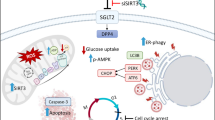
Abstract
The isocitrate dehydrogenase (IDH), which participates in the TCA cycle, is an important key enzyme in regulating cell metabolism. The effect of the metabolic IDH enzyme on cancer pathogenesis has recently been shown in different types of cancer. However, the role of wild-type (wt) IDH1 in the development of colon cancer is still unknown. Our study investigated the role of the IDH1 enzyme in key hallmarks of colon cancer using various methods such as wound healing, cell cycle, colony formation ability, invasion, and apoptosis analysis. Furthermore, cell metabolism was investigated by pyruvate analysis, dinitrosalicylic acid, and HPLC methods. In addition, CRISPR/Cas9 tool was utilized to knockout the IDH1 gene in colon adenocarcinoma cells (SW620). Further studies were performed in two isogenic IDH1 KO clones. Our findings in both clones suggest that IDH1 KO results in G0/G1 arrest, and reduces proliferation by approximately twofold compared to IDH1 WT cells. In addition, the invasion, migration, and colony formation abilities of IDH1 KO clones were significantly decreased accompanied by significant morphological changes. In the context of metabolism, intracellular glucose, pyruvate, αKG, and malate levels were decreased, while the intracellular citrate level was increased in IDH1 KO clones as compared to IDH1 WT cells. Our results reveal that wt IDH1 knockout leads to a decrease in the aggressive features of colon cancer cells. In conclusion, we reported that wt IDH1 has an effective role in colon cancer progression and could be a potential therapeutic target.







Similar content being viewed by others
Data Availability
All the generated or analyzed datas during the current study are included in this manuscript. The datas in the present study are available from the corresponding author on reasonable request.
Abbreviations
- IDH:
-
Isocitrate dehydrogenase
- wt:
-
Wild-type
- FH:
-
Fumarate hydratase
- SDH:
-
Succinate dehydrogenase
- αKG:
-
α-Ketoglutarate
- MeOH:
-
Methanol
- DNS:
-
Dinitrosalicylic acid
- DNPH:
-
2,4-Dinitrophenylhydrazine
- TBS:
-
Tris-buffered saline
- RT-qPCR:
-
Reverse transcriptase quantitative polymerase chain reaction
References
Alavi M, Rai M (2021) Antisense RNA, the modified CRISPR-Cas9, and metal/metal oxide nanoparticles to inactivate pathogenic bacteria. CMBR 1:52–59
Atalay EB, Kayali HA (2022) The elevated D-2-hydroxyglutarate level found as a characteristic metabolic change of colon cancer in both in vitro and in vivo models. Biochem Biophys Res Commun 627:191–199. https://doi.org/10.1016/j.bbrc.2022.08.019
Ayar Kayali H, Tarhan L (2006) Vancomycin antibiotic production and TCA-glyoxalate pathways depending on the glucose concentration in Amycolatopsis orientalis. Enzyme Microb Technol 38:727–734
Barnes LD, Kuehn GD, Atkinson DE (1971) Yeast diphosphopyridine nucleotide specific isocitrate dehydrogenase purification and some properties. Biochem 10:3939–3944
Biedermann J, Preussler M, Conde M, Peitzsch M, Richter S, Wiedemuth R (2019) Mutant IDH1 differently affects redox state and metabolism in glial cells of normal and tumor origin. Cancers 11:2028
Bin H, Mei H, Hui W, Bing Z (2022) The correlation between miR -34a-3p, miR -31, PLEK2 and the occurrence, development and prognosis of colorectal cancer. Cell Mol Biol (noisy-Le-Grand) 68:192–200
Brabetz O, Alla V, Angenendt L, Schliemann C, Berdel WE, Arteaga MF, Mikesch JH (2017) RNA-guided CRISPR-Cas9 system-mediated engineering of acute myeloid leukemia mutations. Mol Ther Nucleic Acids 6:243–248
Cardaci S, Ciriolo MR (2012) TCA cycle defects and cancer: when metabolism tunes redox state. Int J Cell Biol. https://doi.org/10.1155/2012/161837
Crooks DR, Maio N, Lang M, Ricketts CJ, Vocke CD, Gurram S, Turan S, Kim YY, Cawthon GM, Sohelian F, Val N, Pfeiffer RM, Jailwala P, Tandon M, Tran B, Fan TMW, Lane AN, Ried T, Wangsa D, Malayeri AA, Merino MJ, Yang Y, Meier JL, Ball MW, Rouault TA, Srinivasan R, Linehan WM (2021) Mitochondrial DNA alterations underlie an irreversible shift to aerobic glycolysis in fumarate hydratase–deficient renal cancer. Sci Signal 14:eabc4436
Dalziel K (1980) Isocitrate dehydrogenase and related oxidative decarboxylases. FEBS Lett 117:45–55
Dang L, White DW, Gross S, Bennett BD, Bittinger MA, Driggers EM, Fantin VR, Jang HG, ** S, Keenan MC, Marks KM, Prins RM, Ward PS, Yen KE, Liau LM, Rabinowitz JD, Cantley LC, Thompson CB, Vander Heiden MG, Su SM (2009) Cancer-associated IDH1 mutations produce 2-hydroxyglutarate. Nature 462:739–744
Franken NA, Rodermond HM, Stap J, Haveman J, Van Bree C (2006) Clonogenic assay of cells in vitro. Nat Protoc 1:2315–2319
Hanahan D (2022) Hallmarks of cancer: new dimensions. Cancer Discov 12:31–46
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144:646–674
