Abstract
Purpose
To identify demographic and imaging features of MRI-detected enhancing lesions without clinical, ultrasound, and mammographic correlation associated with false-positive outcomes, impacting patient care.
Materials and methods
A retrospective multi-institutional study of imaging studies and patient’s chart review of consecutive women with MRI-detected enhancing lesions without clinical, mammogram, or ultrasound correlation between January and December 2018, who underwent MRI-guided biopsy. According to the BI-RADS lexicon, lesions’ frequency and imaging features were recorded. The demographic and imaging characteristics variables were correlated with histopathology as the gold standard and an uneventful follow-up of at least one year. Univariate logistic regression analysis was used to explore the correlation between the baseline variables such as age, genetic mutation, family history of breast cancer, personal history of breast cancer, MRI indication, background parenchymal enhancement, and MRI characteristic of the lesion with the false-positive results in main data and subgroup analysis.
Results
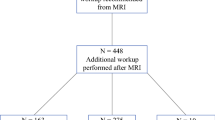
Two hundred nineteen women (median age 49 years; range 26–85 years) with 219 MRI-detected enhancing lesions that underwent MRI-guided vacuum-assisted biopsy during the study period fulfilled the study criteria and formed the study cohort. Out of 219, 180 lesions (82.2%) yielded benign pathology results, including 137 benign outcomes (76%) and 43 high-risk lesions (24%). Most demographic and imaging characteristics variables did not help to differentiate malignant from benign lesions. The variables that showed statistically significant association with true-positive results in univariate analyses were age (OR 1.05; 95% CI 1.02–1.08; p = 0.0015), irregular mass-lesion shape when compared with oval/round mass lesion (OR 11.2; 95% CI 1.6–78.4; p = 0.015), and clumped and clustered ring of enhancement when compared with homogeneous (OR 3.22, 95% CI 1.40–7.40; p = 0.0058). For participants with mass breast lesion, the hyperintense signal on the T2-weighted sequence (compared to the normal fibroglandular signal) was significantly related to the false-positive result (OR 0.13; 95% CI 0.02–0.76; p = 0.024).
Conclusion
Young patients, oval/round mass-lesion shape, and homogeneous pattern of non-mass enhancement showed the strongest association with false-positive results of enhancing lesions depicted by MRI. For participants with mass breast lesion, T2-bright mass lesion showed significant association with false-positive result. It may impact the patient’s management with a suggestion of follow-up rather than interventional procedure when these demographic and imaging parameters are present, consequently decreasing the patient’s anxiety and health care costs.




Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Kuhl CK, Weigel S, Schrading S et al (2010) Prospective multicenter cohort study to refine management recommendations for women at elevated familial risk of breast cancer: the EVA trial. J Clin Oncol 28(9):1450–1457
Lehman CD, Isaacs C, Schnall MD et al (2007) Cancer yield of mammography, MR, and US in high-risk women: prospective multi-institution breast cancer screening study. Radiology 244(2):381–388
Sardanelli F, Podo F, D’Agnolo G et al (2007) High breast cancer risk Italian trial. Multicenter comparative multimodality surveillance of women at genetic-familial high risk for breast cancer (HIBCRIT study): interim results. Radiology 242(3):698–715
Sung JS, Lee CH, Morris EA, Oeffinger KC, Dershaw DD (2011) Screening breast MR imaging in women with a history of chest irradiation. Radiology 259(1):65–71
Freitas V, Scaranelo A, Menezes R, Kulkarni S, Hodgson D, Crystal P (2013) Added cancer yield of breast magnetic resonance imaging screening in women with a prior history of chest radiation therapy. Cancer 119(3):495–503
Mendelson EB, Berg WA (2015) Training and standards for performance, interpretation, and structured reporting for supplemental breast cancer screening. AJR Am J Roentgenol 204:265–268
Health Quality Ontario (2016) Magnetic resonance imaging as an adjunct to mammography for breast cancer screening in women at less than high risk for breast cancer: a health technology assessment. Ont Health Technol Assess Ser 16(20):1–30
Kuhl CK, Strobel K, Bieling H, Leutner C, Schild HH, Schrading S (2017) Supplemental breast MR imaging screening of women with average risk of breast cancer. Radiology 283:361–370
Kuhl CK (2018) Abbreviated breast MRI for screening women with dense breast: the EA1141 trial. Br J Radiol 91(1090):20170441
Bakker MF et al (2019) Supplemental MRI screening for women with extremely dense breast tissue. N Engl J Med 381(22):2091–2102
Kuhl CK, Keulers A, Strobel K et al (2018) Not all false positive diagnoses are equal: on the prognostic implications of false positive diagnoses made in breast MRI versus in mammography/digital tomosynthesis screening. Breast Cancer Res 20:13
Van Zee KJ, Barrio AV, Tchou J (2016) Society of Surgical Oncology Breast Disease Site Work Group. Treatment and long-term risks for patients with a diagnosis of ductal carcinoma in situ. JAMA Oncol 2:397–398
Dabbous FM, Dolecek TA, Berbaum ML et al (2017) Impact of a false-positive screening mammogram on subsequent screening behavior and stage at breast cancer diagnosis. Cancer Epidemiol Biomarkers Prev 26:397–403
