Abstract
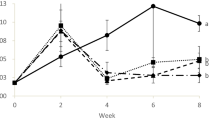
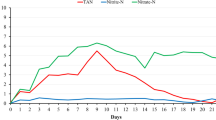
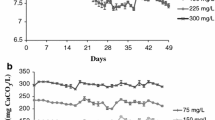
Strategies, such as the biological denitrification process, must be adopted to treat rich-nitrate water from biofloc systems, making it suitable for culture or proper disposal. This study evaluated the effect of water reuse from a BFT system after being subjected to a biological anaerobic denitrification process on shrimp performance and water quality in the rearing of Litopenaeus vannamei. A 63-day experiment was carried out at the Marine Station of Aquaculture of the Federal University of Rio Grande. L. vannamei juveniles (1.30 ± 0.48 g) were stocked in 150-L tanks at a density of 500 shrimp/m3. Four treatments (with three replicas each) were tested: natural seawater and denitrified seawater, both with and without biofloc inoculum. Temperature, salinity, dissolved oxygen, pH, ammonia, nitrite, nitrate, alkalinity, and total suspended solids (TSS) in the water were monitored. Ammonia and nitrite levels were higher without biofloc inoculum, while nitrate levels increased with inoculum use. Alkalinity and TSS were significantly higher in denitrified water with inoculum, driven mainly by the initial higher concentrations. A 100% mortality was found in the treatment using denitrified water without inoculum, probably due to the presence of byproducts after the denitrification process, but no differences in survival (>89%) were found in the other treatments. The biofloc inoculum helped with higher productivity (3.03 ± 0.08 kg/m3) and lower FCR (1.48 ± 0.05) in the treatment using seawater compared to the treatment in which no inoculum was used (2.62 ± 0.18 kg/m3 and 1.79 ± 0.16), in addition to serving as a biological treatment in denitrified seawater, readjusting the water for shrimp farming. As the growth performance of shrimp raised in denitrified water showed no differences compared to natural seawater, utilizing denitrified seawater from a biofloc system seems feasible for shrimp cultivation. However, alternatives for water treatment after the denitrification process need to be investigated.


Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Ahmad A, Sheikh Abdullah SR, Hasan HA, Othman AR, Izzati IN (2021) Aquaculture industry: supply and demand, best practices, effluent and its current issues and treatment technology. J Environ Manage 287(March):112271. https://doi.org/10.1016/j.jenvman.2021.112271
Aich N, Nama S, Biswal A, Paul T (2021) A review on recirculating aquaculture systems: challenges and opportunities for sustainable aquaculture. Innov Farm 5(1):17–24
Albina P, Durban N, Bertron A, Albrecht A, Robinet J-C, Erable B (2019) Influence of hydrogen electron donor, alkaline pH, and high nitrate concentrations on microbial denitrification: a review. Int J Mol Sci 20(20):5163. https://doi.org/10.3390/ijms20205163
Alves Neto I, Brandão H, Furtado PS, Wasielesky W Jr (2019) Acute toxicity of nitrate in Litopenaeus vannamei juveniles at low salinity levels. Ciência Rural 49(1). https://doi.org/10.1590/0103-8478cr20180439
Aminot A, Chaussepied M (1983) Manuel des analyses chimiques en milieu marin. Brest: CNEXO, Paris
AOAC (1990) Official methods of analysis of the association of official analytical chemists, 15th edn. Association of Official Analytical Chemists
APHA-AWWA-WEF (2005) Standard methods for the examination of water and wastewater, 20th edn. American Public Health Association, American Water Works Association, Water Environment Federation, Washington, DC
