Abstract
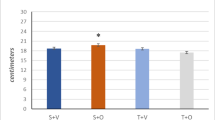
Dietary restriction (DR) is a potential intervention for ameliorating ageing-related damages. Mitochondrial quality control is the key mechanism for regulating cellular functions in skeletal muscle. This study aimed to explore the effect of age and DR on the homeostasis of mitochondrial quality control in skeletal muscle. To study the effect of age on mitochondrial homeostasis, young (3 months old) male C57BL/6J mice were fed ad libitum (AL) until 7 (Young), 14 (Middle), and 19 months (Aged) of age. For the DR intervention, 60% of AL intake was given to the mice at 3 months of age until they reached 19 months of age (16 months). The quadriceps femoris muscle was collected for further analysis. Significant changes in the skeletal muscle were noticed during the transition between middle age and the elderly stages. An accumulation of collagen was observed in the muscle after middle age. Compared with the Middle muscle, Aged muscle displayed a greater expression of VDAC, and lower expressions of mitochondrial dynamic proteins and OXPHOS proteins. The DR intervention attenuated collagen content and elongated the sarcomere length in the skeletal muscle during ageing. In addition, DR adjusted the abnormalities in mitochondrial morphology in the Aged muscle. DR downregulated VDAC expression, but upregulated OPA1 and DRP1 expressions. Taken together, greater pathological changes were noticed in the skeletal muscle during ageing, especially in the transition between middle age and the elderly, whereas early-onset DR attenuated the muscular ageing via normalising partial functions of mitochondria.





Similar content being viewed by others
Data availability
Data are included as electronic supplementary materials.
Abbreviations
- AL:
-
Ad libitum
- DR:
-
Dietary restriction
- DRP1:
-
Dynamin-related protein 1
- FIS1:
-
Mitochondrial fission 1 protein
- GAPDH:
-
Glyceraldehyde 3-phosphate dehydrogenase
- MDA:
-
Malondialdehyde
- MFN2:
-
Mitofusin-2
- MQC:
-
Mitochondrial quality control
- OPA1:
-
Optic Atrophy 1
- OXPHOS:
-
Oxidative phosphorylation
- PGC-1α:
-
PPARγ coactivator 1 α
- PPARγ:
-
Peroxisome proliferator- activated receptor gamma
- ROS:
-
Reactive oxygen species
- TEM:
-
Transmission electron microscopy
- VDAC:
-
Voltage-dependent anion channel
References
Bellanti F, Lo Buglio A, Vendemiale G (2021) Mitochondrial impairment in sarcopenia. BiologyBasel. https://doi.org/10.3390/biology10010031
Boengler K, Kosiol M, Mayr M, Schulz R, Rohrbach S (2017) Mitochondria and ageing: role in heart, skeletal muscle and adipose tissue. J Cachexia Sarcopeni 8:349–369. https://doi.org/10.1002/jcsm.12178
Chen D, Steele AD, Lindquist S, Guarente L (2005) Increase in activity during calorie restriction requires Sirt1. Science 310:1641–1641. https://doi.org/10.1126/science.1118357
Chen CN, Lin SY, Liao YH, Li ZJ, Wong AM (2015) Late-onset caloric restriction alters skeletal muscle metabolism by modulating pyruvate metabolism. Am J Physiol Endocrinol Metab 308:E942-949. https://doi.org/10.1152/ajpendo.00508.2014
Chen CN, Liao YH, Tsai SC, Thompson LV (2019) Age-dependent effects of caloric restriction on mTOR and ubiquitin-proteasome pathways in skeletal muscles. Geroscience 41:871–880. https://doi.org/10.1007/s11357-019-00109-8
Chen Q, Huang WS, Capanoglu E, Amrouche AT, Lu BY (2023) Targeting mitochondrial quality control in muscle aging: natural dietary products as potential interventions. Food Front 4:1206–1241. https://doi.org/10.1002/fft2.261
Chiang CH, Li SJ, Zhang TR, Chen CY (2022) Long-term dietary restriction ameliorates ageing-related renal fibrosis in male mice by normalizing mitochondrial functions and autophagy. Biogerontology 23:731–740. https://doi.org/10.1007/s10522-022-09993-8
Chiang CH, Li SJ, Lin YH, Wang PY, Hsu PS, Lin SP et al (2023) Early-onset caloric restriction alleviates ageing-associated steatohepatitis in male mice via restoring mitochondrial homeostasis. Biogerontology 24:391–401. https://doi.org/10.1007/s10522-023-10023-4
