Abstract
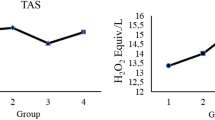
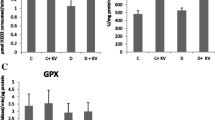
The liver damage caused by Diabetes Mellitus (DM) has attracted increasing attention in recent years. Liver injury in DM can be caused by ferroptosis, a form of cell death caused by iron overload. However, the role of iron transporters in this context is still not clear. Herein, we attempted to shed light on the pathophysiological mechanism of ferroptosis. DM was induced in 8-week-old male rats by streptozotocin (STZ) before assessment of the degree of liver injury. Together with histopathological changes, variations in glutathione peroxidase 4 (GPX4), glutathione (GSH), superoxide dismutase (SOD), transferrin receptor 1 (TFR1), ferritin heavy chain (FTH), ferritin light chain (FTL), ferroportin and Prussian blue staining, were monitored in rat livers before and after treatment with Fer-1. In the liver of STZ-treated rats, GSH and SOD levels decreased, whereas those of malondialdehyde (MDA) increased. Expression of TFR1, FTH and FTL increased whereas that of glutathione peroxidase 4 (GPX4) and ferroportin did not change significantly. Prussian blue staining showed that iron levels increased. Histopathology showed liver fibrosis and decreased glycogen content. Fer-1 treatment reduced iron and MDA levels but GSH and SOD levels were unchanged. Expression of FTH and FTL was reduced whereas that of ferroportin showed a mild decrease. Fer-1 treatment alleviated liver fibrosis, increased glycogen content and mildly improved liver function. Our study demonstrates that ferroptosis is involved in DM-induced liver injury. Regulating the levels of iron transporters may become a new therapeutic strategy in ferroptosis-induced liver injury.





Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article. The supplemental data can be obtained from the first author, K.W. (Wukp@mail2.sysu.edu.cn).
References
Abdul Y et al (2021) Deferoxamine treatment prevents post-stroke vasoregression and neurovascular unit remodeling leading to improved functional outcomes in type 2 male diabetic rats: Role of endothelial ferroptosis. Transl Stroke Res 12(4):615–630
Cao D, Wang C, Zhou L (2023) Identification and comprehensive analysis of ferroptosis-related genes as potential biomarkers for the diagnosis and treatment of proliferative diabetic retinopathy by bioinformatics methods. Exp Eye Res 232:109513
Cho NH et al (2018) IDF diabetes atlas: global estimates of diabetes prevalence for 2017 and projections for 2045. Diabetes Res Clin Pract 138:271–281
Dixon SJ et al (2012) Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell 149(5):1060–1072
El-Serag HB, Hampel H, Javadi F (2006) The association between diabetes and hepatocellular carcinoma: a systematic review of epidemiologic evidence. Clin Gastroenterol Hepatol 4(3):369–380
Fan X et al (2022) Downregulation of fatty acid binding protein 4 alleviates lipid peroxidation and oxidative stress in diabetic retinopathy by regulating peroxisome proliferator-activated receptor gamma-mediated ferroptosis. Bioengineered 13(4):10540–10551
Fujii J, Homma T, Osaki T (2022) Superoxide radicals in the execution of cell death. Antioxidants (Basel) 11(3):501
Gao H et al (2022) Aberrant iron distribution via hepatocyte-stellate cell axis drives liver lipogenesis and fibrosis. Cell Metab 34(8):1201-1213.e5
Govindarajan S et al (2021) Aloe vera carbohydrates regulate glucose metabolism through improved glycogen synthesis and downregulation of hepatic gluconeogenesis in diabetic rats. J Ethnopharmacol 281:114556
Graham RM et al (2007) Liver iron transport. World J Gastroenterol 13(35):4725–4736
Hansen JB, Moen IW, Mandrup-Poulsen T (2014) Iron: the hard player in diabetes pathophysiology. Acta Physiol (Oxf) 210(4):717–732
Harrison AV, Lorenzo FR, McClain DA (2023) Iron and the pathophysiology of diabetes. Annu Rev Physiol 85:339–362
Holstein A et al (2002) Clinical implications of hepatogenous diabetes in liver cirrhosis. J Gastroenterol Hepatol 17(6):677–681
Honarmand EK et al (2012) The catalytic center of ferritin regulates iron storage via Fe(II)-Fe(III) displacement. Nat Chem Biol 8(11):941–948
