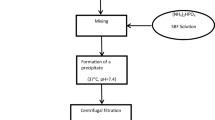
Abstract
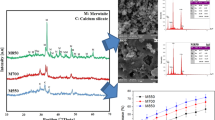
Calcium phosphates are used in biomedical materials, such as bone cement, bone replacement material and drug carriers, as they are the major inorganic constituents of hard human tissue. Different reverse microemulsions were prepared to synthesize brushite materials with diverse shapes and sizes. SEM analysis showed that from ME1 an irregular amorphous material was obtained, those from ME2 formed macroscopic sheets and nanoparticles synthesized from ME3 formed nanorods-like structures. Due to their size and shape, those nanoparticles were used as drug carriers for controlled drug release. The results suggest a direct effect of the type of reverse microemulsion used, and that the amount of water added to the reverse microemulsion may also affect the phase of the final material. An evaluation of adsorption and desorption of the antibiotic norfloxacin was carried out on the nanoparticles obtained from ME3 and demonstrated that the nanoparticles could be used not only as a tissue engineering material but also as a controlled drug delivery system.





Similar content being viewed by others
References
Aguilar-Frutis, M., Kumar, S., Falcony, C.: Spray-pyrolyzed hydroxyapatite thin-film coatings. Surf. Coat. Technol. 204(6–7), 1116–1120 (2009). https://doi.org/10.1016/j.surfcoat.2009.07.021
Alkhraisat, M.H., Rueda, C., Cabrejos-Azama, J., Lucas-Aparicio, J., Mariño, F.T., Torres García-Denche, J., Jerez, L.B., Gbureck, U., Cabarcos, E.L.: Loading and release of doxycycline hyclate from strontium-substituted calcium phosphate cement. Acta Biomater. 6(4), 1522–1528 (2010). https://doi.org/10.1016/j.actbio.2009.10.043
Asami, K.: Dielectric relaxation in a water–oil–Triton X-100 microemulsion near phase inversion. Langmuir 21(20), 9032–9037 (2005). https://doi.org/10.1021/la050235u
Bohner, M., Tadier, S., van Garderen, N., de Gasparo, A., Döbelin, N., Baroud, G.: Synthesis of spherical calcium phosphate particles for dental and orthopedic applications. Biomatter 3(2), e25103 (2013). https://doi.org/10.4161/biom.25103
Cai, Y., Liu, Y., Yan, W., Hu, Q., Tao, J., Zhang, M., Shi, Z., Tang, R.: Role of hydroxyapatite nanoparticle size in bone cell proliferation. J. Mater. Chem. 17(36), 3780–3787 (2007). https://doi.org/10.1039/b705129h
Chen, J., Wang, Y., Chen, X., Ren, L., Lai, C., He, W., Zhang, Q.: A simple sol–gel technique for synthesis of nanostructured hydroxyapatite, tricalcium phosphate and biphasic powders. Mater. Lett. 65(12), 1923–1926 (2011). https://doi.org/10.1016/j.matlet.2011.03.076
Chen, X., Zhao, Z., Chen, A., Li, H.: Pulsed electrodeposition of hydroxyapatite on titanium substrate in solution containing hydrogen peroxide. Trans. Nonferr, Met. Soc. China 17(3), 617–621 (2007). https://doi.org/10.1016/S1003-6326(07)60144-7
Cho, J.S., Lee, J.-C., Rhee, S.-H.: Effect of precursor concentration and spray pyrolysis temperature upon hydroxyapatite particle size and density. J. Biomed. Mater. Res. B 104(2), 422–430 (2016). https://doi.org/10.1002/jbm.b.33406
Costa, P., Sousa Lobo, J.M.: Evaluation of mathematical models describing drug release from estradiol transdermal systems. Drug Dev. Ind. Pharm. 29(1), 89–97 (2003). https://doi.org/10.1081/ddc-120016687
