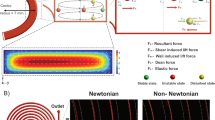
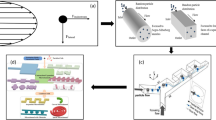
Abstract
The inertial-based spiral microfluidic separation device is the most sought after for a number of medical settings, because of its simplicity, high throughput, and rejection from an external source at a lower cost than other types of devices. Spiral microchannels have shown an optimistic outcome for biological cell manipulations as the hydrodynamic particle–particle interactions in these devices are known to have a significant impact on focusing behavior. There have been a plethora of attempts to decipher the forces and migration principle of particles in these intricate microchannels, but the influence of cross-sectional geometries and its fine control over the dean vortices on effective particle manipulation in optimum microfluidic design is yet to be discussed in detail to the tee. This is the first time to compare dean flow mechanism thereby providing deep insights on distinct cross-sectional areas (rectangular, trapezoidal, hybrid/complex and stair-like) of spiral geometry, biocompatibility and its fabrication methods for effective cell focusing and separation. This study insists on the influence of cross-sectional geometry in spiral separation devices to widen the horizons of the existing applications even though the fabrication of the same is still challenging. The advent of 3D printing and other similar technologies seems promising to address the fabrication issues in the near future. This review enables the researchers to focus on intricate cross sections to tune the dean flow for improving the separation performance which paves the way to a slew of new applications in clinical diagnostics and other research areas in the biomedical field.







Similar content being viewed by others
References
Abdulla A, Liu W, Gholamipour-Shirazi A et al (2018) High-Throughput Isolation of Circulating Tumor Cells Using Cascaded Inertial Focusing Microfluidic Channel. Anal Chem 90:4397–4405. https://doi.org/10.1021/acs.analchem.7b04210
Alba-Loureiro TC, Munhoz CD, Martins JO et al (2007) Neutrophil function and metabolism in individuals with diabetes mellitus. Brazilian J Med Biol Res. https://doi.org/10.1590/S0100-879X2006005000143
Al-Halhouli A, Al-Faqheri W, Alhamarneh B et al (2018) Spiral microchannels with trapezoidal cross section fabricated by femtosecond laser ablation in glass for the inertial separation of microparticles. Micromachines. https://doi.org/10.3390/mi9040171
Asmolov ES (1999) The inertial lift on a spherical particle in a plane poiseuille flow at large channel Reynolds number. J Fluid Mech. https://doi.org/10.1017/S0022112098003474
Aya-Bonilla CA, Marsavela G, Freeman JB et al (2017) Isolation and detection of circulating tumour cells from metastatic melanoma patients using a slanted spiral microfluidic device. Oncotarget. https://doi.org/10.18632/oncotarget.18641
Bagdade JD, Root RK, Bulger RJ (1974) Impaired leukocyte function in patients with poorly controlled diabetes. Diabetes. https://doi.org/10.2337/diab.23.1.9
Balasubramaniam L, Arayanarakool R, Marshall SD et al (2017) Impact of cross-sectional geometry on mixing performance of spiral microfluidic channels characterized by swirling strength of Dean-vortices. J Micromech Microeng. https://doi.org/10.1088/1361-6439/aa7fc8
Bayareh M (2020) An updated review on particle separation in passive microfluidic devices. Chem Eng Process Process Intensif 153. https://doi.org/10.1016/j.cep.2020.107984
Berezovski M, Drabovich A, Krylova SM et al (2005) Nonequilibrium capillary electrophoresis of equilibrium mixtures: a universal tool for development of aptamers. J Am Chem Soc. https://doi.org/10.1021/ja042394q
