2 Role of CAV1 in migration
The presence of CAV1 at the plasma membrane is necessary for migration and invasion processes, since disruption of membrane rafts by targeting CAV1 or cholesterol reduces the metastatic potential of cancer cells [53, 66, 156]. Concerning lipid droplet biogenesis, CAV1 promotes accumulation of lipids and proteins at specific sites in the ER before entering these organelles [70]. Moreover, a positively charged sequence in the CSD along with the last 20 residues of CAV1 are held responsible for the sorting of CAV1 into lipid droplets [70, 74]. Simulation of lipid droplet biogenesis reveals that CAV1 reduces bilayer thickness of lipid aggregates and thereby reduces the energy barrier to facilitate pinching-off of these aggregates from the host bilayer [129].
In the context of cancer, these anti-lipotoxic organelles tend to accumulate in tumor cells with an aggressive phenotype, as well as in tumor cells exposed to hypoxic or nutrient/lipid-deprived conditions. Furthermore, recent studies suggest that these dynamic organelles are capable of abolishing nutrient and oxidative stress and, therefore, promote tumor cell survival and growth by protecting, for instance, their contents from damage due to peroxidation [128, 156]. Moreover, while the majority of normal tissues synthesize new structural lipids by using extracellular pools of lipids, tumor cells may prefer de novo fatty acid synthesis, despite the availability of these extracellular lipids, in order to meet their specific lipid requirements and/or to maintain proliferation in a stressful environment [128, 141].
Human metastatic breast cancer cell lines, such as the triple negative MDA-MB-231 cells, contain greater levels of cholesterol and fatty acids in lipid droplets when compared to non-metastatic and hormone-responsive breast cancer cells, like MCF-7. Coincidentally, MDA-MB-231 cells express high endogenous levels of CAV1, LDL receptors, and acetyl-CoA:cholesterol acyltransferase 1 (ACAT1) enzymes [2, 3, 156], which facilitate incorporation into LDL particles and promote proliferation [2]. Moreover, Siddiqui and colleagues showed that the migration potential of MDA-MB-231 cells is dependent on ACAT1 and correlates with increased lipid accumulation in these cells [3]. Furthermore, treatment with ACAT1 inhibitors reduces LDL receptor expression and LDL-enhanced proliferation [3]. Another report shows that the treatment of triple negative breast cancer cells with bitter melon extract reduces the accumulation of esterified cholesterol, ACAT1, and LDL receptor expression, thereby reducing tumor growth in mammospheres implanted into mice [152]. Interestingly, ACAT1 inhibition has been reported to promote downregulation of the CAV1/MAPK pathway, which enhances pancreatic cancer aggressivity [79]. To what extent the presence of CAV1 in EVs may aid in drug delivery is an intriguing but as yet unresolved question.
8 Conclusions
In summary, we focused this discussion initially on literature dealing with what we defined as canonical roles of CAV1, as a modulator of signaling events at the plasma membrane. There, CAV1 is largely considered a scaffolding protein that functions within caveolae; however, while roles for the non-caveolar protein in regulating signaling via phosphorylation on tyrosine-14 or modifying plasma membrane lipid composition have been described, they need to be explored in greater detail in the future. Furthermore, we now know that CAV1 expression alters mitochondrial function in a number of ways, yet the relevance of these mechanisms in cancer is still poorly understood, as is also the case for peroxisomal CAV1. The importance of CAV1 in lipid transport is well-established, but again more research on the regulation of these processes and their relevance to tumor biology is necessary. Thus, as stated at the onset, CAV1 is present beyond the plasma membrane in numerous subcellular compartments. However, the precise function of CAV1 at these sites often still remains to be defined, as does the role of these pools in processes related to the development of cancer (non-canonical roles, Fig. 2). Undoubtedly, more research in this area is required to shed light on these emerging new functions of the protein. Finally, understanding how CAV1 participates in EV genesis and function is an exciting new field of CAV1 research that holds considerable promise in the development of cancer therapies.
Signaling events modulated by CAV1 at the plasma membrane (canonical role) and at several subcellular localizations (non-canonical role). Proteins regulated by CAV1 functioning as a tumor suppressor are indicated in purple. Alternatively, those proteins modulated by CAV1 functioning as a tumor promoter are shown in green. CSD, caveolin-1 scaffolding domain; pY14-CAV1, phosphorylated caveolin-1; MVB, multivesicular body; MAMs, mitochondria-associated membranes. For protein abbreviations and more details, see the respective sections of the main text
References
Anand, S., Samuel, M., Kumar, S., & Mathivanan, S. (2019). Ticket to a bubble ride: cargo sorting into exosomes and extracellular vesicles. Biochimica et Biophysica Acta. Proteins and Proteomics, 1867(12), 140203 https://doi.org/10.1016/j.bbapap.2019.02.005.
Antalis, C. J., Arnold, T., Rasool, T., Lee, B., Buhman, K. K., & Siddiqui, R. A. (2010). High ACAT1 expression in estrogen receptor negative basal-like breast cancer cells is associated with LDL-induced proliferation. Breast Cancer Research and Treatment, 122(3), 661–670 https://doi.org/10.1007/s10549-009-0594-8.
Antalis, C. J., Uchida, A., Buhman, K. K., & Siddiqui, R. A. (2011). Migration of MDA-MB-231 breast cancer cells depends on the availability of exogenous lipids and cholesterol esterification. Clinical & Experimental Metastasis, 28(8), 733–741 https://doi.org/10.1007/s10585-011-9405-9.