Dai, X., Shivkumar, S.: Electrospinning of PVA-calcium phosphate sol precursors for the production of fibrous hydroxyapatite. J. Am. Ceram. Soc. 90(5), 1412–1419 (2007). https://doi.org/10.1111/j.1551-2916.2007.01569.x
Dorozhkin, S.: Calcium orthophosphate cements and concretes. Materials 2(1), 221 (2009a)
Dorozhkin, S.: Nanodimensional and nanocrystalline apatites and other calcium orthophosphates in biomedical engineering, biology and medicine. Materials 2(4), 1975 (2009b)
Dosen, A., Giese, R.F.: Thermal decomposition of brushite, CaHPO4·2H2O to monetite CaHPO4 and the formation of an amorphous phase. Am. Mineral. 96(2–3), 368–373 (2011). https://doi.org/10.2138/am.2011.3544
Durdu, S., Deniz, Ö.F., Kutbay, I., Usta, M.: Characterization and formation of hydroxyapatite on Ti6Al4V coated by plasma electrolytic oxidation. J. Alloys Compd 551, 422–429 (2013). https://doi.org/10.1016/j.jallcom.2012.11.024
Feng, W., Mu-sen, L., Yu-peng, L., Yong-xin, Q.: A simple sol–gel technique for preparing hydroxyapatite nanopowders. Mater. Lett. 59(8–9), 916–919 (2005). https://doi.org/10.1016/j.matlet.2004.08.041
Gbureck, U., Vorndran, E., Barralet, J.E.: Modeling vancomycin release kinetics from microporous calcium phosphate ceramics comparing static and dynamic immersion conditions. Acta Biomater. 4(5), 1480–1486 (2008). https://doi.org/10.1016/j.actbio.2008.02.027
Giger, E.V., Puigmartí-Luis, J., Schlatter, R., Castagner, B., Dittrich, P.S., Leroux, J.-C.: Gene delivery with bisphosphonate-stabilized calcium phosphate nanoparticles. J. Control. Release 150(1), 87–93 (2011). https://doi.org/10.1016/j.jconrel.2010.11.012
Ginebra, M.-P., Canal, C., Espanol, M., Pastorino, D., Montufar, E.B.: Calcium phosphate cements as drug delivery materials. Adv. Drug Deliv. Rev. 64(12), 1090–1110 (2012). https://doi.org/10.1016/j.addr.2012.01.008
Giocondi, J.L., El-Dasher, B.S., Nancollas, G.H., Orme, C.A.: Molecular mechanisms of crystallization impacting calcium phosphate cements. Philos. Trans. R. Soc. Lond. A 368(1917), 1937–1961 (2010). https://doi.org/10.1098/rsta.2010.0006
Grabielle-Madelmont, C., Lesieur, S., Ollivon, M.: Characterization of loaded liposomes by size exclusion chromatography. J. Biochem. Biophys. Methods 56(1–3), 189–217 (2003). https://doi.org/10.1016/S0165-022X(03)00059-9
Guo, X., Yan, H., Zhao, S., Li, Z., Li, Y., Liang, X.: Effect of calcining temperature on particle size of hydroxyapatite synthesized by solid-state reaction at room temperature. Adv. Powder Technol. 24(6), 1034–1038 (2013). https://doi.org/10.1016/j.apt.2013.03.002
Guo, Y.-P., Guo, L.-H., Yao, Y., Ning, C.-Q., Guo, Y.-J.: Magnetic mesoporous carbonated hydroxyapatite microspheres with hierarchical nanostructure for drug delivery systems. Chem. Commun. (Camb. UK) 47(44), 12215–12217 (2011). https://doi.org/10.1039/c1cc15190h
Guo, Y.-P., Yao, Y.-B., Guo, Y.-J., Ning, C.-Q.: Hydrothermal fabrication of mesoporous carbonated hydroxyapatite microspheres for a drug delivery system. Microporous Mesoporous Mater. 155, 245–251 (2012). https://doi.org/10.1016/j.micromeso.2012.01.037