Colleluori G, Villareal DT (2021) Aging, obesity, sarcopenia and the effect of diet and exercise intervention. Exp Gerontol 155:111561. https://doi.org/10.1016/j.exger.2021.111561
Corbianco S, Dini M, Bongioanni P, Carboncini MC, Cavallini G (2020) Exercise training in ad libitum and food-restricted old rats: effects on metabolic and physiological parameters. Biogerontology 21:69–82. https://doi.org/10.1007/s10522-019-09844-z
Faitg J, Leduc-Gaudet JP, Reynaud O, Ferland G, Gaudreau P, Gouspillou G (2019) Effects of aging and caloric restriction on fiber type composition, mitochondrial morphology and dynamics in rat oxidative and glycolytic muscles. Front Physiol 10:420. https://doi.org/10.3389/fphys.2019.00420
Gallardo CM, Hsu CT, Gunapala KM, Parfyonov M, Chang CH, Mistlberger RE et al (2014) Behavioral and neural correlates of acute and scheduled hunger in C57BL/6 mice. PLoS ONE 9:e95990. https://doi.org/10.1371/journal.pone.0095990
Green CL, Lamming DW, Fontana L (2022) Molecular mechanisms of dietary restriction promoting health and longevity. Nat Rev Mol Cell Biol 23:56–73. https://doi.org/10.1038/s41580-021-00411-4
Gutierrez-Casado E, Khraiwesh H, Lopez-Dominguez JA, Montero-Guisado J, Lopez-Lluch G, Navas P et al (2019) The impact of aging, calorie restriction and dietary fat on autophagy markers and mitochondrial ultrastructure and dynamics in mouse skeletal muscle. J Gerontol A Biol Sci Med Sci 74:760–769. https://doi.org/10.1093/gerona/gly161
Hagen JL, Krause DJ, Baker DJ, Fu MH, Tarnopolsky MA, Hepple RT (2004) Skeletal muscle aging in F344BN F1-hybrid rats: I. Mitochondrial dysfunction contributes to the age-associated reduction in VO2max. J Gerontol A Biol Sci Med Sci 59:1099–1110. https://doi.org/10.1093/gerona/59.11.1099
Ham DJ, Borsch A, Chojnowska K, Lin S, Leuchtmann AB, Ham AS et al (2022) Distinct and additive effects of calorie restriction and rapamycin in aging skeletal muscle. Nat Commun 13:2025. https://doi.org/10.1038/s41467-022-29714-6
Joseph AM, Adhihetty PJ, Buford TW, Wohlgemuth SE, Lees HA, Nguyen LM et al (2012) The impact of aging on mitochondrial function and biogenesis pathways in skeletal muscle of sedentary high- and low-functioning elderly individuals. Aging Cell 11:801–809. https://doi.org/10.1111/j.1474-9726.2012.00844.x
Leduc-Gaudet JP, Picard M, Pelletier FS, Sgarioto N, Auger MJ, Vallée J et al (2015) Mitochondrial morphology is altered in atrophied skeletal muscle of aged mice. Oncotarget 6:17923–17937. https://doi.org/10.18632/oncotarget.4235
Li XD, Rebrin I, Forster MJ, Sohal RS (2012) Effects of age and caloric restriction on mitochondrial protein oxidative damage in mice. Mech Ageing Dev 133:30–36. https://doi.org/10.1016/j.mad.2011.12.001
Li H, Kumar Sharma L, Li Y, Hu P, Idowu A, Liu D et al (2013) Comparative bioenergetic study of neuronal and muscle mitochondria during aging. Free Radic Biol Med 63:30–40. https://doi.org/10.1016/j.freeradbiomed.2013.04.030
Li SJ, Lin YH, Chiang CH, Wang PY, Chen CY (2022) Early-onset dietary restriction maintains mitochondrial health, autophagy and ER function in the left ventricle during aging. J Nutr Biochem 101:108944. https://doi.org/10.1016/j.jnutbio.2022.108944
Liu SZ, Marcinek DJ (2017) Skeletal muscle bioenergetics in aging and heart failure. Heart Fail Rev 22:167–178. https://doi.org/10.1007/s10741-016-9586-z
Lopez-Dominguez JA, Khraiwesh H, Gonzalez-Reyes JA, Lopez-Lluch G, Navas P, Ramsey JJ et al (2013) Dietary fat modifies mitochondrial and plasma membrane apoptotic signaling in skeletal muscle of calorie-restricted mice. Age 35:2027–2044. https://doi.org/10.1007/s11357-012-9492-9
McKiernan SH, Colman RJ, Lopez M, Beasley TM, Aiken JM, Anderson RM et al (2011) Caloric restriction delays aging-induced cellular phenotypes in rhesus monkey skeletal muscle. Exp Gerontol 46:23–29. https://doi.org/10.1016/j.exger.2010.09.011
Miljkovic N, Lim JY, Miljkovic I, Frontera WR (2015) Aging of skeletal muscle fibers. Ann Rehabil Med 39:155–162. https://doi.org/10.5535/arm.2015.39.2.155