Ariotti, N., Fernández-Rojo, M. A., Zhou, Y., Hill, M. M., Rodkey, T. L., Inder, K. L., Tanner, L. B., Wenk, M. R., Hancock, J. F., & Parton, R. G. (2014). Caveolae regulate the nanoscale organization of the plasma membrane to remotely control Ras signaling. The Journal of Cell Biology, 204(5), 777–792 https://doi.org/10.1083/jcb.201307055.
Ariotti, N., Rae, J., Leneva, N., Ferguson, C., Loo, D., Okano, S., Hill, M. M., Walser, P., Collins, B. M., & Parton, R. G. (2015). Molecular characterization of caveolin-induced membrane curvature. Journal of Biological Chemistry, 290(41), 24875–24890 https://doi.org/10.1074/jbc.M115.644336.
Arpaia, E., Blaser, H., Quintela-Fandino, M., Duncan, G., Leong, H. S., Ablack, A., Nambiar, S. C., Lind, E. F., Silvester, J., Fleming, C. K., Rufini, A., Tusche, M. W., Brüstle, A., Ohashi, P. S., Lewis, J. D., & Mak, T. W. (2012). The interaction between caveolin-1 and Rho-GTPases promotes metastasis by controlling the expression of alpha5-integrin and the activation of Src, Ras and Erk. Oncogene, 31(7), 884–896 https://doi.org/10.1038/onc.2011.288.
Asterholm, I. W., Mundy, D. I., Weng, J., Anderson, R. G. W., & Scherer, P. E. (2012). Altered mitochondrial function and metabolic inflexibility associated with loss of caveolin-1. Cell Metabolism, 15(2), 171–185 https://doi.org/10.1016/j.cmet.2012.01.004.
Aung, C. S., Hill, M. M., Bastiani, M., Parton, R. G., & Parat, M.-O. (2011). PTRF–cavin-1 expression decreases the migration of PC3 prostate cancer cells: role of matrix metalloprotease 9. European Journal of Cell Biology, 90(2–3), 136–142 https://doi.org/10.1016/j.ejcb.2010.06.004.
Barth, J. M. I., & Köhler, K. (2014). How to take autophagy and endocytosis up a notch. BioMed Research International, 2014, 960803 https://doi.org/10.1155/2014/960803.
Bartz, R., Zhou, J., Hsieh, J.-T., Ying, Y., Li, W., & Liu, P. (2008). Caveolin-1 secreting LNCaP cells induce tumor growth of caveolin-1 negative LNCaP cells in vivo. International Journal of Cancer, 122(3), 520–525 https://doi.org/10.1002/ijc.23142.
Bastiani, M., Liu, L., Hill, M. M., Jedrychowski, M. P., Nixon, S. J., Lo, H. P., Abankwa, D., Luetterforst, R., Fernandez-Rojo, M., Breen, M. R., Gygi, S. P., Vinten, J., Walser, P. J., North, K. N., Hancock, J. F., Pilch, P. F., & Parton, R. G. (2009). MURC/cavin-4 and cavin family members form tissue-specific caveolar complexes. The Journal of Cell Biology, 185(7), 1259–1273 https://doi.org/10.1083/jcb.200903053.
Bender, F. C., Reymond, M. A., Bron, C., & Quest, A. F. (2000). Caveolin-1 levels are down-regulated in human colon tumors, and ectopic expression of caveolin-1 in colon carcinoma cell lines reduces cell tumorigenicity. Cancer Research, 60(20), 5870–5878.
Bonuccelli, G., Whitaker-Menezes, D., Castello-Cros, R., Pavlides, S., Pestell, R. G., Fatatis, A., Witkiewicz, A. K., Vander Heiden, M. G., Migneco, G., Chiavarina, B., Frank, P. G., Capozza, F., Flomenberg, N., Martinez-Outschoorn, U. E., Sotgia, F., & Lisanti, M. P. (2010). The reverse Warburg effect: glycolysis inhibitors prevent the tumor promoting effects of caveolin-1 deficient cancer associated fibroblasts. Cell Cycle (Georgetown, Texas), 9(10), 1960–1971 https://doi.org/10.4161/cc.9.10.11601.
Bosch, M., Marí, M., Herms, A., Fernández, A., Fajardo, A., Kassan, A., Giralt, A., Colell, A., Balgoma, D., Barbero, E., González-Moreno, E., Matias, N., Tebar, F., Balsinde, J., Camps, M., Enrich, C., Gross, S. P., García-Ruiz, C., Pérez-Navarro, E., et al. (2011). Caveolin-1 deficiency causes cholesterol-dependent mitochondrial dysfunction and apoptotic susceptibility. Current Biology: CB, 21(8), 681–686 https://doi.org/10.1016/j.cub.2011.03.030.
Boscher, C., & Nabi, I. R. (2013). Galectin-3- and phospho-caveolin-1-dependent outside-in integrin signaling mediates the EGF motogenic response in mammary cancer cells. Molecular Biology of the Cell, 24(13), 2134–2145 https://doi.org/10.1091/mbc.e13-02-0095.
Boucrot, E., Howes, M. T., Kirchhausen, T., & Parton, R. G. (2011). Redistribution of caveolae during mitosis. Journal of Cell Science, 124(Pt 12), 1965–1972 https://doi.org/10.1242/jcs.076570.
Bourseau-Guilmain, E., Menard, J. A., Lindqvist, E., Indira Chandran, V., Christianson, H. C., Cerezo Magaña, M., Lidfeldt, J., Marko-Varga, G., Welinder, C., & Belting, M. (2016). Hypoxia regulates global membrane protein endocytosis through caveolin-1 in cancer cells. Nature Communications, 7, 11371 https://doi.org/10.1038/ncomms11371.
