Abstract
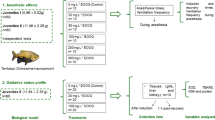
The objective of the present study was to evaluate the action of benzocaine and eugenol over anesthesia stages and the use of three anticoagulants (ethylenediamine tetraacetic acid, sodium citrate, and sodium heparin) over hematological and biochemical parameters and erythrocyte osmotic fragility in tambaquis Colossoma macropomum. For that, fish were distributed into two experimental groups: G1: fish anesthetized with benzocaine and G2: fish anesthetized with eugenol. Induction and recovery times of anesthesia were evaluated and blood samples with different anticoagulants were collected for hematological and biochemical analysis and osmotic erythrocyte osmotic fragility. Fish anesthetized with eugenol showed a longer period of anesthesia, shorter induction time, and longer recovery time. In addition, fish anesthetized with benzocaine by using sodium citrate showed leukopenia and lymphopenia and morphological alterations when compared to the other anticoagulants. Furthermore, fish anesthetized with eugenol by using sodium citrate showed significantly lower values (P < 0.05) of hematocrit and hemoglobin when compared to other anticoagulants and there were significant differences in erythrocyte osmotic fragility between anticoagulants in fish anesthetized with eugenol. The results observed in this study indicate that eugenol is a good alternative as anesthetics for tambaquis when compared to benzocaine because of its rapid anesthesia induction and few alterations in blood parameters. In addition, the use of EDTA facilitates the erythrocyte conservation and cause few blood alterations compared to other anticoagulants.


Similar content being viewed by others
References
Allen P (1994) Changes in the hematological profile of the cichlid Oreochromis aureus (Steindachner) during acute inorganic Mercury intoxication. Comp Biochem Physiol C Toxicol Pharmacol 108:117–121. https://doi.org/10.1016/1367-8280(94)90097-3
Aydın B, Barbas LAL (2020) Sedative and anesthetic properties of essential oils and their active compounds in fish: a review. Aquaculture 520:734999. https://doi.org/10.1016/j.aquaculture.2020.734999
Barros MM, Ranzani Paiva MJT, Pezzato LE (2009) Hematological response and growth performance of Nile tilapia fed diets containing folic acid. Aquaculture Research 40:95–98. https://doi.org/10.1111/j.1365-2109.2009.02175.x
Baynes JW, Dominiczark MH (2005) Bioquímica Médica. Mosby, Elsevier
Bittencourt NLR, Molinari LM, Scoaris DO, Pedroso RB, Nakamura CV, Nakamura TU, Abreu Filho BA, Diasfilho BP (2003) Hematological and biochemical values for Nile tilapia Oreochromis niloticus cultured in semi-intensive system. Acta Scientiarum 25:385–389
Castro KS, Sousa AG, Fugimura MMS, Vaz LJ, Marcusso PF, Claudiano GS (2019) Estabilidade das variáveis hematológicas em sangue de Colossoma macropomum armazenado com diferentes anticoagulantes. Ragros 11:171–180. https://doi.org/10.18542/ragros.v11i2.8963
Claudiano GS, Yunis-Aguinaga J, Marinho-Neto FA, Miranda RL, Martins IM, Otani FS, Mundim AV, Marzocchi-Machado CM, Moraes JRE, Moraes FR (2019) Hematological and immune changes in Piaractus mesopotamicus in the sepsis induced by Aeromonas hydrophila. Fish Shellfish Immunol 88:259–265. https://doi.org/10.1016/j.fsi.2019.01.044
Cosenza GR, Claudiano GS, Marcusso PF, Eto SF, Manrique WG, Loureiro BA, Shimada MT, Salvador R, Moraes JRE, Moraes FR (2014) Influence of glyceryl guaiacolate ether on anesthetics in tilapia compared to benzocaine and eugenol. Rev. MVZ Córdoba 19:3944–3953. https://doi.org/10.21897/rmvz.114