Hilbrig, F., Freitag, R.: Isolation and purification of recombinant proteins, antibodies and plasmid DNA with hydroxyapatite chromatography. Biotechnol. J. 7(1), 90–102 (2012). https://doi.org/10.1002/biot.201100015
Holzmann, D., Holzinger, D., Hesser, G., Schmidt, T., Knor, G.: Hydroxyapatite nanoparticles as novel low-refractive index additives for the long-term UV-photoprotection of transparent composite materials. J. Mater. Chem. 19(43), 8102–8106 (2009). https://doi.org/10.1039/b912116a
Hou, Z., Li, G., Lian, H., Lin, J.: One-dimensional luminescent materials derived from the electrospinning process: preparation, characteristics and application. J. Mater. Chem. 22(12), 5254–5276 (2012). https://doi.org/10.1039/c2jm15638e
Hulshoff, J.E.G., Hayakawa, T., van Dijk, K., Leijdekkers-Govers, A.F.M., van der Waerden, J.P.C.M., Jansen, J.A.: Mechanical and histologic evaluation of Ca–P plasma-spray and magnetron sputter-coated implants in trabecular bone of the goat. J. Biomed. Mater. Res. 36(1), 75–83 (1997). https://doi.org/10.1002/(sici)1097-4636(199707)36:1%3c75:aid-jbm9%3e3.0.co;2-i
Kandori, K., Kuroda, T., Togashi, S., Katayama, E.: Preparation of calcium hydroxyapatite nanoparticles using microreactor and their characteristics of protein adsorption. J. Phys. Chem. B 115(4), 653–659 (2011). https://doi.org/10.1021/jp110441e
Kandori, K., Mizumoto, S., Toshima, S., Fukusumi, M., Morisada, Y.: Effects of heat treatment of calcium hydroxyapatite particles on the protein adsorption behavior. J. Phys. Chem. B 113(31), 11016–11022 (2009). https://doi.org/10.1021/jp904481z
Kar, A., Raja, K.S., Misra, M.: Electrodeposition of hydroxyapatite onto nanotubular TiO2 for implant applications. Surf. Coat. Technol. 201(6), 3723–3731 (2006). https://doi.org/10.1016/j.surfcoat.2006.09.008
Koutsopoulos, S.: Synthesis and characterization of hydroxyapatite crystals: a review study on the analytical methods. J. Biomed. Mater. Res. A 62(4), 600–612 (2002). https://doi.org/10.1002/jbm.10280
Kozlova, D., Chernousova, S., Knuschke, T., Buer, J., Westendorf, A.M., Epple, M.: Cell targeting by antibody-functionalized calcium phosphate nanoparticles. J. Mater. Chem. 22(2), 396–404 (2012). https://doi.org/10.1039/c1jm14683a
Kumar, S., Simpson, D., Smart, R.S.C.: Plasma processing for inducing bioactivity in stainless steel orthopaedic screws. Surf. Coat. Technol. 202(4–7), 1242–1246 (2007). https://doi.org/10.1016/j.surfcoat.2007.07.075
Lai, C., Tang, S., Wang, Y., Wei, K.: Formation of calcium phosphate nanoparticles in reverse microemulsions. Mater. Lett. 59(2), 210–214 (2005). https://doi.org/10.1016/j.matlet.2004.08.037
Landín, M., Martínez-Pacheco, R., Gómez-Amoza, J.L., Souto, C., Concheiro, A., Rowe, R.C.: The effect of country of origin on the properties of dicalcium phosphate dihydrate powder. Int. J. Pharm. 103(1), 9–18 (1994). https://doi.org/10.1016/0378-5173(94)90198-8
Layrolle, P., Ito, A., Tateishi, T.: Sol–gel synthesis of amorphous calcium phosphate and sintering into microporous hydroxyapatite bioceramics. J. Am. Ceram. Soc. 81(6), 1421–1428 (1998). https://doi.org/10.1111/j.1151-2916.1998.tb02499.x
Li, J., Yang, Y., Huang, L.: Calcium phosphate nanoparticles with an asymmetric lipid bilayer coating for siRNA delivery to the tumor. J. Control. Release 158(1), 108–114 (2012). https://doi.org/10.1016/j.jconrel.2011.10.020