Bravo-Sagua, R., Parra, V., López-Crisosto, C., Díaz, P., Quest, A. F. G., & Lavandero, S. (2017). Calcium transport and signaling in mitochondria. Comprehensive physiology 7(2), 623–634 https://doi.org/10.1002/cphy.c160013.
Bravo-Sagua, R., Parra, V., Ortiz-Sandoval, C., Navarro-Marquez, M., Rodríguez, A. E., Diaz-Valdivia, N., Sanhueza, C., Lopez-Crisosto, C., Tahbaz, N., Rothermel, B. A., Hill, J. A., Cifuentes, M., Simmen, T., Quest, A. F. G., & Lavandero, S. (2019). Caveolin-1 impairs PKA-DRP1-mediated remodelling of ER-mitochondria communication during the early phase of ER stress. Cell Death and Differentiation, 26(7), 1195–1212 https://doi.org/10.1038/s41418-018-0197-1.
Byrne, D. P., Dart, C., & Rigden, D. J. (2012). Evaluating caveolin interactions: do proteins interact with the caveolin scaffolding domain through a widespread aromatic residue-rich motif? PLoS One, 7(9), e44879 https://doi.org/10.1371/journal.pone.0044879.
Cai, J., Zhao, X.-L., Liu, A.-W., Nian, H., & Zhang, S.-H. (2011). Apigenin inhibits hepatoma cell growth through alteration of gene expression patterns. Phytomedicine: International Journal of Phytotherapy and Phytopharmacology, 18(5), 366–373 https://doi.org/10.1016/j.phymed.2010.08.006.
Cai, M., Sun, X., Wang, W., Lian, Z., Wu, P., Han, S., Chen, H., & Zhang, P. (2018). Disruption of peroxisome function leads to metabolic stress, mTOR inhibition, and lethality in liver cancer cells. Cancer Letters, 421, 82–93 https://doi.org/10.1016/j.canlet.2018.02.021.
Campos, A., Salomon, C., Bustos, R., Díaz, J., Martínez, S., Silva, V., Reyes, C., Díaz-Valdivia, N., Varas-Godoy, M., Lobos-González, L., & Quest, A. F. (2018). Caveolin-1-containing extracellular vesicles transport adhesion proteins and promote malignancy in breast cancer cell lines. Nanomedicine (London, England), 13(20), 2597–2609 https://doi.org/10.2217/nnm-2018-0094.
Campos, A., Burgos-Ravanal, R., González, M. F., Huilcaman, R., Lobos González, L., & Quest, A. F. G. (2019). Cell intrinsic and extrinsic mechanisms of caveolin-1-enhanced metastasis. Biomolecules, 9(8) https://doi.org/10.3390/biom9080314.
Capiod, T. (2016). Extracellular calcium has multiple targets to control cell proliferation. Advances in Experimental Medicine and Biology, 898, 133–156 https://doi.org/10.1007/978-3-319-26974-0_7.
Cárdenas, C., Miller, R. A., Smith, I., Bui, T., Molgó, J., Müller, M., Vais, H., Cheung, K.-H., Yang, J., Parker, I., Thompson, C. B., Birnbaum, M. J., Hallows, K. R., & Foskett, J. K. (2010). Essential regulation of cell bioenergetics by constitutive InsP3 receptor Ca2+ transfer to mitochondria. Cell, 142(2), 270–283 https://doi.org/10.1016/j.cell.2010.06.007.
Chatterjee, M., Ben-Josef, E., Robb, R., Vedaie, M., Seum, S., Thirumoorthy, K., Palanichamy, K., Harbrecht, M., Chakravarti, A., & Williams, T. M. (2017). Caveolae-mediated endocytosis is critical for albumin cellular uptake and response to albumin-bound chemotherapy. Cancer Research, 77(21), 5925–5937 https://doi.org/10.1158/0008-5472.CAN-17-0604.
Chen, Y.-G. (2009). Endocytic regulation of TGF-beta signaling. Cell Research, 19(1), 58–70 https://doi.org/10.1038/cr.2008.315.
Cheng, J. P. X., & Nichols, B. J. (2016). Caveolae: one function or many? Trends in Cell Biology, 26(3), 177–189 https://doi.org/10.1016/j.tcb.2015.10.010.
Choudhury, A., Marks, D. L., Proctor, K. M., Gould, G. W., & Pagano, R. E. (2006). Regulation of caveolar endocytosis by syntaxin 6-dependent delivery of membrane components to the cell surface. Nature Cell Biology, 8(4), 317–328 https://doi.org/10.1038/ncb1380.
Chung, Y.-C., Kuo, J.-F., Wei, W.-C., Chang, K.-J., & Chao, W.-T. (2015). Caveolin-1 dependent endocytosis enhances the chemosensitivity of HER-2 positive breast cancer cells to trastuzumab emtansine (T-DM1). PLoS One, 10(7), e0133072 https://doi.org/10.1371/journal.pone.0133072.
Cohen, A. W., Schubert, W., Brasaemle, D. L., Scherer, P. E., & Lisanti, M. P. (2005). Caveolin-1 expression is essential for proper nonshivering thermogenesis in brown adipose tissue. Diabetes, 54(3), 679–686 https://doi.org/10.2337/diabetes.54.3.679.
Collins, B. M., Davis, M. J., Hancock, J. F., & Parton, R. G. (2012). Structure-based reassessment of the caveolin signaling model: do caveolae regulate signaling through caveolin-protein interactions? Developmental Cell, 23(1), 11–20 https://doi.org/10.1016/j.devcel.2012.06.012.