Hao HX, Khalimonchuk O, Schraders M, Dephoure N, Bayley JP, Kunst H, Devilee P, Cremers CWRJ, Schiffman JD, Bentz BG, Gygi SP, Winge DR, Kremer H, Rutter J (2009) SDH5, a gene required for flavination of succinate dehydrogenase, is mutated in paraganglioma. Sci 325:1139–1142
Hao J, Wei H, Qi Y, Liu H (2022) miR-129-5p plays an anticancer role in colon cancer by targeting RSF1. Cell Mol Biol 67:196–201
**ek M, Chylinski K, Fonfara I, Hauer M, Doudna JA, Charpentier A (2012) A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Sci 337:816–821
Khuwijitjaru P, Koomyart I, Kobayashi T, Adachi S (2017) Hydrolysis of konjac flour under subcritical water conditions. Chiang Mai J Sci 44:988–992
Krebs H, Johnson WA (1937) The role of citric acid in intermediate metabolism in animal tissues. Enzymologia 4:148–156
Li L, **aoyu H, Trent JC (2019) Depletion of mutant IDH1 impairs chondrosarcoma growth by downregulating integrins. J Clin Oncol 37:11031–11031
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2− ΔΔCT method. Methods 25:402–408
Moure CJ, Diplas BH, Chen LH, Yang R, Pirozzi CJ, Wang Z, Spasojevic I, Waitkus MS, He Y, Yan H (2019) CRISPR editing of mutant IDH1 R132H induces a CpG methylation-low state in patient-derived glioma models of G-CIMP. Mol Cancer Res 17:2042–2050
Neitzel C, Demuth P, Wittmann S, Fahrer J (2020) Targeting altered energy metabolism in colorectal cancer: oncogenic reprogramming, the central role of the TCA cycle and therapeutic opportunities. Cancers 12:1731
Pollard PJ, Ratcliffe PJ (2009) Puzzling Patterns of Predisposition. Sci 324:192–194
Qiao S, Lu W, Glorieux C, Li J, Zeng P, Meng N (2021) Wild-type IDH2 protects nuclear DNA from oxidative damage and is a potential therapeutic target in colorectal cancer. Oncogene 40:5880–5892
Ran FA, Hsu PD, Wright J, Agarwala V, Scott DA, Zhang F (2013) Genome engineering using the CRISPR-Cas9 system. Nat Protoc 8:2281–2308
Shi J, Fan L, Li B, Pan H (2022) Molecular Mechanism of Integrin αvβ6 in Liver Metastasis of Colon Cancer Based on SDF-1/CXCR4. Cell Mol Biol 67:88–95
Sjöblom T, Jones S, Wood LD, Parsons DW, Lin J, Barber TD (2006) The consensus coding sequences of human breast and colorectal cancers. Sci 314:268–274
Špačková J, Gotvaldová K, Dvořák A, Urbančoková A, Pospíšilová K, Větvička D, Leguina-Ruzzi A, Tesařová P, Vítek L, Ježek P, Smolková K (2021) Biochemical background in mitochondria affects 2HG production by IDH2 and ADHFE1 in breast carcinoma. Cancers 13:1709
Su L, Zhang X, Zheng L, Wang M, Zhu Z, Li P (2020) Mutation of isocitrate dehydrogenase 1 in cholangiocarcinoma impairs tumor progression by inhibiting isocitrate metabolism. Front Endocrinol 11:189
Subasi E, Atalay EB, Erdogan D, Sen B, Pakyapan B, Ayar Kayalı H (2020) Synthesis and characterization of thiosemicarbazone-functionalized organoruthenium (II)-arene complexes: Investigation of antitumor characteristics in colorectal cancer cell lines. Mater Sci Eng C 106:110152
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71:209–249
Wang B, Ye Y, Yang X, Liu B, Wang Z, Chen S (2020) SIRT 2-dependent IDH 1 deacetylation inhibits colorectal cancer and liver metastases. EMBO Rep 21:e48183
Wen L, Zhang Y, Yang B, Han F, Ebadi AG, Toughani M (2020) Knockdown of Angiopoietin-like protein 4 suppresses the development of colorectal cancer. Cell Mol Biol 66:117–124
Wu SS, Li QC, Yin CQ, Xue W, Song CQ (2020) Advances in CRISPR/Cas-based gene therapy in human genetic diseases. Theranostics 10:4374
** Y, Xu P (2021) Global colorectal cancer burden in 2020 and projections to 2040. Transl Oncol 14:101174
Yang H, **e S, Liang B, Tang Q, Liu H, Wang D, Huang G (2021) Exosomal IDH1 increases the resistance of colorectal cancer cells to 5-Fluorouracil. J Cancer 12:4862
Yoo KS, Lee EJ, Patil BS (2011) Underestimation of pyruvic acid concentrations by fructose and cysteine in 2, 4-dinitrophenylhydrazine-mediated onion pungency test. J Food Sci 76:C1136–C1142
Acknowledgements
We gratefully thank doctoral student Ece Çakıroğlu for her contribution to the CRISPR/Cas9 method.
Funding
The authors declare that there were not any funds, grants, or other support during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
EBA performed the experiments and analyzed the data and writing- the original draft; SS analyzed the data in the CRISPR/Cas9 method and Writing—review & editing; HAK coordinated all aspects of this work, Writing—review & editing, Supervision HAK contributed to the study conception and design.
Corresponding author
Ethics declarations
Conflict of ınterest
There have no relevant financial or non-financial interests by all the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Atalay, E.B., Senturk, S. & Kayali, H.A. Wild-type IDH1 Knockout Leads to G0/G1 Arrest, Impairs Cancer Cell Proliferation, Altering Glycolysis, and the TCA Cycle in Colon Cancer. Biochem Genet 61, 1470–1486 (2023). https://doi.org/10.1007/s10528-022-10325-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10528-022-10325-1