Lin, K., Pan, J., Chen, Y., Cheng, R., Xu, X.: Study the adsorption of phenol from aqueous solution on hydroxyapatite nanopowders. J. Hazard. Mater. 161(1), 231–240 (2009). https://doi.org/10.1016/j.jhazmat.2008.03.076
Lin, K., Wu, C., Chang, J.: Advances in synthesis of calcium phosphate crystals with controlled size and shape. Acta Biomater. 10(10), 4071–4102 (2014). https://doi.org/10.1016/j.actbio.2014.06.017
Liu, D., Savino, K., Yates, M.Z.: Microstructural engineering of hydroxyapatite membranes to enhance proton conductivity. Adv. Funct. Mater. 19(24), 3941–3947 (2009). https://doi.org/10.1002/adfm.200900318
Loo, S.C.J., Siew, Y.E., Ho, S., Boey, F.Y.C., Ma, J.: Synthesis and hydrothermal treatment of nanostructured hydroxyapatite of controllable sizes. J. Mater. Sci. Mater. Med. 19(3), 1389–1397 (2008). https://doi.org/10.1007/s10856-007-3261-9
Malik, M.A., Wani, M.Y., Hashim, M.A.: Microemulsion method: a novel route to synthesize organic and inorganic nanomaterials. Arab. J. Chem. 5(4), 397–417 (2012). https://doi.org/10.1016/j.arabjc.2010.09.027
Manso, M., Jiménez, C., Morant, C., Herrero, P., Martínez-Duart, J.M.: Electrodeposition of hydroxyapatite coatings in basic conditions. Biomaterials 21(17), 1755–1761 (2000). https://doi.org/10.1016/S0142-9612(00)00061-2
Metoki, N., Mandler, D., Eliaz, N.: Effect of decorating titanium with different self-assembled monolayers on the electrodeposition of calcium phosphate. Cryst. Growth Des. 16(5), 2756–2764 (2016). https://doi.org/10.1021/acs.cgd.6b00057
Mizutani, Y., Hattori, M., Okuyama, M., Kasuga, T., Nogami, M.: Large-sized hydroxyapatite whiskers derived from calcium tripolyphosphate gel. J. Eur. Ceram. Soc. 25(13), 3181–3185 (2005). https://doi.org/10.1016/j.jeurceramsoc.2004.07.028
Morrison, C.J., Gagnon, P., Cramer, S.M.: Purification of monomeric mAb from associated aggregates using selective desorption chromatography in hydroxyapatite systems. Biotechnol. Bioeng. 108(4), 813–821 (2011). https://doi.org/10.1002/bit.22971
Moulik, S.P., Das, M.L., Bhattacharya, P.K., Das, A.R.: Thermodynamics of microemulsion formation. 1. Enthalpy of solution of water in binary (Triton X 100 + butanol) and ternary (heptane + Triton X 100 + butanol) mixtures and heat capacity of the resulting systems. Langmuir 8(9), 2135–2139 (1992). https://doi.org/10.1021/la00045a012
Moulik, S.P., Paul, B.K.: Structure, dynamics and transport properties of microemulsions. Adv. Colloid Interface Sci. 78(2), 99–195 (1998). https://doi.org/10.1016/S0001-8686(98)00063-3
Mouthuy, P.-A., Crossley, A., Ye, H.: Fabrication of calcium phosphate fibres through electrospinning and sintering of hydroxyapatite nanoparticles. Mater. Lett. 106, 145–150 (2013). https://doi.org/10.1016/j.matlet.2013.04.110
Niu, N., Wang, D., Huang, S., Li, C., He, F., Gai, S., Li, X., Yang, P.: Controlled synthesis of luminescent F-substituted strontium hydroxyapatite with hierarchical structures for drug delivery. CrystEngComm 14(5), 1744–1752 (2012). https://doi.org/10.1039/c1ce06265d
