Abstract
Purpose
Tumor-infiltrating lymphocytes (TILs) have been positively correlated with response to systemic therapy for triple-negative and HER2 + subtypes and improved clinical outcomes in early breast cancer (BC). Less is known about TILs in metastatic sites, particularly brain metastases (BM), where unique immune regulation governs stromal composition. Reactive glial cells actively participate in cytokine-mediated T cell stimulation. The impact of prior medical therapy (chemotherapy, endocrine, and HER2-targeted therapy) on the presence of TILs and gliosis in human breast cancer brain metastases (BCBM) has not been previously reported.
Methods
We examined prior treatment data for 133 patients who underwent craniotomy for resection of BMs from the electronic medical record. The primary endpoint was overall survival (OS) from the time of BM diagnosis. We examined the relationship between prior systemic therapy exposure and the histologic features of gliosis, necrosis, hemorrhage, and lymphocyte infiltration (LI) in BCBMs resected at subsequent craniotomy in univariate analyses.
Results
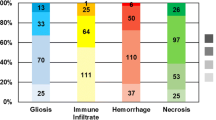
Complete treatment data were available for 123 patients. BCBM LI was identified in 35 of 116 (30%) patients who had received prior systemic treatment versus 5 of 7 (71.4%) who had not {significant by Fisher’s exact test p = 0.045}. There were no statistically significant relationships between prior systemic therapy and the three other histologic variables examined.
Conclusions
This observation suggests that systemic therapy may interfere with the immune response to BCBMs and cause exhaustion of anti-tumor immunity. This motivates clinical investigation of strategies to enhance LI for therapeutic benefit to improve outcomes for patients with BCBMs.


Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are not publicly available due to patient confidentiality but are available from the corresponding author on reasonable request.
Abbreviations
- BBB:
-
Blood–brain barrier
- BM:
-
Brain metastases
- BC:
-
Breast cancer
- BCBM:
-
Breast cancer brain metastases
- GPA:
-
Graded Prognostic Assessment
- H and E:
-
Hematoxylin and eosin
- HR:
-
Hormone receptor
- HER2:
-
Human epidermal growth factor receptor 2
- IL-1β:
-
Interleukin-1β
- IQR:
-
Interquartile range
- LI:
-
Lymphocyte infiltration
- MBC:
-
Metastatic breast cancer
- OS:
-
Overall survival
- PD-L1:
-
Programmed cell death ligand 1
- RT:
-
Radiotherapy
- SERPINS:
-
Serine protease inhibitors
- SRS:
-
Stereotactic radiosurgery
- TNBC:
-
Triple-negative breast cancer
- TILs:
-
Tumor-infiltrating lymphocytes
- USA:
-
United States of America
- WBRT:
-
Whole brain radiation therapy
References
Cancer of the Breast (Female) - Cancer Stat Facts SEER. [cited 2022 Aug 1]. Available from: https://seer.cancer.gov/statfacts/html/breast.html
Barnholtz-Sloan JS, Sloan AE, Davis FG, Vigneau FD, Lai P, Sawaya RE (2004) Incidence proportions of brain metastases in patients diagnosed (1973 to 2001) in the Metropolitan Detroit Cancer Surveillance System. J Clin Oncol 22:2865–2872. https://doi.org/10.1200/JCO.2004.12.149
Kennecke H, Yerushalmi R, Woods R, Cheang M, Voduc D, Speers CH, Nielsen TO, Gelmon K (2010) Metastatic Behavior of Breast Cancer Subtypes. JCO 28:3271–3277. https://doi.org/10.1200/JCO.2009.25.9820
Denkert C, von Minckwitz G, Darb-Esfahani S, Lederer B, Heppner BI et al (2018) Tumour-infiltrating lymphocytes and prognosis in different subtypes of breast cancer: a pooled analysis of 3771 patients treated with neoadjuvant therapy. Lancet Oncol 19:40–50. https://doi.org/10.1016/S1470-2045(17)30904-X
Hamilton R, Krauze M, Romkes M, Omolo B, Konstantinopoulos P et al (2013) Pathologic and gene expression features of metastatic melanomas to the brain (MBM). Cancer 119:2737–2746. https://doi.org/10.1002/cncr.28029
Sperduto PW, Mesko S, Li J, Cagney D, Aizer A et al (2020) Beyond an updated graded prognostic assessment (Breast GPA): a prognostic index and trends in treatment and survival in breast cancer brain metastases from 1985 to today. Int J Radiat Oncol Biol Phys. https://doi.org/10.1016/j.ijrobp.2020.01.051