Costa LFA, Claudiano G.S, Ramos-Espinoza FC, Marinho-Neto FA, Marcusso PF, Yunis-Aguinaga J, Moraes FR; Moraes JRE (2020). Hematological and glycemic changes in Oreochromis niloticus subjected to acute stress by syphoning. J Vet Sc Public Health 7: 97-101. https://doi.org/10.4025/rcvsp.v7i2.46639
Delbon MCE, Paiva MJTR (2012) Eugenol em juvenis de tilápia do Nilo: concentrações e administrações sucessivas. Bol Inst Pesca 38:43–52
Drabkin DL, Austin JH (1935) Spectrophotometric studies II. Preparations from washed blood cells; nitric oxide hemoglobin and sulfhemoglobin. J Biol Chem 112(1):51–65
Fazio F (2019) Fish hematology analysis as an important tool of aquaculture: a review. Aquaculture 500:237–242. https://doi.org/10.1016/j.aquaculture.2018.10.030
Gimbo RY, Saita MV, Gonçalves AFN, Takahashi LS (2008) Diferentes concentrações de benzocaína na indução anestésica do lambari-do raboamarelo (Astyanax altiparanae). Rev Brasa Saúde Prod Anim 9(2):350–357
Goldenfarb PB, Bowyer FP, Hall E, Brosious E (1971) Reproducibility in the hematology laboratory: the microhematocrit determination. Am J Clin Pathol 56:35–39
Grush J, Noakes DLG, Moccia RD (2004) The efficacy of clove oil as an anesthetic for the zebrafish, Danio rerio (Hamilton). Zebrafish 1:46–53. https://doi.org/10.1089/154585404774101671
Hasan M, Bart AN (2007) Improved survival of rohu, Labeo rohita (Hamilton-Buchanan) and silver carp, Hypophthalmichthys molitrix (Valenciennes) fingerlings using low-dose quinaldine and benzocaine during transport. Aquac Res 38:50–58. https://doi.org/10.1111/j.1365-2109.2006.01628.x
Hikasa Y, Takase K, Ogasawara T, Ogasawara S, Ogasawara T, Ogasawara S (1986) Anaesthesia and recovery with tricain methansulfonate, eugenol and thiopental sodium in carp, Cyprinus carpio. Jpn J Sci 48(2):341–351
Holloway AC, Keene JL, Noakes DG, Moccia RD (2004) Effects of clove oil and MS-222 on blood hormone profiles in rainbow trout Oncorhynchus mykiss. Aquac Res 35:1025–1030. https://doi.org/10.1111/j.1365-2109.2004.01108.x
Inoue L, Afonso L, Iwama G, Moraes G (2005) Effects of clove oil on the stress response of matrinxã (Brycon cephalus) subjected to transport. Acta Amazon 35:145–151. https://doi.org/10.1590/S0044-59672005000200018
Inoue LAKA, Boi**k CL, Ribeiro PT, Silva AMD, Affonso EG (2011) Avaliação de respostas metabólicas do tambaqui exposto ao eugenol em banhos anestésicos. Acta Amazon 41(2):327–332. https://doi.org/10.1590/S0044-59672011000200020
Ishikawa NM, Ranzani-Paiva MJT, Lombardi JV (2008) Total leukocyte counts methods in fish, Oreochromis niloticus. Arch Vet Sci 13(1):54–63
Ishikawa NM, Pádua SB, Satake F, Hisano H, Jerônimo GT, Martins ML (2010) Heparina e Na2 EDTA como anticoagulantes para surubim híbrido (Pseudoplatystoma reticulatum × P. corruscan): eficácia e alterações hematológicas. Cienc Rural 40:1557–1561. https://doi.org/10.1590/S0103-84782010005000113
Iversen M, Finstad B, Mckinley RS, Eliassen RA (2003) The efficacy of metomidate, clove oil, Aqui-STM and Benzoak® as anaesthetics in Atlantic salmon (Salmo salar L.) smolts, and their potential stress-reducing capacity. Aquaculture 221(1-4):549–566. https://doi.org/10.1016/S0044-8486(03)00111-X
Lătărełu A, Teusdea V, Furnaris F, Bunea R, Mitrănescu E (2013) Comparative study of the effect of different anticoagulants on blood cell morphology in common carp (C. carpio) and rainbow trout (O. mykiss). Bull UASVM Anim Sci Biotechnol 70(2):284–288. https://doi.org/10.15835/BUASVMCN-ASB:70:2:9473