Naruse M, Trappe S, Trappe TA (2023) Human skeletal muscle-specific atrophy with aging: a comprehensive review. J Appl Physiol 134:900–914. https://doi.org/10.1152/japplphysiol.00768.2022
Rana A, Oliveira MP, Khamoui AV, Aparicio R, Rera M, Rossiter HB et al (2017) Promoting Drp1-mediated mitochondrial fission in midlife prolongs healthy lifespan of Drosophila melanogaster. Nat Commun 8:448. https://doi.org/10.1038/s41467-017-00525-4
Ringholm S, Gudiksen A, Halling JF, Qoqaj A, Rasmussen PM, Prats C et al (2023) Impact of aging and lifelong exercise training on mitochondrial function and network connectivity in human skeletal muscle. J Gerontol a-Biol 78:373–383. https://doi.org/10.1093/gerona/glac164
Sayed RKA, de Leonardis EC, Guerrero-Martínez JA, Rahim I, Mokhtar DM, Saleh AM et al (2016) Identification of morphological markers of sarcopenia at early stage of aging in skeletal muscle of mice. Exp Gerontol 83:22–30. https://doi.org/10.1016/j.exger.2016.07.007
Sciorati C, Rigamonti E, Manfredi AA, Rovere-Querini P (2016) Cell death, clearance and immunity in the skeletal muscle. Cell Death Differ 23:927–937. https://doi.org/10.1038/cdd.2015.171
Serna JDC, Caldeira da Silva CC, Kowaltowski AJ (2020) Functional changes induced by caloric restriction in cardiac and skeletal muscle mitochondria. J Bioenerg Biomembr 52:269–277. https://doi.org/10.1007/s10863-020-09838-4
Sharples AP, Hughes DC, Deane CS, Saini A, Selman C, Stewart CE (2015) Longevity and skeletal muscle mass: the role of IGF signalling, the sirtuins, dietary restriction and protein intake. Aging Cell 14:511–523. https://doi.org/10.1111/acel.12342
Su Y, Claflin DR, Huang MX, Davis CS, Macpherson PCD, Richardson A et al (2021) Deletion of neuronal CuZnSOD accelerates age-associated muscle mitochondria and calcium handling dysfunction that is independent of denervation and precedes sarcopenia. Int J Mol Sci 22:10735. https://doi.org/10.3390/ijms221910735
Tian Q, Mitchell BA, Zampino M, Fishbein KW, Spencer RG, Ferrucci L (2022) Muscle mitochondrial energetics predicts mobility decline in well-functioning older adults: the Baltimore longitudinal study of aging. Aging Cell 21:e13552. https://doi.org/10.1111/acel.13552
Triolo M, Bhattacharya D, Hood DA (2022) Denervation induces mitochondrial decline and exacerbates lysosome dysfunction in middle-aged mice. Aging 14:8900–8913. https://doi.org/10.18632/aging.204365
van Norren K, Rusli F, van Dijk M, Lute C, Nagel J, Dijk FJ et al (2015) Behavioural changes are a major contributing factor in the reduction of sarcopenia in caloric-restricted ageing mice. J Cachexia Sarcopeni 6:253–268. https://doi.org/10.1002/jcsm.12024
WHO (2023). World Heatlh Statistics. World Health Organization
**e WQ, **ao WF, Tang K, Wu YX, Hu PW, Li YS et al (2020) Caloric restriction: implications for sarcopenia and potential mechanisms. Aging 12:24441–24452. https://doi.org/10.18632/aging.103987
Acknowledgements
All authors read and approved the final manuscript. The authors declare that there are no conflicts of interest. The authors are grateful to Dr. Harry J. Mersmann for reviewing the manuscript and English proofreading.
Funding
This work was supported by the Research Grant MOST 110-2320-B-002-039-MY3 (from the Ministry of Science and Technology, Taiwan).
Author information
Authors and Affiliations
Contributions
TR Zhang obtained, analysed, and interpreted the data, and drafted the article. CH Chiang performed the acquisition, analysis, and interpretation of the data. TC Hsu and CY Wang performed the data acquisition. CY Chen developed the conception and design of the study and obtained the funding. All authors approved the final version of the article, including the authorship list.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, TR., Chiang, CH., Hsu, TC. et al. Age and dietary restriction modulate mitochondrial quality in quadriceps femoris muscle of male mice. Biogerontology 25, 447–459 (2024). https://doi.org/10.1007/s10522-023-10086-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10522-023-10086-3