Schreiber RD, Old LJ, Smyth MJ (2011) Cancer immunoediting: integrating immunity’s roles in cancer suppression and promotion. Science 331:1565–1570. https://doi.org/10.1126/science.1203486
Yang I, Tihan T, Han SJ, Wrensch MR, Wiencke J, Sughrue ME, Parsa AT (2010) CD8+ T-cell infiltrate in newly diagnosed glioblastoma is associated with long-term survival. J Clin Neurosci 17:1381–1385. https://doi.org/10.1016/j.jocn.2010.03.031
Ogiya R, Niikura N, Kumaki N, Bianchini G, Kitano S et al (2016) Comparison of tumor-infiltrating lymphocytes between primary and metastatic tumors in breast cancer patients. Cancer Sci 107:1730–1735. https://doi.org/10.1111/cas.13101
Duchnowska R, Pęksa R, Radecka B, Mandat T, Trojanowski T et al (2016) Immune response in breast cancer brain metastases and their microenvironment: the role of the PD-1/PD-L axis. Breast Cancer Res 18:43. https://doi.org/10.1186/s13058-016-0702-8
Cimino-Mathews A, Thompson E, Taube JM, Ye X, Lu Y et al (2016) PD-L1 (B7–H1) expression and the immune tumor microenvironment in primary and metastatic breast carcinomas. Hum Pathol 47:52–63. https://doi.org/10.1016/j.humpath.2015.09.003
Zhu X, Zhang Q, Wang D, Liu C, Han B, Yang JM (2019) Expression of PD-L1 attenuates the positive impacts of high-level tumor-infiltrating lymphocytes on prognosis of triple-negative breast cancer. Cancer Biol Ther 20(8):1105–1112. https://doi.org/10.1080/15384047.2019.1595282
Harter PN, Bernatz S, Scholz A, Zeiner PS, Zinke J et al (2015) Distribution and prognostic relevance of tumor-infiltrating lymphocytes (TILs) and PD-1/PD-L1 immune checkpoints in human brain metastases. Oncotarget 6:40836–40849. https://doi.org/10.18632/oncotarget.5696
Streit WJ, Conde JR, Fendrick SE, Flanary BE, Mariani CL (2005) Role of microglia in the central nervous system’s immune response. Neurol Res 27:685–691. https://doi.org/10.1179/016164105X49463a
Wasilewski D, Priego N, Fustero-Torre C, Valiente M (2017) Reactive astrocytes in brain metastasis. Front Oncol 7:298. https://doi.org/10.3389/fonc.2017.00298
Samdani AF, Kuchner EB, Rhines L, Adamson DC, Lawson C, Tyler B, Brem H, Dawson VL, Dawson TM (2004) Astroglia induce cytotoxic effects on brain tumors via a nitric oxide-dependent pathway both in vitro and in vivo. Neurosurgery. https://doi.org/10.1227/01.neu.0000119576.76193.b8
Sambade MJ, Prince G, Deal AM, Trembath D, McKee M et al (2019) Examination and prognostic implications of the unique microenvironment of breast cancer brain metastases. Breast Cancer Res Treat 176:321–328. https://doi.org/10.1007/s10549-019-05211-1
Fitzgerald DP, Palmieri D, Hua E, Hargrave E, Steeg PS et al (2008) Reactive glia are recruited by highly proliferative brain metastases of breast cancer and promote tumor cell colonization. Clin Exp Metastasis 25:799–810. https://doi.org/10.1007/s10585-008-9193-z
Schwartz H, Blacher E, Amer M, Livneh N, Abramovitz L et al (2016) Incipient melanoma brain metastases instigate astrogliosis and neuroinflammation. Cancer Res 76:4359–4371. https://doi.org/10.1158/0008-5472.CAN-16-0485
**ng F, Kobayashi A, Okuda H, Watabe M, Pai SK et al (2013) Reactive astrocytes promote the metastatic growth of breast cancer stem-like cells by activating Notch signalling in brain. EMBO Mol Med 5:384–396. https://doi.org/10.1002/emmm.201201623
Teng F, Mu D, Meng X, Kong L, Zhu H, Liu S, Zhang J, Yu J (2015) Tumor infiltrating lymphocytes (TILs) before and after neoadjuvant chemoradiotherapy and its clinical utility for rectal cancer. Am J Cancer Res 5:2064–2074
Tramm T, Vinter H, Vahl P, Özcan D, Alsner J, Overgaard J (2022) Tumor-infiltrating lymphocytes predict improved overall survival after post-mastectomy radiotherapy: a study of the randomized DBCG82bc cohort. Acta Oncol 61:153–162. https://doi.org/10.1080/0284186X.2021.1989629
Kovács A, Stenmark Tullberg A, Werner Rönnerman E, Holmberg E, Hartman L et al (2019) Effect of radiotherapy after breast-conserving surgery depending on the presence of tumor-infiltrating lymphocytes: a long-term follow-up of the SweBCG91RT randomized trial. J Clin Oncol 37:1179–1187. https://doi.org/10.1200/JCO.18.02157