Hardesty LA, Lind KE, Gutierrez EJ (2016) Compliance with screening mammography guidelines after a false-positive mammogram. J Am Coll Radiol 13:1032–1038
Román M, Castells X, Hofvind S, von Euler-Chelpin M (2016) Risk of breast cancer after false-positive results in mammographic screening. Cancer Med 5:1298–1306
Nelson HD, Pappas M, Cantor A, Griffin J, Daeges M, Humphrey L (2016) Harms of breast cancer screening: systematic review to update the 2009 U.S. preventive services task force recommendation. Ann Intern Med 164:256–267
Melnikow J, Fenton JJ, Whitlock EP, et al (2016) Supplemental screening for breast cancer in women with dense breasts: a systematic review for the U.S. Preventive Service Task Force. Report no. 14-05201-EF-3. Rockville, MD: Agency for Healthcare Research and Quality
Menezes GL, Knuttel FM, Stehouwer BL, Pijnappel RM, van den Bosch MA (2014) Magnetic resonance imaging in breast cancer: a literature review and future perspectives. World J Clin Oncol 5:61–70
Sardanelli F, Boetes C, Borisch B et al (2010) Magnetic resonance imaging of the breast: recommendations from the EUSOMA working group. Eur J Cancer 46:1296–1316
Baltzer PAT, Benndorf M, Dietzel M et al (2010) False-positive findings at contrast-enhanced breast MRI: a BI-RADS descriptor study. Am J Roentgenol 194(6):1658–1663
Myers KS, Kamel IR, Macura KJ (2015) MRI-guided breast biopsy: outcomes and effect on patient management. Clin Breast Cancer 15(2):143–152
ACR Practice Parameter for the Performance of Contrast Enhanced Magnetic Resonance Imaging (MRI) of the Breast. American College of Radiology. Revised 2018. https://www.acr.org/-/media/acr/files/practice-parameters/mr-contrast-breast.pdf. Assessed 07 Jan 2022
American College of Radiology (2013) BI-RADS: MRI: breast imaging reporting and data system: BI-RADS atlas. Reston: American College of Radiology
Sickles EA, D’Orsi CJ (2013) ACR BI-RADS follow-up and outcome monitoring. In: ACR BI-RADS Atlas, Breast Imaging Reporting and Data System. Reston: American College of Radiology
Mahoney MC (2008) Initial clinical experience with a new MRI vacuum-assisted breast biopsy device. J Magn Reson Imaging 28:900–9054
Orel SG, Rosen M, Mies C, Schnall MD (2006) MR imaging-guided 9-gauge vacuum-assisted core-needle breast biopsy: initial experience. Radiology 238:54–615
Lehman CD, Deperi ER, Peacock S, McDonough MD, Demartini WB, Shook J (2005) Clinical experience with MRI-guided vacuum-assisted breast biopsy. AJR 184:1782–1787
Liberman L, Morris EA, Dershaw DD, Abramson AF, Tan LK (2003) MR imaging of the ipsilateral breast in women with percutaneously proven breast cancer. AJR Am J Roentgenol 180(4):901–910
Perlet C, Heywang-Kobrunner SH, Heinig A et al (2006) Magnetic resonance-guided, vacuum-assisted breast biopsy: results from a European multicenter study of 538 lesions. Cancer 106:982–990
Han BK, Schnall MD, Orel SG, Rosen M (2008) Outcome of MRI-guided breast biopsy. AJR 191:1798–1804
Liberman L, Bracero N, Morris E, Thornton C, Dershaw DD (2005) MRI-guided 9-gauge vacuum-assisted breast biopsy: initial clinical experience. AJR 185:183–193
Malhaire C, El Khoury C, Thibault F et al (2010) Vacuum-assisted biopsies under MR guidance: results of 72 procedures. Eur Radiol 20:1554e–1562e
Rauch GM, Dogan BE, Smith TB, Liu P, Yang WT (2012) Outcome analysis of 9-gauge MRI-guided vacuum-assisted core needle breast biopsies. AJR Am J Roentgenol 198(2):292–299
Hoogerbrugge N, Kamm YJL, Bult P et al (2008) The impact of a false-positive MRI on the choice for mastectomy in BRCA mutation carriers. Ann Oncol 19:655–659
Sharma U, Jagannathan NR (2022) Magnetic resonance imaging (MRI) and MR spectroscopic methods in understanding breast cancer biology and metabolism. Metabolites 12:295. https://doi.org/10.3390/metabo12040295
Fardanesh R, Marino MA, Avendano D, Leithner D, Pinker K, Thakur SB (2019) Proton MR spectroscopy in the breast: technical innovations and clinical applications. J Magn Reson Imaging 50:1033–1046. https://doi.org/10.1002/jmri.26700
Codari M, Schiaffino S, Sardanelli F, Trimboli RM (2019) Artificial intelligence for breast MRI in 2008–2018: a systematic map** review. Am J Roentgenol 212:280–292. https://doi.org/10.2214/AJR.18.20389
Ye D-M, Wang H-T, Yu T (2020) The application of radiomics in breast MRI: a review. Technol Cancer Res Treat. https://doi.org/10.1177/1533033820916191
Reig B (2021) Radiomics and deep learning methods in expanding the use of screening breast MRI. Eur Radiol 31(8):5863–5865. https://doi.org/10.1007/s00330-021-08056-9
Jiang Y, Edwards AV, Newstead GM (2021) Artificial intelligence applied to breast MRI for improved diagnosis. Radiology 298(1):38–46. https://doi.org/10.1148/radiol.2020200292
Funding
The authors declare that no funds, grants, or other supports were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by AA, VF, and XL. The first draft of the manuscript was written by AA and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of University Health Network (May 14, 2020/REB#20-5119.0).
Consent to participate and consent to publish
As a retrospective study, the patient’s informed consent and also consent to publish was waived.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Alikhassi, A., Li, X., Au, F. et al. False-positive incidental lesions detected on contrast-enhanced breast MRI: clinical and imaging features. Breast Cancer Res Treat 198, 321–334 (2023). https://doi.org/10.1007/s10549-023-06861-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-023-06861-y