Ohta, K., Monma, H., Takahashi, S.: Adsorption characteristics of proteins on calcium phosphates using liquid chromatography. J. Biomed. Mater. Res. A 55(3), 409–414 (2001). https://doi.org/10.1002/1097-4636(20010605)55:3%3c409:aid-jbm1030%3e3.0.co;2-z
Oliva, J., Cama, J., Cortina, J.L., Ayora, C., De Pablo, J.: Biogenic hydroxyapatite (Apatite II™) dissolution kinetics and metal removal from acid mine drainage. J. Hazard. Mater. 213–214, 7–18 (2012). https://doi.org/10.1016/j.jhazmat.2012.01.027
Pang, X., Zhitomirsky, I.: Electrodeposition of hydroxyapatite–silver–chitosan nanocomposite coatings. Surf. Coat. Technol. 202(16), 3815–3821 (2008). https://doi.org/10.1016/j.surfcoat.2008.01.022
Parent, M., Baradari, H., Champion, E., Damia, C., Viana-Trecant, M.: Design of calcium phosphate ceramics for drug delivery applications in bone diseases: a review of the parameters affecting the loading and release of the therapeutic substance. J. Control. Release 252, 1–17 (2017). https://doi.org/10.1016/j.jconrel.2017.02.012
Putz, H., Brandenburg, K.: MATCH! Phase Identification from Powder Diffraction. Crystal Impact, Bonn (2003)
Ramirez-Gutierrez, C.F., Palechor-Ocampo, A.F., Londoño-Restrepo, S.M., Millán-Malo, B.M., Rodriguez-García, M.E.: Cooling rate effects on thermal, structural, and microstructural properties of bio-hydroxyapatite obtained from bovine bone. J. Biomed. Mater. Res. B 104(2), 339–344 (2016). https://doi.org/10.1002/jbm.b.33401
Ren, F., Ding, Y., Ge, X., Lu, X., Wang, K., Leng, Y.: Growth of one-dimensional single-crystalline hydroxyapatite nanorods. J. Cryst. Growth 349(1), 75–82 (2012). https://doi.org/10.1016/j.jcrysgro.2012.04.003
Ren, F., Leng, Y., Ding, Y., Wang, K.: Hydrothermal growth of biomimetic carbonated apatite nanoparticles with tunable size, morphology and ultrastructure. CrystEngComm 15(11), 2137–2146 (2013). https://doi.org/10.1039/c3ce26884e
Rong, D., Chen, P., Yang, Y., Li, Q., Wan, W., Fang, X., Zhang, J., Han, Z., Tian, J., Ouyang, J.: Fabrication of gelatin/PCL electrospun fiber mat with bone powder and the study of its biocompatibility. J. Funct. Biomater. 7(1), 6 (2016). https://doi.org/10.3390/jfb7010006
Sahoo, S., Chakraborti, C.K., Behera, P.K., Mishra, S.C.: FTIR and Raman spectroscopic investigations of a norfloxacin/Carbopol934 polymeric suspension. J. Young Pharm. 4(3), 138–145 (2012). https://doi.org/10.4103/0975-1483.100017
Sanosh, K.P., Chu, M.-C., Balakrishnan, A., Kim, T.N., Cho, S.-J.: Sol–gel synthesis of pure nano sized β-tricalcium phosphate crystalline powders. Curr. Appl. Phys. 10(1), 68–71 (2010). https://doi.org/10.1016/j.cap.2009.04.014
Shao, F., Liu, L., Fan, K., Cai, Y., Yao, J.: Ibuprofen loaded porous calcium phosphate nanospheres for skeletal drug delivery system. J. Mater. Sci. 47(2), 1054–1058 (2012). https://doi.org/10.1007/s10853-011-5894-9
Singh, S., Bhardwaj, P., Singh, V., Aggarwal, S., Mandal, U.K.: Synthesis of nanocrystalline calcium phosphate in microemulsion—effect of nature of surfactants. J. Colloid Interface Sci. 319(1), 322–329 (2008). https://doi.org/10.1016/j.jcis.2007.09.059
Strey, R.: Microemulsion microstructure and interfacial curvature. Colloid Polym. Sci. 272(8), 1005–1019 (1994). https://doi.org/10.1007/bf00658900