Schmid P, Adams S, Rugo HS, Schneeweiss A, Barrios CH et al (2018) Atezolizumab and nab-paclitaxel in advanced triple-negative breast cancer. N Engl J Med 379:2108–2121. https://doi.org/10.1056/NEJMoa1809615
Cortes J, Cescon DW, Rugo HS, Nowecki Z, Im SA et al (2020) Pembrolizumab plus chemotherapy versus placebo plus chemotherapy for previously untreated locally recurrent inoperable or metastatic triple-negative breast cancer (KEYNOTE-355): a randomised, placebo-controlled, double-blind, phase 3 clinical trial. Lancet 396:1817–1828. https://doi.org/10.1016/S0140-6736(20)32531-9
Stereotactic Radiation and Nivolumab in the Management of Metastatic Breast Cancer Brain Metastases. [cited 2022 Aug 1]. Available from: https://clinicaltrials.gov/ct2/show/NCT03807765
Atezolizumab in Combination With Pertuzumab Plus High-dose Trastuzumab for the Treatment of Central Nervous System Metastases in Patients With Her2-positive Breast Cancer [cited 2022 Aug 1]. Available from: https://clinicaltrials.gov/ct2/show/NCT03449238
Pembrolizumab And Stereotactic Radiosurgery (SRS) Of Selected Brain Metastases In Breast Cancer Patients [cited 2022 Aug 1]. Available from: https://clinicaltrials.gov/ct2/show/NCT03417544
Zacharakis N, Huq LM, Seitter SJ, Kim SP, Gartner JJ et al (2022) Breast cancers are immunogenic: immunologic analyses and a phase ii pilot clinical trial using mutation-reactive autologous lymphocytes. J Clin Oncol. https://doi.org/10.1200/JCO.21.02170
O’Brien ER, Kersemans V, Tredwell M, Checa B, Serres S et al (2014) Glial activation in the early stages of brain metastasis: TSPO as a diagnostic biomarker. J Nucl Med 55(2):275–280. https://doi.org/10.2967/jnumed.113.127449
Acknowledgements
We would like to thank Dr Jedd Wolchok from Memorial Sloan Kettering Cancer Center, Immuno-Oncology Service, New York, USA, for his advice and comments.
Funding
This research received no external funding. Dr. Anders is a Translating Duke Health Scholar and is supported by the Translating Duke Health Initiative (TDHI).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by AB, SC, AD, MM, MS, and DT, Supervision was performed by CA, EB, and AS. Statistical analysis was performed by YC. The first draft of the manuscript was written by SC and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interests
CKA discloses Research funding: PUMA, Lilly, Merck, Seattle Genetics, Nektar, Tesaro, G1-Therapeutics, ZION, Novartis, Pfizer, Elucida; Compensated consultant role: Genentech, Eisai, IPSEN, Seattle Genetics, Astra Zeneca, Novartis, Immunomedics, Elucida, Athenex; Royalties: UpToDate, Jones and Bartlett. The other authors have no relevant financial or non-financial interests to disclose.
Ethical approval
The study was conducted according to the guidelines of the Declaration of Helsinki. Ethical review and approval were waived for this study, as this was a retrospective observational study of data obtained for clinical purposes. This study was registered under Memorial Sloan Kettering IRB waiver #WA0125-06.
Informed consent
Patient consent was waived due to retrospective data being collected anonymously for a quality improvement process that aimed to improve patient care and outcomes through systematic review of care against explicit criteria and standards.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The abstract of this paper was presented at the 2018 ASCO Annual Meeting as a poster presentation with interim findings. The abstract was published in Journal of Clinical Oncology 2018 36:15_suppl, 2063-2063. DOI: 10.1200/JCO.2018.36.15_suppl.2063 Journal of Clinical Oncology 36, no. 15_suppl (May 20, 2018) 2063-2063.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chew Minmin, S., Bacotti, A., Chen, Y. et al. Impact of prior systemic therapy on lymphocytic infiltration in surgically resected breast cancer brain metastases. Breast Cancer Res Treat 199, 99–107 (2023). https://doi.org/10.1007/s10549-023-06908-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-023-06908-0