Bhagat AAS, Kuntaegowdanahalli SS, Papautsky I (2008) Continuous particle separation in spiral microchannels using dean flows and differential migration. Lab Chip 8:1906–1914. https://doi.org/10.1039/b807107a
Bhagat AAS, Bow H, Hou HW et al (2010a) Microfluidics for cell separation. Med Biol Eng Comput 48:999–1014. https://doi.org/10.1007/s11517-010-0611-4
Bhagat AAS, Kuntaegowdanahalli SS, Kaval N et al (2010b) Inertial microfluidics for sheath-less high-throughput flow cytometry. Biomed Microdevices. https://doi.org/10.1007/s10544-009-9374-9
Bhagat AAS, Kuntaegowdanahalli SS, Papautsky I (2008) Enhanced particle filtration in straight microchannels using shear-modulated inertial migration. In: Physics of fluids. https://doi.org/10.1063/1.2998844
Bhattacharjee N, Parra-Cabrera C, Kim YT et al (2018) Desktop-Stereolithography 3D-printing of a poly(dimethylsiloxane)-based material with sylgard-184 properties. Adv Mater. https://doi.org/10.1002/adma.201800001
Bhattacharjee N, Urrios A, Kang S, Folch A (2016) The upcoming 3D-printing revolution in microfluidics. Lab Chip 16. https://doi.org/10.1039/c6lc00163g
Birch CM, Hou HW, Han J, Niles JC (2015) Identification of malaria parasite-infected red blood cell surface aptamers by inertial microfluidic SELEX (I-SELEX). Sci Rep. https://doi.org/10.1038/srep11347
Bohl B, Steger R, Zengerle R, Koltay P (2005) Multi-layer SU-8 lift-off technology for microfluidic devices. J Micromechanics Microengineering. https://doi.org/10.1088/0960-1317/15/6/002
Brahim WME, Khoo BLUA, Wu L et al (2016) Ultra-fast, label-free isolation of circulating tumor cells from blood using spiral microfluidics. Nat Protoc 11:134–148. https://doi.org/10.1038/nprot.2016.003
Bridle H, Kersaudy-Kerhoas M, Miller B, et al (2012) Detection of Cryptosporidium in miniaturised fluidic devices. Water Res 46. https://doi.org/10.1016/j.watres.2012.01.010
Burke JM, Zubajlo RE, Smela E, White IM (2014) High-throughput particle separation and concentration using spiral inertial filtration. Biomicrofluidics. https://doi.org/10.1063/1.4870399
Caffiyar MY, Lim KP, Kamal Basha IH et al (2020) Label-free, high-throughput assay of human dendritic cells from whole-blood samples with microfluidic inertial separation suitable for resource-limited manufacturing. Micromachines. https://doi.org/10.3390/mi11050514
Catarino SO, Rodrigues RO, Pinho D, et al (2019) Blood cells separation and sorting techniques of passive microfluidic devices: from fabrication to applications. Micromachines. https://doi.org/10.3390/mi10090593
Çetin B, Li D (2011) Dielectrophoresis in microfluidics technology. Electrophoresis 32:2410–2427. https://doi.org/10.1002/elps.201100167
Chen H (2018) A triplet parallelizing spiral microfluidic chip for continuous separation of tumor cells. Sci Rep 8:1–8. https://doi.org/10.1038/s41598-018-22348-z
Cheung YW, Dirkzwager RM, Wong WC et al (2018) Aptamer-mediated plasmodium-specific diagnosis of malaria. Biochimie. https://doi.org/10.1016/j.biochi.2017.10.017
Chiu PL, Chang CH, Lin YL et al (2019) Rapid and safe isolation of human peripheral blood B and T lymphocytes through spiral microfluidic channels. Sci Rep. https://doi.org/10.1038/s41598-019-44677-3
Choi S, Song S, Choi C, Park JK (2007) Continuous blood cell separation by hydrophoretic filtration. Lab Chip 7:1532–1538. https://doi.org/10.1039/b705203k
Choi S, Song S, Choi C, Park JK (2008) Sheathless focusing of microbeads and blood cells based on hydrophoresis. Small 4:634–641. https://doi.org/10.1002/smll.200700308
Chung AJ (2019) A minireview on inertial microfluidics fundamentals: inertial particle focusing and secondary flow. Biochip J 13:53–63. https://doi.org/10.1016/j.watres.2012.01.010