Mafuvadze B, Erlwanger KH (2007) The effect of EDTA, Heparin and storage on the erythrocyte osmotic fragility, plasma osmolality and hematocrit of adult ostriches (Struthio camelus). Arch Vet Sci 77:427–434
Nussey G, Van Vuren JHJ, Du Preez HH (1995) Effects of copper on the haematological and osmoregulation of the Mozambique tilapia. Oreochromis mossambicus (Cichlidae). Comp Biochem Physiol C Toxicol Pharmacol 111:369–380. https://doi.org/10.1016/0742-8413(95)00063-1
Okamoto MH, Tesser MB, Louzada LR, Santos RA, Sampaio LA (2009) Benzocaína e eugenol como anestésicos para juvenis do pampo Trachinotus marginatus. Ciência Rural 39:866–870. https://doi.org/10.1590/S0103-84782008005000100
Pádua SB, Neto JD, Sakabe R, Claudiano GS, Chaga EC, Pilarski F (2013) Variáveis hematológicas em tambaquis anestesiados com óleo de cravo e benzocaína. Pesq Agrop Bras 48(8):1171–1174. https://doi.org/10.1590/S0100-204X2013000800056
Palmer L, Briggs C, Mcfadden S, Zini G, Burthem J, Rozenberg G, Proytcheva M, Machin SJ (2015) ICSH recommendations for the standardization of nomenclature and grading of peripheral blood cell morphological features. Int Jnl Lab Hem 37:287–303. https://doi.org/10.1111/ijlh.12327
Parpart AK, Lorenz PB, Parpart ER, Gregg JR, Chase AM (1947) The osmotic resistance (fragility) of human red cells. J Clin Invest Ann Arbor 26:636–640. https://doi.org/10.1172/JCI101847
Priborsky J, Velisek J (2018) A review of three commonly used fish anesthetics. Rev Fish Sci Aqua 26(4):417–442. https://doi.org/10.1080/23308249.2018.1442812
Prince A, Powell C (2000) Clove oil as an anesthetic for invasive field procedures on adult rainbow trout. N Am J Fish Manag 20(4):1029–1032. https://doi.org/10.1577/1548-8675(2000)0202.0.CO;2
Ross LG, Ross BR (2008) Anaesthetic and sedative techniques for aquatic animals. Blackwell Science, Oxford
Roubach R, Gomes LC, Leão Fonseca FA, Val AL (2005) Eugenol as an efficacious anaesthetic for tambaqui, Colossoma macropomum (Cuvier). Aquac Res 36(11):1056–1061. https://doi.org/10.1111/j.1365-2109.2005.01319.x
Salvador R, Claudiano GS, Loureiro BA, Marcusso PF, Eto SF, Pilarski F, Toazza CS, Moraes JRE, Moraes FR (2013) Performance and hematological profile of Nile tilapia fed with Saccharomyces cerevisiae and vaccinated against Streptococcus agalactiae. Pesq Agropec Bras 48(8):892–898. https://doi.org/10.1590/S0100-204X2013000800012
Sarkar M, Barari SK, Mandal DB, Nandankar UA, Basu A, Mohanty TK, Ray S (1999) The effect of anti-coagulants on the osmotic fragility of erythrocytes in the yak (Poephagus grunniens). Vet J 157:91–93. https://doi.org/10.1053/tvjl.1998.0257
Sladky KK, Swanson CR, Stoskopf MK, Loomis MR, Lewbart GA (2001) Comparative efficacy of tricaine methanesulfonate and clove oil for use as anesthetics in red pacu (Piaractus brachypomis). Amer J Vet Res 62:337–342. https://doi.org/10.2460/ajvr.2001.62.337
Souza RAR, Carvalho CVA, Nunes FF, Scopel BR, Guarizi JD, Tsuzuki MY (2018) Comparative effect of benzocaine, menthol and eugenol as anesthetics for juvenile fat snook. Bol Inst Pesca 38(3):247–255
Souza C, Baldissera M, Baldisserotto B, Heinzmann B, Martos-Sitcha JÁ, Mancera JM (2019) Essential oils as stress-reducing agents for fish aquaculture: a review. Front Physiol 10:785. https://doi.org/10.3389/fphys.2019.00785
Sudagara M, Mohammadizarejabada A, Mazandarania R, Pooralimotlagha S (2000) The efficacy of clove powder as an anesthetic and its effects on hematological parameters on roach (Rutilus rutilus). Journal of Aquaculture Feed Science and Nutrition 1:1–5
Tavares-Dias M, Sandrin EFS (1998) Influence of anticoagulants and blood storage on hematological values in tambaqui, Colossoma macropomum. Acta Sci. 20:151–155. https://doi.org/10.4025/actascibiolsci.v20i0.4465