Czernek, L., & Düchler, M. (2017). Functions of cancer-derived extracellular vesicles in immunosuppression. Archivum Immunologiae et Therapiae Experimentalis, 65(4), 311–323 https://doi.org/10.1007/s00005-016-0453-3.
Deb, M., Sengupta, D., Kar, S., Rath, S. K., Roy, S., Das, G., & Patra, S. K. (2016). Epigenetic drift towards histone modifications regulates CAV1 gene expression in colon cancer. Gene, 581(1), 75–84 https://doi.org/10.1016/j.gene.2016.01.029.
Di Guglielmo, G. M., Le Roy, C., Goodfellow, A. F., & Wrana, J. L. (2003). Distinct endocytic pathways regulate TGF-beta receptor signalling and turnover. Nature Cell Biology, 5(5), 410–421 https://doi.org/10.1038/ncb975.
Díaz, J., Mendoza, P., Ortiz, R., Díaz, N., Leyton, L., Stupack, D., Quest, A. F. G., & Torres, V. A. (2014). Rab5 is required in metastatic cancer cells for caveolin-1-enhanced Rac1 activation, migration and invasion. Journal of Cell Science, 127(Pt 11), 2401–2406 https://doi.org/10.1242/jcs.141689.
Díaz-Valdivia, N. I., Calderón, C. C., Díaz, J. E., Lobos-González, L., Sepulveda, H., Ortíz, R. J., Martinez, S., Silva, V., Maldonado, H. J., Silva, P., Wehinger, S., Burzio, V. A., Torres, V. A., Montecino, M., Leyton, L., & Quest, A. F. G. (2017). Anti-neoplastic drugs increase caveolin-1-dependent migration, invasion and metastasis of cancer cells. Oncotarget, 8(67), 111943–111965. https://doi.org/10.18632/oncotarget.22955.
Fairbank, M., St-Pierre, P., & Nabi, I. R. (2009). The complex biology of autocrine motility factor/phosphoglucose isomerase (AMF/PGI) and its receptor, the gp78/AMFR E3 ubiquitin ligase. Molecular BioSystems, 5(8), 793–801 https://doi.org/10.1039/b820820b.
Felicetti, F., Parolini, I., Bottero, L., Fecchi, K., Errico, M. C., Raggi, C., Biffoni, M., Spadaro, F., Lisanti, M. P., Sargiacomo, M., & Carè, A. (2009). Caveolin-1 tumor-promoting role in human melanoma. International Journal of Cancer, 125(7), 1514–1522 https://doi.org/10.1002/ijc.24451.
Filippin, L., Magalhães, P. J., Di Benedetto, G., Colella, M., & Pozzan, T. (2003). Stable interactions between mitochondria and endoplasmic reticulum allow rapid accumulation of calcium in a subpopulation of mitochondria. Journal of Biological Chemistry, 278(40), 39224–39234 https://doi.org/10.1074/jbc.M302301200.
Frank, P. G., Pavlides, S., Cheung, M. W.-C., Daumer, K., & Lisanti, M. P. (2008). Role of caveolin-1 in the regulation of lipoprotein metabolism. American Journal of Physiology. Cell Physiology, 295(1), C242–C248 https://doi.org/10.1152/ajpcell.00185.2008.
Fridolfsson, H. N., Kawaraguchi, Y., Ali, S. S., Panneerselvam, M., Niesman, I. R., Finley, J. C., Kellerhals, S. E., Migita, M. Y., Okada, H., Moreno, A. L., Jennings, M., Kidd, M. W., Bonds, J. A., Balijepalli, R. C., Ross, R. S., Patel, P. M., Miyanohara, A., Chen, Q., Lesnefsky, E. J., et al. (2012). Mitochondria-localized caveolin in adaptation to cellular stress and injury. FASEB Journal : Official Publication of the Federation of American Societies for Experimental Biology, 26(11), 4637–4649 https://doi.org/10.1096/fj.12-215798.
Fridolfsson, H. N., Roth, D. M., Insel, P. A., & Patel, H. H. (2014). Regulation of intracellular signaling and function by caveolin. The FASEB Journal, 28(9), 3823–3831 https://doi.org/10.1096/fj.14-252320.
Galbiati, F., Volonte, D., Brown, A. M., Weinstein, D. E., Ben-Ze’ev, A., Pestell, R. G., & Lisanti, M. P. (2000). Caveolin-1 expression inhibits Wnt/beta-catenin/Lef-1 signaling by recruiting beta-catenin to caveolae membrane domains. The Journal of Biological Chemistry, 275(30), 23368–23377 https://doi.org/10.1074/jbc.M002020200.
Gandre-Babbe, S., & van der Bliek, A. M. (2008). The novel tail-anchored membrane protein Mff controls mitochondrial and peroxisomal fission in mammalian cells. Molecular Biology of the Cell, 19(6), 2402–2412 https://doi.org/10.1091/mbc.e07-12-1287.
Gao, Y., Li, L., Li, T., Ma, L., Yuan, M., Sun, W., Cheng, H. L., Niu, L., Du, Z., Quan, Z., Fan, Y., Fan, J., Luo, C., & Wu, X. (2019). Simvastatin delays castration-resistant prostate cancer metastasis and androgen receptor antagonist resistance by regulating the expression of caveolin-1. International Journal of Oncology, 54(6), 2054–2068 https://doi.org/10.3892/ijo.2019.4774.