Ballester ELC, Abreu PC, Cavalli RO, Emerenciano M, de Abreu L, Wasielesky W (2010) Effect of practical diets with different protein levels on the performance of Farfantepenaeus paulensis juveniles nursed in a zero exchange suspended microbial flocs intensive system. Aquac Nutr 16(2):163–172. https://doi.org/10.1111/j.1365-2095.2009.00648.x
Biesterfeld S, Farmer G, Russell P, Figueroa L (2001) Effect of alkalinity type and concentration on nitrifying biofilm activity. Proc Water Environ Fed 16:277–291. https://doi.org/10.2175/193864701790901951
Brandão H, Xavier ÍV, Santana GKK, Santana HJK, Krummenauer D, Wasielesky W (2021) Heterotrophic versus mixed BFT system: impacts on water use, suspended solids production and growth performance of Litopenaeus vannamei. Aquac Eng 95:102194. https://doi.org/10.1016/j.aquaeng.2021.102194
Brol J, Müller L, Prates ECA, de Farias BS, Pedrosa VF, de Almeida Pinto LA, Sant’anna Cadaval TR, Tesser MB, Wasielesky W, Ventura-Lima J (2021) Dietary chitosan supplementation in Litopenaeus vannamei reared in a biofloc system: effect on antioxidant status facing saline stress. Aquaculture 544(May):737034. https://doi.org/10.1016/j.aquaculture.2021.737034
da Costa JB, Rodgher S, Daniel LA, Espíndola ELG (2014) Toxicity on aquatic organisms exposed to secondary effluent disinfected with chlorine, peracetic acid, ozone and UV radiation. Ecotoxicology 23(9):1803–1813. https://doi.org/10.1007/s10646-014-1346-z
da SLGP, Krummenauer D, Poersch LH, Rosas VT, Wasielesky W (2020) Hyperintensive stocking densities for Litopenaeus vannamei grow-out in biofloc technology culture system. J World Aquac Soc 51(6):1290–1300. https://doi.org/10.1111/jwas.12718
Domínguez Henao L, Turolla A, Antonelli M (2018) Disinfection by-products formation and ecotoxicological effects of effluents treated with peracetic acid: a review. Chemosphere 213:25–40. https://doi.org/10.1016/j.chemosphere.2018.09.005
Ebeling JM, Timmons MB, Bisogni JJ (2006) Engineering analysis of the stoichiometry of photoautotrophic, autotrophic, and heterotrophic removal of ammonia–nitrogen in aquaculture systems. Aquaculture 257(1–4):346–358. https://doi.org/10.1016/j.aquaculture.2006.03.019
FAO (2022) The State of World Fisheries and Aquaculture 2022. FAO, Rome, Italy
Furtado PS, Campos BR, Serra FP, Klosterhoff M, Romano LA, Wasielesky W (2015a) Effects of nitrate toxicity in the Pacific white shrimp, Litopenaeus vannamei, reared with biofloc technology (BFT). Aquac Int 23(1):315–327. https://doi.org/10.1007/s10499-014-9817-z
Furtado PS, Fugimura MMS, Monserrat JM, Souza DM, Garcia LDO, Wasielesky W (2015b) Acute effects of extreme pH and its influences on the survival and biochemical biomarkers of juvenile White Shrimp, Litopenaeus vannamei. Mar Freshw Behav Physiol 48(6):417–429. https://doi.org/10.1080/10236244.2015.1086539
Furtado PS, Gaona CAP, Poersch LH, Wasielesky W (2014) Application of different doses of calcium hydroxide in the farming shrimp Litopenaeus vannamei with the biofloc technology (BFT). Aquac Int 22(3):1009–1023. https://doi.org/10.1007/s10499-013-9723-9
Gaona CAP, Poersch LH, Krummenauer D, Foes GK, Wasielesky WJ (2011) The effect of solids removal on water quality, growth and survival of Litopenaeus vannamei in a biofloc technology culture system. Int J Recirc Aquac 12(1):54–73. https://doi.org/10.21061/ijra.v12i1.1354
Garza de Yta A, Rouse DB, Davis DA (2004) Influence of nursery period on the growth and survival of Litopenaeus vannamei under pond production conditions. J World Aquac Soc 35(3):357–365. https://doi.org/10.1111/j.1749-7345.2004.tb00099.x