Sun, H., Su, F.-Z., Ni, J., Cao, Y., He, H.-Y., Fan, K.-N.: Gold supported on hydroxyapatite as a versatile multifunctional catalyst for the direct tandem synthesis of imines and oximes. Angew. Chem. Int. Ed. 48(24), 4390–4393 (2009). https://doi.org/10.1002/anie.200900802
Sun, Y., Guo, G., Wang, Z., Guo, H.: Synthesis of single-crystal HAP nanorods. Ceram. Int. 32(8), 951–954 (2006). https://doi.org/10.1016/j.ceramint.2005.07.023
Tamimi, F., Torres, J., Kathan, C., Baca, R., Clemente, C., Blanco, L., Cabarcos, E.L.: Bone regeneration in rabbit calvaria with novel monetite granules. J. Biomed. Mater. Res. 87A(4), 980–985 (2008). https://doi.org/10.1002/jbm.a.31842
Tian, Z., Huang, W., Liang, Y.: Preparation of spherical nanoparticles of LaAlO3 via the reverse microemulsion process. Ceram. Int. 35(2), 661–664 (2009). https://doi.org/10.1016/j.ceramint.2008.01.020
Tsui, Y.C., Doyle, C., Clyne, T.W.: Plasma sprayed hydroxyapatite coatings on titanium substrates. Part 1: mechanical properties and residual stress levels. Biomaterials 19(22), 2015–2029 (1998). https://doi.org/10.1016/S0142-9612(98)00103-3
Tucker, B.E., Cottell, C.M., Auyeungt, R.C.Y., Spector, M., Nancollas, G.H.: Pre-conditioning and dual constant composition dissolution kinetics of pulsed laser deposited hydroxyapatite thin films on silicon substrates. Biomaterials 17(6), 631–637 (1996). https://doi.org/10.1016/0142-9612(96)88714-X
Uskoković, V., Uskoković, D.P.: Nanosized hydroxyapatite and other calcium phosphates: chemistry of formation and application as drug and gene delivery agents. J. Biomed. Mater. Res. B 96B(1), 152–191 (2011). https://doi.org/10.1002/jbm.b.31746
Vallet-Regí, M., González-Calbet, J.M.: Calcium phosphates as substitution of bone tissues. Prog. Solid State Chem. 32(1–2), 1–31 (2004). https://doi.org/10.1016/j.progsolidstchem.2004.07.001
Vukomanovic, M., Zunic, V., Otonicar, M., Repnik, U., Turk, B., Skapin, S.D., Suvorov, D.: Hydroxyapatite/platinum bio-photocatalyst: a biomaterial approach to self-cleaning. J. Mater. Chem. 22(21), 10571–10580 (2012). https://doi.org/10.1039/c2jm00136e
Wang, J., Chong, P.F., Ng, S.C., Gan, L.M.: Microemulsion processing of manganese zinc ferrites. Mater. Lett. 30(2), 217–221 (1997). https://doi.org/10.1016/S0167-577X(96)00200-5
Wei, W., Zhang, X., Cui, J., Wei, Z.: Interaction between low molecular weight organic acids and hydroxyapatite with different degrees of crystallinity. Colloids Surf. A 392(1), 67–75 (2011). https://doi.org/10.1016/j.colsurfa.2011.09.034
Wei, X., Yates, M.Z.: Yttrium-doped hydroxyapatite membranes with high proton conductivity. Chem. Mater. 24(10), 1738–1743 (2012). https://doi.org/10.1021/cm203355h
Welzel, T., Meyer-Zaika, W., Epple, M.: Continuous preparation of functionalised calcium phosphate nanoparticles with adjustable crystallinity. Chem. Commun. (Camb. UK) (2004). https://doi.org/10.1039/b402521k
Wu, L., Dou, Y., Lin, K., Zhai, W., Cui, W., Chang, J.: Hierarchically structured nanocrystalline hydroxyapatite assembled hollow fibers as a promising protein delivery system. Chem. Commun. (Camb. UK) 47(42), 11674–11676 (2011). https://doi.org/10.1039/c1cc14709a