Jiang G, Zhang BB (2003) Glucagon and regulation of glucose metabolism. Am J Physiol Endocrinol Metab 284(4):E671–E678
** T, Chen C (2022) Umbelliferone delays the progression of diabetic nephropathy by inhibiting ferroptosis through activation of the Nrf-2/HO-1 pathway. Food Chem Toxicol 163:112892
Li S et al (2021) Inhibition of ferroptosis by up-regulating Nrf2 delayed the progression of diabetic nephropathy. Free Radic Biol Med 162:435–449
Liu C et al (2022a) Glia maturation factor-beta induces ferroptosis by impairing chaperone-mediated autophagic degradation of ACSL4 in early diabetic retinopathy. Redox Biol 52:102292
Liu SH, Ku CY, Chiang MT (2022) Polysaccharide-rich red algae (Gelidium amansii) hot-water extracts alleviate abnormal hepatic lipid metabolism without suppression of glucose intolerance in a streptozotocin/nicotinamide-induced diabetic rat model. Molecules 27(4):1447
Loomba R et al (2012) Association between diabetes, family history of diabetes, and risk of nonalcoholic steatohepatitis and fibrosis. Hepatology 56(3):943–951
Mehta KJ, Farnaud SJ, Sharp PA (2019) Iron and liver fibrosis: Mechanistic and clinical aspects. World J Gastroenterol 25(5):521–538
Miotto G et al (2020) Insight into the mechanism of ferroptosis inhibition by ferrostatin-1. Redox Biol 28:101328
Pietrangelo A (2016) Iron and the liver. Liver Int 36(Suppl 1):116–123
Rochette L, Dogon G, Rigal E, Zeller M, Cottin Y, Vergely C (2022) Lipid peroxidation and iron metabolism: two corner stones in the homeostasis control of ferroptosis. Int J Mol Sci 24(1):449
Song JX et al (2022) Liraglutide attenuates hepatic iron levels and ferroptosis in db/db mice. Bioengineered 13(4):8334–8348
Stancic A, Velickovic K, Markelic M et al (2022) Involvement of ferroptosis in diabetes-induced liver pathology. Int J Mol Sci 23(16):9309
Sung KC, Wild SH, Byrne CD (2013) Resolution of fatty liver and risk of incident diabetes. J Clin Endocrinol Metab 98(9):3637–3643
Tang W et al (2022) Caveolin-1 alleviates diabetes-associated cognitive dysfunction through modulating neuronal ferroptosis-mediated mitochondrial homeostasis. Antioxid Redox Signal 37(13–15):867–886
Tian H et al (2021) Activation of NRF2/FPN1 pathway attenuates myocardial ischemia-reperfusion injury in diabetic rats by regulating iron homeostasis and ferroptosis. Cell Stress Chaperones 27(2):149–164
Tomic D, Shaw JE, Magliano DJ (2022) The burden and risks of emerging complications of diabetes mellitus. Nat Rev Endocrinol 18(9):525–539
Ursini F, Maiorino M (2020) Lipid peroxidation and ferroptosis: the role of GSH and GPx4. Free Radic Biol Med 152:175–185
Wang C et al (2012) Increased risk of hepatocellular carcinoma in patients with diabetes mellitus: a systematic review and meta-analysis of cohort studies. Int J Cancer 130(7):1639–1648
Wang CY, Babitt JL (2019) Liver iron sensing and body iron homeostasis. Blood 133(1):18–29
Wang Y et al (2020) Ferroptosis involves in renal tubular cell death in diabetic nephropathy. Eur J Pharmacol 888:173574
Wu K, Fei L, Wang X et al (2022) ZIP14 is involved in iron deposition and triggers ferroptosis in diabetic nephropathy. Metallomics 14(7):mfac034
Younossi ZM et al (2019) The global epidemiology of NAFLD and NASH in patients with type 2 diabetes: a systematic review and meta-analysis. J Hepatol 71(4):793–801
Zhang W, Lu J, Wang Y et al (2023) Canagliflozin attenuates lipotoxicity in cardiomyocytes by inhibiting inflammation and ferroptosis through activating AMPK pathway. Int J Mol Sci 24(1):858
Acknowledgements
Many thanks to all the authors for their contributions. We would like to thank Sun Yat-sen University for providing animal laboratory and experimental equipment.
Funding
The research was financially supported by the GuangDong Basic and Applied Basic Research Foundation of China (2023A1515111043).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Conceptualization: K.W., J.C., and Y.Z.; methodology: K.W.,J.L.,X.X.,E.Z.L.J.,XH.Y.; writing—original draft preparation:K.W.,M.H.; writing—review and editing: K.W., Y.Z.,and M.H.. All authors read and approved the final manuscript.