Hostins B, Wasielesky W, Decamp O, Bossier P, De Schryver P (2019) Managing input C/N ratio to reduce the risk of acute hepatopancreatic necrosis disease (AHPND) outbreaks in biofloc systems – a laboratory study. Aquaculture 508(2018):60–65. https://doi.org/10.1016/j.aquaculture.2019.04.055
Jarusutthirak C, Amy G (2006) Role of soluble microbial products (SMP) in membrane fouling and flux decline. Environ Sci Technol 40(3):969–974. https://doi.org/10.1021/es050987a
Jiang L, Pan L, Fang-Bo (2005) Effect of dissolved oxygen on immune parameters of the white shrimp Litopenaeus vannamei. Fish Shellfish Immunol 18(2):185–188. https://doi.org/10.1016/j.fsi.2004.07.001
Kirchman DL (2018) Processes in anoxic environments. In: Processes in microbial ecology, 2nd edn. Oxford University Press, pp 195–216
Krummenauer D, Samocha T, Poersch L, Lara G, Wasielesky W (2014) The reuse of water on the culture of pacific white shrimp, Litopenaeus vannamei in BFT system. J World Aquac Soc 45(1):3–14. https://doi.org/10.1111/jwas.12093
Kuhn DD, Smith SA, Boardman GD, Angier MW, Marsh L, Flick GJ (2010) Chronic toxicity of nitrate to Pacific white shrimp, Litopenaeus vannamei: impacts on survival, growth, antennae length, and pathology. Aquaculture 309(1–4):109–114. https://doi.org/10.1016/j.aquaculture.2010.09.014
Lara G, Honda M, Poersch L, Wasielesky W (2017) The use of biofilm and different feeding rates in biofloc culture system: the effects in shrimp growth parameters. Aquac Int 25(5):1959–1970. https://doi.org/10.1007/s10499-017-0151-0
Letelier-Gordo CO, Huang X, Aalto SL, Pedersen PB (2020) Activated sludge denitrification in marine recirculating aquaculture system effluent using external and internal carbon sources. Aquac Eng 90(May):102096. https://doi.org/10.1016/j.aquaeng.2020.102096
Lin Y-C, Chen J-C (2001) Acute toxicity of ammonia on Litopenaeus vannamei Boone juveniles at different salinity levels. J Exp Mar Bio Ecol 259(1):109–119. https://doi.org/10.1016/s0022-0981(01)00227-1
Lin Y-C, Chen J-C (2003) Acute toxicity of nitrite on Litopenaeus vannamei (Boone) juveniles at different salinity levels. Aquaculture 224(1–4):193–201. https://doi.org/10.1016/S0044-8486(03)00220-5
Liu C, Olivares CI, Pinto AJ, Lauderdale CV, Brown J, Selbes M, Karanfil T (2017) The control of disinfection byproducts and their precursors in biologically active filtration processes. Water Res 124:630–653. https://doi.org/10.1016/j.watres.2017.07.080
Luo G, Xu J, Meng H (2020) Nitrate accumulation in biofloc aquaculture systems. Aquaculture 520:734675. https://doi.org/10.1016/j.aquaculture.2019.734675
Martins MA, Poli MA, Legarda EC, Pinheiro IC, Carneiro RFS, Pereira SA, Martins ML, Gonçalves P, Schleder DD, do Nascimento Vieira F (2020) Heterotrophic and mature biofloc systems in the integrated culture of Pacific white shrimp and Nile tilapia. Aquaculture 514(August 2019):734517. https://doi.org/10.1016/j.aquaculture.2019.734517
Melo Filho MES, Owatari MS, Mouriño JLP, Lapa KR, Soares HM (2020) Application of nitrification and denitrification processes in a direct water reuse system for pacific white shrimp farmed in biofloc system. Aquac Eng 88(August 2019):102043. https://doi.org/10.1016/j.aquaeng.2020.102043
Noguera-Oviedo K, Aga DS (2016) Lessons learned from more than two decades of research on emerging contaminants in the environment. J Hazard Mater 316:242–251. https://doi.org/10.1016/j.jhazmat.2016.04.058
Philippot L (2002) Denitrifying genes in bacterial and Archaeal genomes. Biochim Biophys Acta - Gene Struct Expr 1577(3):355–376. https://doi.org/10.1016/S0167-4781(02)00420-7