Gaus, K., Le Lay, S., Balasubramanian, N., & Schwartz, M. A. (2006). Integrin-mediated adhesion regulates membrane order. Journal of Cell Biology, 174(5), 725–734 https://doi.org/10.1083/jcb.200603034.
Geletu, M., Mohan, R., Arulanandam, R., Berger-Becvar, A., Nabi, I. R., Gunning, P. T., & Raptis, L. (2018). Reciprocal regulation of the cadherin-11/Stat3 axis by caveolin-1 in mouse fibroblasts and lung carcinoma cells. Biochimica Et Biophysica Acta. Molecular Cell Research, 1865(5), 794–802 https://doi.org/10.1016/j.bbamcr.2018.02.004.
Gottlieb-Abraham, E., Shvartsman, D. E., Donaldson, J. C., Ehrlich, M., Gutman, O., Martin, G. S., & Henis, Y. I. (2013). Src-mediated caveolin-1 phosphorylation affects the targeting of active Src to specific membrane sites. Molecular Biology of the Cell, 24(24), 3881–3895 https://doi.org/10.1091/mbc.E13-03-0163.
Guo, Y.-L., Zhu, T.-N., Guo, W., Dong, Z.-M., Zhou, Z., Cui, Y.-J., & Zhao, R.-J. (2016). Aberrant CpG island shore region methylation of CAV1 is associated with tumor progression and poor prognosis in gastric cardia adenocarcinoma. Archives of Medical Research, 47(6), 460–470 https://doi.org/10.1016/j.arcmed.2016.10.005.
Gupta, R., Toufaily, C., & Annabi, B. (2014). Caveolin and cavin family members: dual roles in cancer. Biochimie, 107(Pt B), 188–202 https://doi.org/10.1016/j.biochi.2014.09.010.
Gupta, V. K., Sharma, N. S., Kesh, K., Dauer, P., Nomura, A., Giri, B., Dudeja, V., Banerjee, S., Bhattacharya, S., Saluja, A., & Banerjee, S. (2018). Metastasis and chemoresistance in CD133 expressing pancreatic cancer cells are dependent on their lipid raft integrity. Cancer Letters, 439, 101–112 https://doi.org/10.1016/j.canlet.2018.09.028.
Guruswamy, S., & Rao, C. V. (2009). Synergistic effects of lovastatin and celecoxib on caveolin-1 and its down-stream signaling molecules: implications for colon cancer prevention. International Journal of Oncology, 35(5), 1037–1043 https://doi.org/10.3892/ijo_00000418.
Ha, T.-K., Her, N.-G., Lee, M.-G., Ryu, B.-K., Lee, J.-H., Han, J., Jeong, S.-I., Kang, M.-J., Kim, N.-H., Kim, H.-J., & Chi, S.-G. (2012). Caveolin-1 increases aerobic glycolysis in colorectal cancers by stimulating HMGA1-mediated GLUT3 transcription. Cancer Research, 72(16), 4097–4109 https://doi.org/10.1158/0008-5472.CAN-12-0448.
Han, T., Kang, D., Ji, D., Wang, X., Zhan, W., Fu, M., ** in touch with the membrane; protein- and lipid-mediated confinement of caveolae to the cell surface. Biochemical Society Transactions, 48(1), 155–163 https://doi.org/10.1042/BST20190386.
Huertas-Martínez, J., Rello-Varona, S., Herrero-Martín, D., Barrau, I., García-Monclús, S., Sáinz-Jaspeado, M., Lagares-Tena, L., Núñez-Álvarez, Y., Mateo-Lozano, S., Mora, J., Roma, J., Toran, N., Moran, S., López-Alemany, R., Gallego, S., Esteller, M., Peinado, M. A., Del Muro, X. G., & Tirado, O. M. (2014). Caveolin-1 is down-regulated in alveolar rhabdomyosarcomas and negatively regulates tumor growth. Oncotarget, 5(20), 9744–9755. https://doi.org/10.18632/oncotarget.2403.
Huertas-Martínez, J., Court, F., Rello-Varona, S., Herrero-Martín, D., Almacellas-Rabaiget, O., Sáinz-Jaspeado, M., Garcia-Monclús, S., Lagares-Tena, L., Buj, R., Hontecillas-Prieto, L., Sastre, A., Azorin, D., Sanjuan, X., López-Alemany, R., Moran, S., Roma, J., Gallego, S., Mora, J., García Del Muro, X., et al. (2017). DNA methylation profiling identifies PTRF/cavin-1 as a novel tumor suppressor in Ewing sarcoma when co-expressed with caveolin-1. Cancer Letters, 386, 196–207 https://doi.org/10.1016/j.canlet.2016.11.020.
Indira Chandran, V., Månsson, A.-S., Barbachowska, M., Cerezo-Magaña, M., Nodin, B., Joshi, B., Koppada, N., Saad, O. M., Gluz, O., Isaksson, K., Borgquist, S., Jirström, K., Nabi, I. R., Jernström, H., & Belting, M. (2020). Hypoxia attenuates trastuzumab uptake and trastuzumab-emtansine (T-DM1) cytotoxicity through redistribution of phosphorylated caveolin-1. Molecular Cancer Research, 18(4), 644–656 https://doi.org/10.1158/1541-7786.MCR-19-0856.
Ingelmo-Torres, M., Gonza’lez-Moreno, E., Kassan, A., Hanzal-Bayer, M., Tebar, F., Herms, A., Grewal, T., Hancock, J. F., Enrich, C., Bosch, M., Gross, S. P., Parton, R. G., & Pol, A. (2009). Hydrophobic and basic domains target proteins to lipid droplets. Traffic, 10(12), 1785–1801 https://doi.org/10.1111/j.1600-0854.2009.00994.x.
Joshi, B., Strugnell, S. S., Goetz, J. G., Kojic, L. D., Cox, M. E., Griffith, O. L., Chan, S. K., Jones, S. J., Leung, S.-P., Masoudi, H., Leung, S., Wiseman, S. M., & Nabi, I. R. (2008). Phosphorylated caveolin-1 regulates rho/ROCK-dependent focal adhesion dynamics and tumor cell migration and invasion. Cancer Research, 68(20), 8210–8220 https://doi.org/10.1158/0008-5472.CAN-08-0343.
Kahlert, C., & Kalluri, R. (2013). Exosomes in tumor microenvironment influence cancer progression and metastasis. Journal of Molecular Medicine (Berlin, Germany), 91(4), 431–437 https://doi.org/10.1007/s00109-013-1020-6.
Karam, J. A., Lotan, Y., Roehrborn, C. G., Ashfaq, R., Karakiewicz, P. I., & Shariat, S. F. (2007). Caveolin-1 overexpression is associated with aggressive prostate cancer recurrence. The Prostate, 67(6), 614–622 https://doi.org/10.1002/pros.20557.
Kassan, A., Herms, A., Fernández-Vidal, A., Bosch, M., Schieber, N. L., Reddy, B. J. N., Fajardo, A., Gelabert-Baldrich, M., Tebar, F., Enrich, C., Gross, S. P., Parton, R. G., & Pol, A. (2013). Acyl-CoA synthetase 3 promotes lipid droplet biogenesis in ER microdomains. The Journal of Cell Biology, 203(6), 985–1001 https://doi.org/10.1083/jcb.201305142.
Koch, J., & Brocard, C. (2012). PEX11 proteins attract Mff and human Fis1 to coordinate peroxisomal fission. Journal of Cell Science, 125(Pt 16), 3813–3826 https://doi.org/10.1242/jcs.102178.
Kojic, L. D., Joshi, B., Lajoie, P., Le, P. U., Cox, M. E., Turbin, D. A., Wiseman, S. M., & Nabi, I. R. (2007). Raft-dependent endocytosis of autocrine motility factor is phosphatidylinositol 3-kinase-dependent in breast carcinoma cells. The Journal of Biological Chemistry, 282(40), 29305–29313 https://doi.org/10.1074/jbc.M704069200.
Lajoie, P., Goetz, J. G., Dennis, J. W., & Nabi, I. R. (2009). Lattices, rafts, and scaffolds: Domain regulation of receptor signaling at the plasma membrane. The Journal of Cell Biology, 185(3), 381–385 https://doi.org/10.1083/jcb.200811059.
Lajoie, P., Goetz, J. G., Dennis, J. W., & Nabi, I. R. (2009). Lattices, rafts, and scaffolds: Domain regulation of receptor signaling at the plasma membrane. The Journal of Cell Biology, 185(3), 381–385 https://doi.org/10.1083/jcb.200811059.
Lara, P., Palma-Florez, S., Salas-Huenuleo, E., Polakovicova, I., Guerrero, S., Lobos-Gonzalez, L., Campos, A., Muñoz, L., Jorquera-Cordero, C., Varas-Godoy, M., Cancino, J., Arias, E., Villegas, J., Cruz, L. J., Albericio, F., Araya, E., Corvalan, A. H., Quest, A. F. G., & Kogan, M. J. (2020). Gold nanoparticle based double-labeling of melanoma extracellular vesicles to determine the specificity of uptake by cells and preferential accumulation in small metastatic lung tumors. Journal of Nanobiotechnology, 18(1), 20 https://doi.org/10.1186/s12951-020-0573-0.
Le, P. U., Guay, G., Altschuler, Y., & Nabi, I. R. (2002). Caveolin-1 is a negative regulator of caveolae-mediated endocytosis to the endoplasmic reticulum. The Journal of Biological Chemistry, 277(5), 3371–3379 https://doi.org/10.1074/jbc.M111240200.
Le, P. U., Guay, G., Altschuler, Y., & Nabi, I. R. (2002). Caveolin-1 is a negative regulator of caveolae-mediated endocytosis to the endoplasmic reticulum. The Journal of Biological Chemistry, 277(5), 3371–3379 https://doi.org/10.1074/jbc.M111240200.
Li, J., Gu, D., Lee, S. S.-Y., Song, B., Bandyopadhyay, S., Chen, S., Konieczny, S. F., Ratliff, T. L., Liu, X., ** in on lipid droplets: insights into cellular stress and cancer. Bioscience Reports, 38(5) https://doi.org/10.1042/BSR20180764.
Sinha, B., Köster, D., Ruez, R., Gonnord, P., Bastiani, M., Abankwa, D., Stan, R. V., Butler-Browne, G., Vedie, B., Johannes, L., Morone, N., Parton, R. G., Raposo, G., Sens, P., Lamaze, C., & Nassoy, P. (2011). Cells respond to mechanical stress by rapid disassembly of caveolae. Cell, 144(3), 402–413 https://doi.org/10.1016/j.cell.2010.12.031.
Sonveaux, P., Végran, F., Schroeder, T., Wergin, M. C., Verrax, J., Rabbani, Z. N., De Saedeleer, C. J., Kennedy, K. M., Diepart, C., Jordan, B. F., Kelley, M. J., Gallez, B., Wahl, M. L., Feron, O., & Dewhirst, M. W. (2008). Targeting lactate-fueled respiration selectively kills hypoxic tumor cells in mice. The Journal of Clinical Investigation, 118(12), 3930–3942 https://doi.org/10.1172/JCI36843.
Sugiura, A., Mattie, S., Prudent, J., & McBride, H. M. (2017). Newly born peroxisomes are a hybrid of mitochondrial and ER-derived pre-peroxisomes. Nature, 542(7640), 251–254 https://doi.org/10.1038/nature21375.
Sundivakkam, P. C., Kwiatek, A. M., Sharma, T. T., Minshall, R. D., Malik, A. B., & Tiruppathi, C. (2009). Caveolin-1 scaffold domain interacts with TRPC1 and IP3R3 to regulate Ca2+ store release-induced Ca2+ entry in endothelial cells. American Journal of Physiology. Cell Physiology, 296(3), C403–C413 https://doi.org/10.1152/ajpcell.00470.2008.
Tagawa, A., Mezzacasa, A., Hayer, A., Longatti, A., Pelkmans, L., & Helenius, A. (2005). Assembly and trafficking of caveolar domains in the cell. The Journal of Cell Biology, 170(5), 769–779 https://doi.org/10.1083/jcb.200506103.
Tahir, S. A., Yang, G., Ebara, S., Timme, T. L., Satoh, T., Li, L., Goltsov, A., Ittmann, M., Morrisett, J. D., & Thompson, T. C. (2001). Secreted caveolin-1 stimulates cell survival/clonal growth and contributes to metastasis in androgen-insensitive prostate cancer. Cancer Research, 61(10), 3882–3885.
Territo, P. R., Mootha, V. K., French, S. A., & Balaban, R. S. (2000). Ca(2+) activation of heart mitochondrial oxidative phosphorylation: role of the F(0)/F(1)-ATPase. American Journal of Physiology. Cell Physiology, 278(2), C423–C435 https://doi.org/10.1152/ajpcell.2000.278.2.C423.
Torrejón, B., Cristóbal, I., Rojo, F., & García-Foncillas, J. (2017). Caveolin-1 is markedly downregulated in patients with early-stage colorectal cancer. World Journal of Surgery, 41(10), 2625–2630 https://doi.org/10.1007/s00268-017-4065-9.
Torres, V. A., Tapia, J. C., Rodríguez, D. A., Párraga, M., Lisboa, P., Montoya, M., Leyton, L., & Quest, A. F. G. (2006). Caveolin-1 controls cell proliferation and cell death by suppressing expression of the inhibitor of apoptosis protein survivin. Journal of Cell Science, 119(Pt 9), 1812–1823 https://doi.org/10.1242/jcs.02894.
Torres, V. A., Tapia, J. C., Rodriguez, D. A., Lladser, A., Arredondo, C., Leyton, L., & Quest, A. F. G. (2007). E-cadherin is required for caveolin-1-mediated down-regulation of the inhibitor of apoptosis protein survivin via reduced beta-catenin-Tcf/Lef-dependent transcription. Molecular and Cellular Biology, 27(21), 7703–7717 https://doi.org/10.1128/MCB.01991-06.
Tsutsumi, S., Hogan, V., Nabi, I. R., & Raz, A. (2003). Overexpression of the autocrine motility factor/phosphoglucose isomerase induces transformation and survival of NIH-3T3 fibroblasts. Cancer Research, 63(1), 242–249.
Urra, H., Torres, V. A., Ortiz, R. J., Lobos, L., Díaz, M. I., Díaz, N., Härtel, S., Leyton, L., & Quest, A. F. G. (2012). Caveolin-1-enhanced motility and focal adhesion turnover require tyrosine-14 but not accumulation to the rear in metastatic cancer cells. PLoS One, 7(4), e33085 https://doi.org/10.1371/journal.pone.0033085.
van den Bosch, H., Schrakamp, G., Hardeman, D., Zomer, A. W., Wanders, R. J., & Schutgens, R. B. (1993). Ether lipid synthesis and its deficiency in peroxisomal disorders. Biochimie, 75(3–4), 183–189 https://doi.org/10.1016/0300-9084(93)90076-5.
van Deurs, B., Roepstorff, K., Hommelgaard, A. M., & Sandvig, K. (2003). Caveolae: anchored, multifunctional platforms in the lipid ocean. Trends in Cell Biology, 13(2), 92–100 https://doi.org/10.1016/s0962-8924(02)00039-9.
van Niel, G., D’Angelo, G., & Raposo, G. (2018). Shedding light on the cell biology of extracellular vesicles. Nature Reviews. Molecular Cell Biology, 19(4), 213–228 https://doi.org/10.1038/nrm.2017.125.
Vander Heiden, M. G., Cantley, L. C., & Thompson, C. B. (2009). Understanding the Warburg effect: the metabolic requirements of cell proliferation. Science, 324(5930), 1029–1033 https://doi.org/10.1126/science.1160809.
Volonte, D., Vyas, A. R., Chen, C., Dacic, S., Stabile, L. P., Kurland, B. F., Abberbock, S. R., Burns, T. F., Herman, J. G., Di, Y. P., & Galbiati, F. (2018). Caveolin-1 promotes the tumor suppressor properties of oncogene-induced cellular senescence. The Journal of Biological Chemistry, 293(5), 1794–1809 https://doi.org/10.1074/jbc.M117.815902.
Walser, P. J., Ariotti, N., Howes, M., Ferguson, C., Webb, R., Schwudke, D., Leneva, N., Cho, K.-J., Cooper, L., Rae, J., Floetenmeyer, M., Oorschot, V. M. J., Skoglund, U., Simons, K., Hancock, J. F., & Parton, R. G. (2012). Constitutive formation of caveolae in a bacterium. Cell, 150(4), 752–763 https://doi.org/10.1016/j.cell.2012.06.042.
Wang, B., Xu, X., Yang, Z., Zhang, L., Liu, Y., Ma, A., Xu, G., Tang, M., **g, T., Wu, L., & Liu, Y. (2019). POH1 contributes to hyperactivation of TGF-β signaling and facilitates hepatocellular carcinoma metastasis through deubiquitinating TGF-β receptors and caveolin-1. EBioMedicine, 41, 320–332 https://doi.org/10.1016/j.ebiom.2019.01.058.
Wang, K., Zhu, X., Mei, D., & Ding, Z. (2018). Caveolin-1 contributes to anoikis resistance in human gastric cancer SGC-7901 cells via regulating Src-dependent EGFR-ITGB1 signaling. Journal of Biochemical and Molecular Toxicology, 32(10), e22202 https://doi.org/10.1002/jbt.22202.
Warburg, O., Wind, F., & Negelein, E. (1927). The metabolism of tumors in the body. The Journal of General Physiology, 8(6), 519–530 https://doi.org/10.1085/jgp.8.6.519.
Wiechen, K., Sers, C., Agoulnik, A., Arlt, K., Dietel, M., Schlag, P. M., & Schneider, U. (2001). Down-regulation of caveolin-1, a candidate tumor suppressor gene, in sarcomas. The American Journal of Pathology, 158(3), 833–839 https://doi.org/10.1016/S0002-9440(10)64031-X.
Witkiewicz, A. K., Whitaker-Menezes, D., Dasgupta, A., Philp, N. J., Lin, Z., Gandara, R., Sneddon, S., Martinez-Outschoorn, U. E., Sotgia, F., & Lisanti, M. P. (2012). Using the “reverse Warburg effect” to identify high-risk breast cancer patients: stromal MCT4 predicts poor clinical outcome in triple-negative breast cancers. Cell Cycle (Georgetown, Texas), 11(6), 1108–1117 https://doi.org/10.4161/cc.11.6.19530.
Woudenberg, J., Rembacz, K. P., van den Heuvel, F. A. J., Woudenberg-Vrenken, T. E., Buist-Homan, M., Geuken, M., Hoekstra, M., Deelman, L. E., Enrich, C., Henning, R. H., Moshage, H., & Faber, K. N. (2010). Caveolin-1 is enriched in the peroxisomal membrane of rat hepatocytes. Hepatology (Baltimore, Md.), 51(5), 1744–1753 https://doi.org/10.1002/hep.23460.
Yang, H., Guan, L., Li, S., Jiang, Y., **ong, N., Li, L., Wu, C., Zeng, H., & Liu, Y. (2016). Mechanosensitive caveolin-1 activation-induced PI3K/Akt/mTOR signaling pathway promotes breast cancer motility, invadopodia formation and metastasis in vivo. Oncotarget, 7(13), 16227–16247. https://doi.org/10.18632/oncotarget.7583.
Yeong, J., Thike, A. A., Ikeda, M., Lim, J. C. T., Lee, B., Nakamura, S., Iqbal, J., & Tan, P. H. (2018). Caveolin-1 expression as a prognostic marker in triple negative breast cancers of Asian women. Journal of Clinical Pathology, 71(2), 161–167 https://doi.org/10.1136/jclinpath-2017-204495.
Yoon, Y., Krueger, E. W., Oswald, B. J., & McNiven, M. A. (2003). The mitochondrial protein hFis1 regulates mitochondrial fission in mammalian cells through an interaction with the dynamin-like protein DLP1. Molecular and Cellular Biology, 23(15), 5409–5420 https://doi.org/10.1128/mcb.23.15.5409-5420.2003.
Yu, D.-M., Jung, S. H., An, H.-T., Lee, S., Hong, J., Park, J. S., Lee, H., Lee, H., Bahn, M.-S., Lee, H. C., Han, N.-K., Ko, J., Lee, J.-S., & Ko, Y.-G. (2017). Caveolin-1 deficiency induces premature senescence with mitochondrial dysfunction. Aging Cell, 16(4), 773–784 https://doi.org/10.1111/acel.12606.
Zhou, M., Chinnaiyan, A. M., Kleer, C. G., Lucas, P. C., & Rubin, M. A. (2002). Alpha-methylacyl-CoA racemase: a novel tumor marker over-expressed in several human cancers and their precursor lesions. The American Journal of Surgical Pathology, 26(7), 926–931 https://doi.org/10.1097/00000478-200207000-00012.
Zimnicka, A. M., Husain, Y. S., Shajahan, A. N., Sverdlov, M., Chaga, O., Chen, Z., Toth, P. T., Klomp, J., Karginov, A. V., Tiruppathi, C., Malik, A. B., & Minshall, R. D. (2016). Src-dependent phosphorylation of caveolin-1 Tyr-14 promotes swelling and release of caveolae. Molecular Biology of the Cell, 27(13), 2090–2106 https://doi.org/10.1091/mbc.E15-11-0756.
Zoeller, R. A., Lake, A. C., Nagan, N., Gaposchkin, D. P., Legner, M. A., & Lieberthal, W. (1999). Plasmalogens as endogenous antioxidants: somatic cell mutants reveal the importance of the vinyl ether. The Biochemical Journal, 338(Pt 3), 769–776.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Simón, L., Campos, A., Leyton, L. et al. Caveolin-1 function at the plasma membrane and in intracellular compartments in cancer. Cancer Metastasis Rev 39, 435–453 (2020). https://doi.org/10.1007/s10555-020-09890-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10555-020-09890-x