Abstract
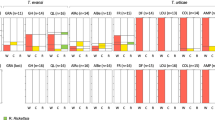
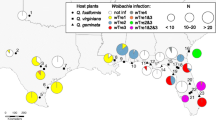
Whereas endosymbiont-induced incompatibility is known to occur in various arthropod taxa, such as spider mites, insects and isopods, it has been rarely reported in plant-inhabiting predatory mites (Acari: Phytoseiidae). Recent cross-breeding studies with the phytoseiid mite Neoseiulus paspalivorus De Leon revealed a complete post-mating reproductive isolation between specimens collected from three geographic origins—Northeast Brazil (South America), Benin and Ghana (West Africa)—even though they are morphologically similar. We carried out a study to assess to what extent these populations exhibit genetic differences and whether endosymbionts are involved in the incompatibility. First, we used the mitochondrial cytochrome oxidase I (COI) gene to assess genetic diversity among the three populations. Second, we used a PCR-based method to check for the presence of Wolbachia and/or Cardinium in these populations, and we determined their phylogenetic relationships using specific primers for Wolbachia and Cardinium 16S rDNA genes. Third, we also conducted a test using an antibiotic (tetracycline) in an attempt to eliminate the symbionts and evaluate their effects on the reproductive compatibility of their host. Based on the DNA sequences of their COI genes, specimens of the three populations appear to be genetically similar. However, the 16S rDNA gene sequences of their associated endosymbionts differed among the three populations: the Benin and Brazil populations harbour different strains of Wolbachia symbionts, whereas the Ghana population harbours Cardinium symbionts. In response to antibiotic treatment females of each of the three populations became incompatible with untreated males of their own population, similar to that observed in crossings between females from one geographic population and males from another. Compatibility was restored in crosses involving uninfected Brazil females and uninfected Benin males, whereas the reciprocal crosses remained incompatible. Cardinium symbionts seem to be essential for oviposition in the Ghana population. It is concluded that their associated bacterial symbionts are the cause of the post-mating reproductive isolation previously observed among the three geographic populations. This insight is relevant to biological control of coconut mites for which N. paspalivorus is an effective predator, because introducing one geographic strain into the population of another (e.g. in field releases or mass cultures) may cause population growth depression.


Similar content being viewed by others
References
Bandi C, Dunn AM, Hurst GD, Rigaud T (2001) Inherited microorganisms, sex-specific virulence and reproductive parasitism. Trends Parasitol 17:88–94
Bordenstein SR (2003) Symbiosis and the origin of the species. In: Bourtzis K, Miller T (eds) Insect symbiosis. CRC Press, Boca Raton, pp 283–304
Bordenstein SR, Werren JH (2003) Host genome determines cytoplasmic incompatibility type in the haplodiploid genus Nasonia. Genetics 164:223–233
Bordenstein SR, O’Hara FP, Werren JH (2001) Wolbachia-induced bidirectional incompatibility precedes other hybrid incompatibility in Nasonia. Nature 409:707–710
Breeuwer JAJ (1997) Wolbachia and cytoplasmic incompatibility in the spider mite Tetranychus urticae and T. turkestani. Heredity 79:41–47
Breeuwer JAJ, Werren JH (1990) Microorganisms associated with chromosome destruction and reproductive isolation between two insect species. Nature 346:558–560
Breeuwer JAJ, Werren JH (1993) Cytoplasmic incompatibility and bacterial density in Nasonia vitripennis. Genetics 135:565–574
Breeuwer JAJ, Werren JH (1995) Hybrid breakdown between two haplodiploid species: the role of nuclear and cytoplasmic genes. Evolution 49:705–717
Charlat S, Bourtzis K, Mercot H (2001) Wolbachia-induced cytoplasmic incompatibility. In: Seckbach J (ed) Symbiosis: mechanism and model systems. Kluwer Academic, Dordrecht, The Netherlands, pp 621–644
Duron O, Bouchon D, Boutin S, Bellamy L, Zhou L, Engelstädter J, Hurst GD (2008) The diversity of reproductive parasites among arthropods: Wolbachia do not walk alone. BMC Biol 6:27. doi:10.1186/1741-7007-6-27
Engelstaedter J, Hurst GDD (2009) The ecology and evolution of microbes that manipulate host reproduction. Annu Rev Ecol Evol Syst 40:127–149
Engelstaedter J, Hammerstein P, Hurst GDD (2007) The evolution of endosymbiont density in doubly infected host species. J Evol Biol 20:685–695
Enigl M, Schausberger P (2007) Incidence of the endosymbionts, Wolbachia, Cardinium and Spiroplasma in phytoseiid mites and associated preys. Exp Appl Acarol 42:75–85
Famah Sourassou N, Hanna R, Zannou I, Moraes GJ, Negloh K, Sabelis MW (2011) Morphological variation and reproductive incompatibility of three coconut-mite-associated populations of predatory mites identified as Neoseiulus paspalivorus (Acari: Phytoseiidae). Exp Appl Acarol 53:323–338
Famah Sourassou N, Hanna R, Zannou I, Breeuwer JAJ, Moraes GJ, Sabelis MW (2012) Morphological, molecular and cross-breeding analysis of geographic populations of coconut-mite associated predatory mites identified as Neoseiulus baraki: evidence for cryptic species? Exp Appl Acarol 57:15–36
Giordano R, O’Neill LS, Robertson HM (1995) Wolbachia infections and the expression of cytoplasmic incompatibility in Drosophila sechellia and D. mauritiana. Genetics 140:1307–1317
Gotoh T, Noda H, Hong XY (2003) Wolbachia distribution and cytoplasmic incompatibility based on a survey of 42 spider mite species (Acari: Tetranychidae) in Japan. Heredity 91:208–216
Gotoh T, Noda H, Fujita T, Iwadate K, Higo Y, Saito S, Ohtsuka S (2005a) Wolbachia and nuclear–nuclear interactions contribute to reproductive incompatibility in the spider mite Panonychus mori (Acari: Tetranychidae). Heredity 94:237–246
Gotoh T, Oku H, Moriya K, Odawara M (2005b) Nucleus-cytoplasm interactions causing reproductive incompatibility between two populations of Tetranychus quercivorus Ehara and Gotoh (Acari: Tetranychidae). Heredity 74:105–114
Gotoh T, Noda H, Ito S (2007) Cardinium symbionts cause cytoplasmic incompatibility in spider mites. Heredity 98:13–20
Hall TA (1999) BioEdit: a user-friendly biological sequence editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp Ser 41:95–98
Hebert PD, Ratnasingham S, deWaard JR (2003) Barcoding animal life: cytochrome c oxidase subunit 1 divergences among closely related species. Proc R Soc B 270(Suppl 1):S96–S99
Hinomoto N, Osakabe M, Gotoh T, Takafuji A (2001) Phylogenetic analysis of the green and red forms of the two-spotted spider mite, Tetranychus urticae Koch (Acari: Tetranychidae), in Japan, based on the mitochondrial cytochrome oxidase subunit I sequences. Appl Entomol Zool 36:459–564
Hinomoto N, Tran PD, Pham AT, Le TBN, Tajima R, Ohashi K, Osakabe M, Takafuji A (2007) Identification of spider mite (Acari: Tetranychidae) by DNA sequence: a case study in the North Vietnam. Int J Acarol 33:53–60
Hoffmann AA, Turelli M (1988) Unidirectional incompatibility in Drosophila simulans: inheritance, geographic variation and fitness effects. Genetics 119:435–444
Hoffmann AA, Turelli M (1997) Cytoplasmic incompatibility in insects. In: O’Neil SL, Hoffmann AA, Werren JH (eds) Influential passengers: inherited microorganisms and arthropod reproduction. Oxford University Press, Oxford, pp 42–80
Hoy MA, Cave FE (1988) Premating and post-mating isolation among populations of Metaseiulus occidentalis (Nesbitt) (Acarina: Phytoseiidae). Hilgardia 56:1–20
Hurst JDD, Jiggins FM, Schulenburg JH, Bertrand D, West SA, Goriceheva II, Zakharov IA, Werren JH, Stouthamer R, Majerus MEN (1999) Male killing Wolbachia in two species of insects. Proc R Soc Lond B 266:735–740
Johanowicz DL, Hoy MA (1998) Experimental induction and termination of non-reciprocal reproductive incompatibility in a parahaploid mite. Entomol Exp Appl 87:51–58
Jukes TH, Cantor CR (1969) Evolution of protein molecules pp. In: Munro HN (ed) Mammalian protein metabolism. Academic Press, New York, USA, pp 21–132
Kajeyama D, Nishimura G, Hoshizaki S, Ishikawa Y (2002) Feminizing Wolbachia in an insect, Ostrinia furnacalis (Lepidoptera: Crambidae). Heredity 88:444–449
Koevoets T, Beukeboom LW (2009) Genetics of postzygotic isolation and Haldane’s rule in haplodiploid. Heredity 102:16–23
Koevoets T, Niehuis O, van de Zande L, Beukeboom LW (2011) Hybrid incompatibilities in the parasitic wasp genus Nasonia. Heredity 108:302–311
Kurtti TJ, Munderloh UG, Andreadis TG, Magnarelli LA (1996) Tick cell culture isolation of an intracellular prokaryote from the tick Ixodes scapularis. J Invertebr Pathol 6:318–321
Laven H (1959) Speciation by cytoplasmic isolation in Culex strains. Evolution 5:370–375
Laven H (1967) Evolution and speciation in Culex pipientis. In: Wright JW, Pal R (eds) Genetics of insects vectors of disease. Holland, Elsevier, pp 251–271
Lawson-Balagbo ML, Gondim MGC, Moraes GJ, Hanna R, Schausberger P (2008) Exploration of acarine fauna on coconut palm in Brazil with emphasis on Aceria guerreronis (Acari: Eriophyidae) and its natural enemies. Bull Entomol Res 98:83–96
Moraes GJ, Lopez PC, Fernando LCP (2004) Phytoseiid mites (Acari: Phytoseiidae) of coconut growing areas in Sri Lanka, with description of three new species. J Acarol Soc Jpn 13:141–160
Navajas M, Boursot P (2003) Nuclear ribosomal DNA monophyly versus mitochondrial DNA polyphyly in two closely related mite species: the influence of life history and molecular drive. Proc R Soc Lond B 270:S124–S127
Navajas M, Gutierrez M, Langnel J (1996) Mitochondrial cytochrome oxidase I in tetranychid mites: a comparison between molecular phylogeny and change of morphological and life history traits. Bull Entomol Res 86:407–417
Navajas M, Langnel J, Gutierrez M, Boursot P (1998) Species wide homogeneity of nuclear ribosomal ITS2 sequences in the spider-mite Tetranychus urticae contrasts with extensive mitochondrial COI polymorphism. Heredity 87:742–752
Navajas M, Gutierrez J, Lagnel L, Fauvel G, Gotoh T (1999) DNA sequences and cross-breeding experiments in the hawthorn spider mite Amphitetranychus viennensis reveal high genetic differentiation between Japanese and French populations. Entomol Exp Appl 90:113–122
Negloh K, Hanna R, Schausberger P (2011) The coconut mite, Aceria guerreronis, in Benin and Tanzania: occurrence, damage and associated acarine fauna. Exp Appl Acarol 55:361–374
Noronha ACS, Moraes GJ (2002) Variaçãoes morfológicas intra e interpopulacionaes de Euseius citrifolius Denmark and Muma e Euseius concordis Chant (Acari, Phytoseiidae). Revista brasileira de Zoologia 19:1111–1122
Noronha ACS, Moraes GJ (2004) Reproductive compatibility between mite populations previously identified as Euseius concordis (Acari: Phytoseiidae). Exp Appl Acarol 32:271–279
O’Neill SL (1989) Cytoplasmic incompatibility in Tribolium confusum. J Invertebr Pathol 53:132–134
O’Neill SL, Karr TL (1990) Bidirectional incompatibility between conspecific populations of Drosophila simulans. Nature 348:178–180
O’Neil SL, Giordano R, Colbert AME, Karl TL, Robertson HM (1992) 16s rDNA phylogenetic analysis of the bacterial endosymbionts associated with cytoplasmic incompatibility in insects. Proc Natl Acad Sci USA 89:2699–2702
Okassa M, Tixier MS, Cheval B, Kreiter S (2009) Molecular and morphological evidence for new species status within the genus Euseius (Acari: Phytoseiidae). Can J Zool 87:689–698
Pannebakker BA, Loppin B, Elemans CPH, Humblot L, Vavre F (2007) Parasitic inhibition of cell death facilitates symbiosis. Proc Natl Acad Sci USA 104:213–215
Pryke SR, Griffith SC (2008) Postzygotic genetic incompatibility between sympatric color morphs. Evolution 637:93–798
Ramadan HAI, El-Banhawy EM, Hassan AA, Afia SI (2004) Genetic variation in the predacious phytoseiid mite, Amblysieus swirskii (Acari: Phytoseiidae): analysis of specific mitochondrial and nuclear sequences. Arab J Biotechnol 7:189–196
Reis AC, Gondim MGC, Moraes GJ, Hanna R, Schausberger P, Lawson-Balagbo ML, Barros R (2008) Population dynamics of Aceria guerreronis Keifer (Acari: Eriophyidae) and associated predators on coconut fruits in Northeastern Brazil. Neotrop Entomol 37:457–462
Rigaud T, Juchault P, Mocquard JP (1997) The evolution of sex determination in isopod crustaceans. BioEssays 19:409–416
Stouthamer R, Breeuwer JAJ, Hurst GDD (1999) Wolbachia pipientis: microbial manipulator of arthropod reproduction. Annu Rev Microbiol 53:71–102
Stouthamer R, Jochemsen P, Platner GR, Pinto JD (2000) Crossing incompatibility between Trichogramma minutum and T. platneri (Hymenoptera: Trichogrammatidae): implications for applications in biological control. Environ Entomol 29:832–837
Telschow A, Hammerstein P, Werren JH (2005) The effects of Wolbachia versus genetic incompatibilities on reinforcement and speciation. Evolution 59:1607–1619
Telschow A, Flor M, Kobayashi Y, Hammerstein P, Werren JH (2007) Wolbachia-induced unidirectional cytoplasmic incompatibility and speciation: mainland–island model. PLoS One 2:e701. doi:10.1371/journal.pone.0000701
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTALW: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680
Tixier M-S, Kreiter S, Barbar Z, Ragusa S, Cheval B (2006a) The status of two cryptic species: Typhlodromus exhilaratus Ragusa and Typhlodromus phialatus Athias-Henriot (Acari: Phytoseiidae): consequences for taxonomy. Zool Scr 35:115–122
Tixier M-S, Kreiter S, Ferragut F, Cheval B (2006b) The suspected synonymy of Kampimodromus hmiminai and Kampimodromus adrianae (Acari: Phytoseiidae): morphological and molecular investigations. Can J Zool 84:1216–1222
Tixier M-S, Kreiter S, Croft BA, Cheval B (2008) Kampimodromus aberrans (Acari: Phytoseiidae) from the USA: morphological and molecular assessment of its density. Bull Entomol Res 98:125–134
Tixier M-S, Guichou S, Kreiter S (2010) Assessment of the usefulness of eight DNA fragments for phylogenetic studies within the family Phytoseiidae. In: Sabelis MW, Bruin J (eds) Trends in acarology. Proceedings of the 12th International Congress, pp 41–47
Toda S, Osakabe M, Kumazaki S (2000) Interspecific diversity of mitochondrial COI sequence in Japanese Panonychus species (Acari: Tetranychidae). Exp Appl Acarol 24:621–629
Toyoshima S, Nakamura M, Nagahama Y, Amano H (2000) Process of egg formation in the female body cavity and fertilization in male eggs of Phytoseiulus persimilis (Acari: Phytoseiidae). Exp Appl Acarol 24:441–451
Uesugi R, Goka K, Osakabe M (2003) Development of genetic differentiation and postzygotic isolation in experimental metapopulations of spider mites. Exp Appl Acarol 31:161–176
Vala F, Breeuwer JAJ, Sabelis MW (2000) Wolbachia-induced ‘hybrid breakdown’ in the two-spotted spider mite Tetranychus urticae Koch. Proc R Soc Lond Ser B 267:1931–1937
Vavre F, Fleury F, Varaldi J, Fouillet P, Boulétreau M (2002) Infection polymorphism and cytoplasmic incompatibility in Hymenoptera-Wolbachia associations. Heredity 88:361–365
Wade MJ, Steven L (1985) Microorganism-mediated reproductive incompatibility in four beetles (Genus Tribolium). Science 227:527–528
Walsh PS, Metzger DA, Higuchi R (1991) Chelex 100 as medium for simple extraction of DNA for PCR-based ty** for forensic material. Biotechniques 10:506–513
Wu K, Hoy MA (2012) Cardinium is associated with reproductive incompatibility in the predatory mite Metaseiulus occidentalis (Acari: Phytoseiidae). J Invertebr Pathol 110:359–365
Zhu L-Y, Zhang K-J, Zhang Y-J, Ge C, Gotoh T, Hong X-Y (2012) Wolbachia strengthens Cardinium-induced cytoplasmic incompatibility in the spider mite Tetranychus piercei McGregor. Curr Microbiol 65:516–523
Zindel R, Gottlieb Y, Aebi A (2011) Arthropod symbioses: a neglected parameter in pest- and disease-control programmes. J Appl Ecol 48:864–872
Acknowledgments
Many thanks go to M. G. C. Gondim Jr for supplying the specimens of N. paspalivorus from Brazil, and R. Houndafoche, P. Sovimi, C. Kededji and B. Bovis for their assistance in maintaining the predator colonies and conducting crossing experiments. This research was supported by the International Institute of Tropical Agriculture (IITA) and the University of Amsterdam, The Netherlands, through a grant from the Board of the Netherlands Foundation for the Advancement of Tropical Research (WOTRO). This study is part of a PhD thesis of the senior author.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Famah Sourassou, N., Hanna, R., Breeuwer, J.A.J. et al. The endosymbionts Wolbachia and Cardinium and their effects in three populations of the predatory mite Neoseiulus paspalivorus . Exp Appl Acarol 64, 207–221 (2014). https://doi.org/10.1007/s10493-014-9820-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10493-014-9820-0