Tavares-Dias M, Moraes FR, Imoto ME (2008) Hematological parameters in two neotropical freshwater teleost, Leporinus macrocephalus (Anostomidae) and Prochilodus lineatus (Prochilodontidae). Biosci J 24(3):96–101
Trinder P (1969) Determination of blood glucose using 4-amino phenazone as oxygen acceptor. J Clin Pathol 22(2):246. https://doi.org/10.1136/jcp.22.2.246-b
Van Vliet KJ, Smit GL, Pieterse JJ, Schoonbee HJ, Van Vuren JHJ (1985) The effects of generally used anticoagulants on the hemolysis of fish erythrocytes. Water SA, Pretoria 2:87–92. https://doi.org/10.1016/j.cbpc.2007.04.004
Vidal LVO, Albinati RCB, Albinati ACL, Mecêdo GR (2006) Utilização do eugenol como anestésico para o manejo de juvenis de pintado (Pseudoplatystoma corruscans). Acta Scientiarum. Biological Sciences 28:275–279. https://doi.org/10.4025/actascibiolsci.v28i3.400
Vidal LVO, Furuya WM, Graciano TS, Schamber CR, da Silva LCR, dos Santos LD, de Souza SR (2007) Eugenol como anestésico para juvenis de matrinxã (Brycon cephalus). Rev Bras Saúde Prod Anim 8:335–342
Vidal LVO, Albinati RCV, Albinati ACL (2008) Eugenol como anestésico para a tilápia-do-nilo. Pesq Agropec Bras 43:1069–1074. https://doi.org/10.1590/S0100-204X2008000800017
Walencik J, Witeska M (2007) The effects of anticoagulants on hematological índices and blood cell morphology of common carp (Cyprinus carpio L.). Comp Biochem Physiol C Toxicol Pharmacol 146:331–335. https://doi.org/10.1016/j.cbpc.2007.04.004
Woynárovich A, Van Anrooy R (2019) Field guide to the culture of tambaqui (Colossoma macropomum, Cuvier, 1816). FAO Fisheries and Aquaculture Technical Paper N°. 624. FAO, Rome 132 p
Yunis-Aguinaga J, Fernandes DC, Eto SF, Claudiano GS, Marcusso PF, Marinho-Neto FA, Fernandes JB, Moraes FR, Moraes JRE (2016) Dietary camu camu, Myrciaria dubia, enhances immunological response in Nile tilapia. Fish Shellfish Immunol 58:284–291. https://doi.org/10.1016/j.fsi.2016.08.030
Availability of data and materials
Not applicable
Code availability
Not applicable
Funding
The authors are grateful for the support of the Federal University of Western Pará –UFOPA (10/2018 PROPPIT/UFOPA).
Author information
Authors and Affiliations
Contributions
Andria Gama Sousa: Formal analysis, investigation, methodology, project administration, writing—original draft preparation
Acácio H. B. Pacheco: Investigation, methodology, writing—review and editing
Gilson A. Siqueira-Pinto: Investigation, visualization, writing—review and editing
Gleika T. J. dos Reis: Investigation, resources, writing—review and editing
Michelle M. S. Fugimura: Investigation, methodology, validation, writing—review and editing
Luciano J. Vaz: Investigation, methodology, validation
Paulo F. Marcusso: Conceptualization, funding acquisition, supervision, writing—review and editing
Fernando C. Ramos-Espinoza: Conceptualization, funding acquisition, supervision, writing—review and editing
Gustavo S. Claudiano: Conceptualization, funding acquisition, supervision, validation, writing—review and editing
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Handling Editor: Gavin Burnell
Rights and permissions
About this article
Cite this article
Sousa, A.G., Pacheco, A.H.B., Siqueira-Pinto, G.A. et al. Comparative study of hematological parameters of Colossoma macropomum anesthetized with benzocaine and eugenol by using different anticoagulants. Aquacult Int 29, 977–988 (2021). https://doi.org/10.1007/s10499-021-00668-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-021-00668-8