Piedrahita RH (2003) Reducing the potential environmental impact of tank aquaculture effluents through intensification and recirculation. Aquaculture 226:35–44. https://doi.org/10.1016/S0044-8486(03)00465-4
Ponce-Palafox J, Martinez-Palacios CA, Ross LG (1997) The effects of salinity and temperature on the growth and survival rates of juvenile white shrimp, Penaeus vannamei, Boone, 1931. Aquaculture 157(1–2):107–115. https://doi.org/10.1016/S0044-8486(97)00148-8
Prates E, Holanda M, Pedrosa VF, Monserrat JM, Wasielesky W (2023a) Compensatory growth and energy reserves changes in the Pacific white shrimp (Litopenaeus vannamei) reared in different temperatures and under feed restriction in biofloc technology system (BFT). Aquaculture 562(September 2022):738821. https://doi.org/10.1016/j.aquaculture.2022.738821
Prates ECA, Damasceno JN, Okamoto M, Holanda M, Costa M, Monserrat JM, Wasielesky W (2023b) Determinação da toxicidade aguda de nitrato para o camarão branco do Pacífico Litopenaeus vannamei. In: Anais Eletrônicos do X Aquaciência. AQUABIO, Florianópolis, SC
Preena PG, Rejish Kumar VJ, Singh ISB (2021) Nitrification and denitrification in recirculating aquaculture systems: the processes and players. Rev Aquac 13(4):2053–2075. https://doi.org/10.1111/raq.12558
Reis WG, Wasielesky W Jr, Abreu PC, Brandão H, Krummenauer D (2023) The influence of different light wavelengths in the culture of the Pacific white shrimp Litopenaeus vannamei reared in BFT using LED lights. Aquaculture 563:738924. https://doi.org/10.1016/j.aquaculture.2022.738924
Reis WG, Wasielesky W, Abreu PC, Brandão H, Krummenauer D (2019) Rearing of the Pacific white shrimp Litopenaeus vannamei (Boone, 1931) in BFT system with different photoperiods: effects on the microbial community, water quality and zootechnical performance. Aquaculture 508(April):19–29. https://doi.org/10.1016/j.aquaculture.2019.04.067
Richardson S, Plewa M, Wagner E, Schoeny R, Demarini D (2007) Occurrence, genotoxicity, and carcinogenicity of regulated and emerging disinfection by-products in drinking water: a review and roadmap for research. Mutat Res Mutat Res 636(1–3):178–242. https://doi.org/10.1016/j.mrrev.2007.09.001
Romanucci V, Siciliano A, Galdiero E, Guida M, Luongo G, Liguori R, Di Fabio G, Previtera L, Zarrelli A (2019) Disinfection by-products and ecotoxic risk associated with hypochlorite treatment of tramadol. Molecules 24(4):693. https://doi.org/10.3390/molecules24040693
Schuler DJ, Boardman GD, Kuhn DD, Flick GJ (2010) Acute toxicity of ammonia and nitrite to pacific white shrimp, Litopenaeus vannamei, at low salinities. J World Aquac Soc 41(3):438–446. https://doi.org/10.1111/j.1749-7345.2010.00385.x
Shanahan JW, Semmens MJ (2015) Alkalinity and pH effects on nitrification in a membrane aerated bioreactor: an experimental and model analysis. Water Res 74:10–22. https://doi.org/10.1016/j.watres.2014.12.055
Silveira LGP, Rosas VT, Krummenauer D, Poersch LH, Wasielesky W Jr (2020) Comparison between horizontal and vertical substrate in shrimp super-intensive culture in bioflocs system. Aquac Eng 96(November 2021):102218. https://doi.org/10.1016/j.aquaeng.2021.102218
Strickland JDH, Parsons TR (1972) A practical handbook of seawater analysis, 2nd edn. Fisheries Research Board Of Canada, Ottawa
UNESCO (1983) Chemical methods for use in marine environmental monitoring. In: Manuals and guides 12. Intergovernmental Oceanographic Commission, Paris, France
Valencia-Castañeda G, Frías-Espericueta MG, Vanegas-Pérez RC et al (2018) Acute toxicity of Ammonia, Nitrite and Nitrate to shrimp litopenaeus vannamei postlarvae in low-salinity water. Bull Environ Contam Toxicol 101:229–234. https://doi.org/10.1007/s00128-018-2355-z