Clime L, Li K, Geissler M et al (2017) Separation and concentration of Phytophthora ramorum sporangia by inertial focusing in curving microfluidic flows. Microfluid Nanofluid. https://doi.org/10.1007/s10404-016-1844-9
Condina MR, Dilmetz BA, RazaviBazaz S et al (2019) Rapid separation and identification of beer spoilage bacteria by inertial microfluidics and MALDI-TOF mass spectrometry. Lab Chip 19:1961–1970. https://doi.org/10.1039/c9lc00152b
Dean WR (1928) LXXII. The stream-line motion of fluid in a curved pipe (second paper). Lond Edinb Dub Philos Mag J Sci. https://doi.org/10.1080/14786440408564513
Delamaire M, Maugendre D, Moreno M et al (1997) Impaired leucocyte functions in diabetic patients. Diabet Med. https://doi.org/10.1002/(SICI)1096-9136(199701)14:1%3c29::AID-DIA300%3e3.0.CO;2-V
Di Carlo D (2009) Inertial microfluidics. Lab Chip 9:3038–3046. https://doi.org/10.1039/b912547g
Di Carlo D, Edd JF, Humphry KJ et al (2009) Particle segregation and dynamics in confined flows. Phys Rev Lett. https://doi.org/10.1103/PhysRevLett.102.094503
Do QV, Van DA, Nguyen VB, Pham VS (2020a) A numerical modeling study on inertial focusing of microparticle in spiral microchannel. AIP Adv. https://doi.org/10.1063/5.0006975
Do QV, Van DA, Nguyen VB, Pham VS (2020b) Erratum: a numerical modeling study on inertial focusing of microparticle in spiral microchannel. AIP Adv 10(1063/5):0006975. https://doi.org/10.1063/5.0025966
Dong Y, Skelley AM, Merdek KD, et al (2013) Microfluidics and circulating tumor cells. J. Mol. Diagnostics 15. https://doi.org/10.1016/j.jmoldx.2012.09.004
Ellington AD, Szostak JW (1990) In vitro selection of RNA molecules that bind specific ligands. Nature. https://doi.org/10.1038/346818a0
Enger J, Goksör M, Ramser K et al (2004) Optical tweezers applied to a microfluidic system. Lab Chip 4:196–200. https://doi.org/10.1039/b307960k
Fallahi H, Zhang J, Phan HP, Nguyen NT (2019) Flexible microfluidics: fundamentals, recent developments, and applications. Micromachines. https://doi.org/10.3390/mi10120830
Forbes TP, Forry SP (2012) Microfluidic magnetophoretic separations of immunomagnetically labeled rare mammalian cells. Lab Chip 12:1471–1479. https://doi.org/10.1039/c2lc40113d
Fuchs BB, Eatemadpour S, Martel-Foley JM et al (2019) Rapid isolation and concentration of pathogenic fungi using inertial focusing on a chip-based platform. Front Cell Infect Microbiol 9:1–9. https://doi.org/10.3389/fcimb.2019.00027
Ghadami S, Kowsari-Esfahan R, Saidi MS, Firoozbakhsh K (2017) Spiral microchannel with stair-like cross section for size-based particle separation. Microfluid Nanofluidics. https://doi.org/10.1007/s10404-017-1950-3
Gong H, Woolley AT, Nordin GP (2016) High density 3D printed microfluidic valves, pumps, and multiplexers. Lab Chip. https://doi.org/10.1039/c6lc00565a
Gong H, Bickham BP, Woolley AT, Nordin GP (2017) Custom 3D printer and resin for 18 μm × 20 μm microfluidic flow channels. Lab Chip. https://doi.org/10.1039/c7lc00644f
Gossett DR, Di Carlo D (2009) Particle focusing mechanisms in curving confined flows. Anal Chem. https://doi.org/10.1021/ac901306y
Gregory AD, Houghton AMG (2011) Tumor-associated neutrophils: New targets for cancer therapy. Cancer Res. 71. https://doi.org/10.1158/0008-5472.CAN-10-2583
Guan G, Wu L, Bhagat AA et al (2013) Spiral microchannel with rectangular and trapezoidal cross-sections for size based particle separation. Sci Rep. https://doi.org/10.1038/srep01475
Hagen DJ, Pemble ME, Karppinen M (2019) Atomic layer deposition of metals: precursors and film growth. Appl. Phys. Rev. 6. https://doi.org/10.1063/1.5087759
Hansson J, Karlsson JM, Haraldsson T et al (2012) Inertial microfluidics in parallel channels for high-throughput applications. