Abstract
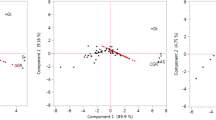
Senegalia macrostachya is a wild, perennial, and promising legume that can help alleviate food insecurity in sub-Saharan Africa’s drought-prone regions. However, it has been for a long time overlooked, underutilized, and scarcely studied. This study shed light on its ecological niche, genetic variation in natural populations, and seeds’ harvest maturity. Three populations of S. macrostachya were selected along an environmental gradient, and their structures, based on their plants’ pod characteristics (i.e., unique morphological differentiation trait amongst the plants), were assessed. Then, the seeds’ development was investigated following their morphological transformation, mass accumulation, and the mobilization of their proteins and secondary metabolites. S. macrostachya has revealed low genetic variation and fitness to poor soils, warm and semi-arid climates in sub-Saharan Africa, prone to droughts and low to medium agricultural productivity. The maturation of the seeds involved the accumulation of specific late embryogenesis abundant proteins (60, 48, and 14 kDa), identified as chilling-responsive dehydrins, and the degradation of the chlorophylls and carotenoids, which could be used as biochemical landmarks of the harvest maturity. Besides, an aging symptom (high seed abortion rate (14–20%)) was observed in the study populations. This work provides unprecedented information that will assist in predicting the eco-climate suitability and climate resilience, identifying the conservation strategies, and planning the harvesting, reforestation programs, and domestication of S. macrostachya. In parallel, the dehydrins identified from S. macrostachya can be tapped into for drought-resistant crop development. Further research is needed to evaluate the impacts of the mother plants’ aging and larger climate gradient on the performance (i.e., seed productivity and regeneration ability) of S. macrostachya.






Similar content being viewed by others
Data availability
All raw data generated during the present study are available from the first and corresponding authors upon request. Besides, the online version contains supplementary material available at https://doi.org/10.1007/s10457-022-00772-5.
References
Aleixandre-Tudo JL, Du Toit W (2018) Chapter 3-The role of UV-visible spectroscopy for phenolic compounds quantification in winemaking. In: Solís-Oviedo RL (ed) Frontiers and New Trends in the Science of Fermented Food and Beverages. IntechOpen, London, pp 1–21. https://doi.org/10.5772/intechopen.79550
Arbonnier M (2000) Arbres, arbustes et lianes des zones sèches d’Afrique de l’Ouest, 1st edn. CIRAD-MNHN-UICN, Paris
Azani N, Babineau M, Bailey CD, Banks H, Barbosa AR, Pinto RB, Boatwright JS, Borges LM et al (2017) A new subfamily classification of the Leguminosae based on a taxonomically comprehensive phylogeny. Taxon 66(1):44–77. https://doi.org/10.12705/661.3
Bance S, Sawadogo P, Ouedraogo L, Yameogo Ganeme S, Balma D, Bognounou G, Ouedraogo L et al (1999) Country study on Burkina Faso biodiversity. Permanent Secretariat of the National Council for the Management of the Environment, Dhaka
Banerjee A, Roychoudhury A (2016) Group II late embryogenesis abundant (LEA) proteins: structural and functional aspects in plant abiotic stress. Plant Growth Regul 79:1–17. https://doi.org/10.1007/s10725-015-0113-3
Batello C, Wade L, Cox S, Pogna N, Bozzini A, Choptiany J (2014) Perennial crops for food security. In: Proceedings of the FAO expert workshop, biodiversity & ecosystem services in agricultural production systems. FAO, Rome, Italy
Blackman SA, Wettlaufer SH, Obendorf RL, Leopold AC (1991) Maturation proteins associated with desiccation tolerance in soybean. Plant Physiol 96:868–874. https://doi.org/10.1104/pp.96.3.868
Bojórquez-Velázquez E, Barrera-Pacheco A, Espitia-Rangel E, Herrera-Estrella A, De La Rosa APB (2019) Protein analysis reveals differential accumulation of late embryogenesis abundant and storage proteins in seeds of wild and cultivated amaranth species. BMC Plant Biol 19:1–17. https://doi.org/10.1186/s12870-019-1656-7
Drabo MS, Shumoy H, Cissé H, Parkouda C, Nikiéma F, Ismail O, Traoré Y, Savadogo A, Raes K (2020) Map** the variability in physical, cooking and nutritional properties of Zamnè, a wild food in Burkina Faso. Food Res Int 138:109810. https://doi.org/10.1016/j.foodres.2020.109810
Drabo MS, Shumoy H, Savadogo A, Raes K (2022) Inventory of human-edible products from native Acacia sensu lato in Africa, America, and Asia: spotlight on Senegalia seeds, overlooked wild legumes in the arid tropics. Food Res Int 159:111596. https://doi.org/10.1016/j.foodres.2022.111596
FAO, SOFI (1999) FAOCLIM database, major environmental constraints for agricultural production project. FAO, Rome
Fernández-Marín B, Milla R, Martín-Robles N, Arc E, Kranner I, Becerril JM, García-Plazaola JI (2014) Side-effects of domestication: cultivated legume seeds contain similar tocopherols and fatty acids but less carotenoids than their wild counterparts. BMC Plant Biol 14:1–11. https://doi.org/10.1186/s12870-014-0385-1
Funk C, Rowland J, Adoum A, Eilerts G, White L (2012) A climate trend analysis of Burkina Faso, famine early warning systems network-informing climate change adaptation series. Fact Sheet 8:2012–3084
Gaisberger H, Kindt R, Loo J, Schmidt M, Bognounou F, Da SS, Diallo OB et al (2017) Spatially explicit multi-threat assessment of food tree species in Burkina Faso: a fine-scale approach. PLoS ONE 12:e0184457. https://doi.org/10.1371/journal.pone.0184457
Global Biodiversity Information Facility (2019) Georeferenciation of Senegalia macrostachya (Rchb. ex DC.) Kyal. & Boatwr. (2013) [WWW Document]. URL www.gbif.org (accessed 12/08/22)
Guffy RD, Hesketh JD, Nelson RL, Bernard RL (1991) Seed growth rate, growth duration and yield in soybean. Biotronics 20:19–30
Harborne JB (1958) Spectral methods of characterizing anthocyanins. Biochem J 70:22–28. https://doi.org/10.1042/bj0700022
Lembicz M, Olejniczak P, Zukowski W, Bogdanowicz AM (2011) Effect of mother plant age on germination and size of seeds and seedlings in the perennial sedge Carex secalina (Cyperaceae). Flora 206:158–163. https://doi.org/10.1016/j.flora.2010.09.006
Leprince O, Pellizzaro A, Berriri S, Buitink J (2017) Late seed maturation: drying without dying. J Exp Bot 68:827–841. https://doi.org/10.1093/jxb/erw363
Lichtenthaler HK, Buschmann C (2001) Unit F4.3—Chlorophylls and carotenoids: measurement and characterization by UV-VIS sprectroscopy. Curr Protoc Food Anal Chem 1(1):1–8. https://doi.org/10.1002/0471142913.faf0403s01
Lv A, Su L, Liu X, **ng Q, Huang B, An Y, Zhou P (2018) Characterization of dehydrin protein, CdDHN4-L and CdDHN4-S and their differential protective roles against abiotic stress in vitro. BMC Plant Biol 18:299. https://doi.org/10.1186/s12870-018-1511-2
Merzlyak MN, Chivkunova OB, Solovchenko AE, Naqvi KR (2008) Light absorption by anthocyanins in juvenile, stressed and senescing leaves. J Exp Bot 59:3903–3911. https://doi.org/10.1093/jxb/ern230
Monma M, Ito M, Saito M, Chikuni K (1994) Carotenoid components in soybean seeds varying with seed color and maturation stage. Biosci Biotechnol Biochem 58:926–930. https://doi.org/10.1271/bbb.58.926
Msika P, Saunois A, Leclere-Bienfait S, Baudoin C (2017) Acacia macrostachya seed extract and compositions containing same. EP 2 506 723 B1
Müller M, Siles L, Cela J, Munné-bosch S (2014) Perennially young : seed production and quality in controlled and natural populations of Cistus albidus reveal compensatory mechanisms that prevent senescence in terms of seed yield and viability. J Exp Bot 65:287–297. https://doi.org/10.1093/jxb/ert372
Ouedraogo LG (1997) Genetische variation und reproduktionssystem in populationen von Acacia macrostachya Reichenbach ex Benth (Mimosaceae) in Burkina Faso. PhD thesis—Univ, Göttingen, pp. 1–105
Phuong NNM, Le TT, Dang MQ, Van Camp J, Raes K (2020) Selection of extraction conditions of phenolic compounds from rambutan (Nephelium lappaceum L.) peel. Food Bioprod Process 122:222–229. https://doi.org/10.1016/j.fbp.2020.05.008
Powell B, Thilsted SH, Ickowitz A, Termote C, Sunderland T, Herforth A (2015) Improving diets with wild and cultivated biodiversity from across the landscape. Food Secur 7:535–554. https://doi.org/10.1007/s12571-015-0466-5
Sanhewe AJ, Ellis RH (1996) Seed development and maturation in Phaseolus vulgaris I. Ability to germinate and to tolerate desiccation. J Exp Bot 47:949–958. https://doi.org/10.1093/jxb/47.7.949
Sawadogo Y, Ganaba S, Tindano E, Some AN (2017) Caractérisation des populations naturelles d’une légumineuse alimentaire sauvage, Senegalia macrostachya (Reichenb. ex DC. Kyal & Boatwr) dans le secteur Nord-soudanien du Burkina Faso. Int J Biol Chem Sci 11:2408–2420. https://doi.org/10.4314/ijbcs.v11i5.36
Schmidt L (2000) Planning and preparation of seed collections. In: Schmidt L (ed) Guide to handling of tropical and subtropical forest seed. Danida Forest Seed Centre, Humlebaek, pp 1–36
Smolikova GN, Medvedev SS (2015) Seed carotenoids: Synthesis, diversity, and functions. Russ J Plant Physiol 62:1–13. https://doi.org/10.1134/S1021443715010136
Smolikova G, Dolgikh E, Vikhnina M, Frolov A, Medvedev S (2017) Genetic and hormonal regulation of chlorophyll degradation during maturation of seeds with green embryos. MDPI Int J Mol Sci 18:5–10. https://doi.org/10.3390/ijms18091993
Souza M, Comin JJ, Moresco R, Maraschin M, Kurtz C, Lovato PE, Lourenzi CR, Pilatti FK, Loss A, Kuhnen S (2021) Exploratory and discriminant analysis of plant phenolic profiles obtained by UV–vis scanning spectroscopy. J Integr Bioinform. https://doi.org/10.1515/jib-2019-0056
USDA (2008) Part 1—Principles and general methods of producing and handling seeds In: Bonner FT, Karrfait RP, Nisley RG (eds) The Woody Plant Seed Manual. USDA, pp. 1–145.
Varaigne-Labeyrie C, Labeyrie V (1981) First data on Bruchidae which attack the pods of legumes in upper Volta of which eight species are man consumed. Ser Entomol 19:83–96
Vassal J (1998) Les acacias au Sénégal-Taxonomie, écologie, principaux intérêts. In: Campas C, Grignon C, Gueye M, Hamon M (eds) L’acacia au Sénégal. Orstom-Isra, Dakar, pp 15–33
Wittig R, Schmidt M, Thiombiano A (2004) Cartes de distribution des espèces du genre Acacia L. au Burkina Faso. Etudes Flore Végétatio Burkina Faso 8:19–26
Yamkoulga M, Waongo A, Sawadogo L, Sanon A (2018) Gestion post-récolte des graines d’Acacia macrostachya Reichenb. ex DC. dans la province du Boulkiemdé au Burkina Faso: diagnostic participatif en milieu paysan. J Appl Biosci 130:13148–13161. https://doi.org/10.4314/jab.v130i1.3
Yamkoulga M, Waongo A, Ilboudo ME, Traoré F, Ilboudo Z, Abudulai M, Sanon A (2021a) Effects of harvesting date on the level of initial infestation and damages caused by insect pests on the seeds of Acacia macrostachya Reichenb. ex DC., in the district of Boulkiemde in Burkina Faso. J Stored Prod Postharvest Res 12:1–7. https://doi.org/10.5897/JSPPR2020.0310
Yamkoulga M, Waongo A, Ilboudo Z, Traoré F, Sanon A (2021b) Effectiveness of hermetic storage using PICS bags and plastic jars for post-harvest preservation of Acacia macrostachya seeds. Adv Entomol 9:20–29. https://doi.org/10.4236/ae.2021.91002
Zida D, Tigabu M, Sawadogo L, Ode PC (2008) Initial seedling morphological characteristics and field performance of two Sudanian savanna species in relation to nursery production period and watering regimes. For Ecol Manage 255:2151–2162. https://doi.org/10.1016/j.foreco.2007.12.029
Acknowledgements
The authors acknowledge Dr. Lambert George Ouedraogo, consultant in natural resources and environment management, for his feedback and the copy of his Ph.D. thesis (see in reference), which has been essential for the design and discussion of this study.
Funding
Moustapha Soungalo Drabo is grateful for Burkina Faso national and Ghent University (BOF 01W05318) Ph.D. scholarships. Habtu Shumoy thanks Ghent University for his post-doc grant (BOF19/PDO/026).
Author information
Authors and Affiliations
Contributions
MSD: Conceptualization, Methodology, Investigation, Formal analysis, Writing—Original Draft; HS: Investigation, Writing—Review & Editing; JK: Resources, Writing—Review & Editing; AS: Resources, Supervision; KR: Methodology, Resources, Supervision, Formal analysis, Writing—Review & Editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Ethics approval
This study did not involve animal subjects or human participants, and no specific permissions were required to sample the fruits and the herbarium specimens. No previous report states Senegalia macrostachya (Rchb. ex DC) Kyal. & Boatwr. (formerly as Acacia macrostachya Rchb. ex DC) is threatened or under protection in any of the sampled locations. The species is listed to have a “least concern” conservation status (https://www.iucnredlist.org/species/144286715/149015544, accessed 12/08/2022).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Drabo, M.S., Shumoy, H., Koala, J. et al. Ecological niche, genetic variation in natural populations, and harvest maturity of Senegalia macrostachya (Rchb. ex DC) Kayl. & Boatwr., a promising wild and perennial edible-seeded crop. Agroforest Syst 97, 1233–1247 (2023). https://doi.org/10.1007/s10457-022-00772-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10457-022-00772-5