**a, L., Lin, K., Jiang, X., Xu, Y., Zhang, M., Chang, J., Zhang, Z.: Enhanced osteogenesis through nano-structured surface design of macroporous hydroxyapatite bioceramic scaffolds via activation of ERK and p38 MAPK signaling pathways. J. Mater. Chem. B 1(40), 5403–5416 (2013). https://doi.org/10.1039/c3tb20945h
Xu, J., White, T., Li, P., He, C., Han, Y.-F.: Hydroxyapatite foam as a catalyst for formaldehyde combustion at room temperature. J. Am. Chem. Soc. 132(38), 13172–13173 (2010). https://doi.org/10.1021/ja1058923
Ye, F., Guo, H., Zhang, H., He, X.: Polymeric micelle-templated synthesis of hydroxyapatite hollow nanoparticles for a drug delivery system. Acta Biomater. 6(6), 2212–2218 (2010). https://doi.org/10.1016/j.actbio.2009.12.014
Ye, G., Troczynski, T.: Hydroxyapatite coatings by pulsed ultrasonic spray pyrolysis. Ceram. Int. 34(3), 511–516 (2008). https://doi.org/10.1016/j.ceramint.2006.11.014
Younes, E., Sanjiv, P., Santiago, G.-R.: A short overview on the biomedical applications of silica, alumina and calcium phosphate-based nanostructured materials. Curr. Med. Chem. 23(39), 4450–4467 (2016). https://doi.org/10.2174/0929867323666161024153459
Yu, W., Sun, T.-W., Qi, C., Ding, Z., Zhao, H., Chen, F., Chen, D., Zhu, Y.-J., Shi, Z., He, Y.: Strontium-doped amorphous calcium phosphate porous microspheres synthesized through a microwave-hydrothermal method using fructose 1,6-bisphosphate as an organic phosphorus source: application in drug delivery and enhanced bone regeneration. ACS Appl. Mater. Interfaces 9(4), 3306–3317 (2017). https://doi.org/10.1021/acsami.6b12325
Zhang, C., Li, C., Huang, S., Hou, Z., Cheng, Z., Yang, P., Peng, C., Lin, J.: Self-activated luminescent and mesoporous strontium hydroxyapatite nanorods for drug delivery. Biomaterials 31(12), 3374–3383 (2010a). https://doi.org/10.1016/j.biomaterials.2010.01.044
Zhang, Y., Huo, M., Zhou, J., Zou, A., Li, W., Yao, C., **e, S.: DDSolver: an add-in program for modeling and comparison of drug dissolution profiles. AAPS J. 12(3), 263–271 (2010b). https://doi.org/10.1208/s12248-010-9185-1
Zhang, Y., Liu, Y., Ji, X., Banks, C.E., Zhang, W.: Conversion of egg-shell to hydroxyapatite for highly sensitive detection of endocrine disruptor bisphenol A. J. Mater. Chem. 21(38), 14428–14431 (2011). https://doi.org/10.1039/c1jm12544c
Zhao, B., Hu, H., Mandal, S.K., Haddon, R.C.: A bone mimic based on the self-assembly of hydroxyapatite on chemically functionalized single-walled carbon nanotubes. Chem. Mater. 17(12), 3235–3241 (2005). https://doi.org/10.1021/cm0500399
Acknowledgements
Financial support from National Council of Science and Technology of Mexico (CONACyT, INNOVAPYME 231008) is greatly appreciated. The authors acknowledge to Josué Romero-Ibarra, for acquiring SEM images, I. D. R.-M. and L. V. H.-H are thankful to DGAPA-UNAM for a Postdoctoral Fellowship.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest. All authors are responsible for the content and the writing of the article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rojas-Montoya, I.D., Fosado-Esquivel, P., Henao-Holguín, L.V. et al. Adsorption/desorption studies of norfloxacin on brushite nanoparticles from reverse microemulsions. Adsorption 26, 825–834 (2020). https://doi.org/10.1007/s10450-019-00138-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10450-019-00138-x