Von Sperling M (2007) Basic principles of wastewater treatment (Biological Wastewater Treatment Volume 2). IWA Publishing, London, UK
Wang J, Zheng F, Yu Z, Chen J, Lu H (2022a) Dissolved organic nitrogen derived from wastewater denitrification: composition and nitrogenous disinfection byproduct formation. J Hazard Mater 440(August):129775. https://doi.org/10.1016/j.jhazmat.2022.129775
Wang Y, Liu H, Yang X, Wang L (2022b) Aquatic toxicity and aquatic ecological risk assessment of wastewater-derived halogenated phenolic disinfection byproducts. Sci Total Environ 809:151089. https://doi.org/10.1016/j.scitotenv.2021.151089
Wasielesky W, Atwood H, Stokes A, Browdy CL (2006) Effect of natural production in a zero exchange suspended microbial floc based super-intensive culture system for white shrimp Litopenaeus vannamei. Aquaculture 258(1–4):396–403. https://doi.org/10.1016/j.aquaculture.2006.04.030
Xavier M, Wasielesky Júnior W, Hostins B, Bequé E, Krummenauer D (2022) The use of a flocculant additive and its effect on biofloc formation, nitrification, and zootechnical performance during the culture of Pacific white shrimp Penaeus vannamei (Boone, 1931) in a BFT system. Lat Am J Aquat Res 50(2):181–196. https://doi.org/10.3856/vol50-issue2-fulltext-2777
Xu W, Xu Y, Su H, Hu X, Xu Y, Li Z, Wen G, Cao Y (2021) Production performance, inorganic nitrogen control and bacterial community characteristics in a controlled biofloc-based system for indoor and outdoor super-intensive culture of Litopenaeus vannamei. Aquaculture 531(June 2020):735749. https://doi.org/10.1016/j.aquaculture.2020.735749
Zar JH (2010) Biostatistical analysis, 5th edn. Prentice Hall, Upper Saddle River
Zheng F, Wang J, **ao R, Chai W, **ng D, Lu H (2021) Dissolved organic nitrogen in wastewater treatment processes: transformation, biosynthesis and ecological impacts. Environ Pollut 273:116436. https://doi.org/10.1016/j.envpol.2021.116436
Acknowledgements
Special thanks to GUABI Animal Health and Nutrition, AQUATEC, TREVISAN, and Al Aqua for donating the experimental diets, shrimp post-larvae, and aeration system respectively.
Funding
The authors are grateful for the financial support provided by the National Council for Scientific and Technological Development (CNPq), Coordination for the Improvement of Higher-Level Personnel (CAPES), and Foundation for Research Support of the State of Rio Grande do Sul (FAPERGS), grant number 23/2551-0000180-2. Wasielesky, W. is a research fellow of CNPq under process number PQ 307741/2022-2.
Author information
Authors and Affiliations
Contributions
HB: conceptualization; methodology; investigation; data curation; formal analysis; writing—original draft; writing—review and editing; visualization. WGR: conceptualization; methodology; investigation; data curation; formal analysis; writing—review and editing. DK: conceptualization; writing—review; supervision; funding acquisition; resources. WWJ: conceptualization; writing—review and editing; supervision; project administration; funding acquisition; resources.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Handling Editor: Mauricio G. C. Emerenciano
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Brandão, H., dos Reis, W.G., Krummenauer, D. et al. Growth performance of Litopenaeus vannamei under biofloc system using denitrified seawater. Aquacult Int 32, 3129–3145 (2024). https://doi.org/10.1007/s10499-023-01315-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-023-01315-0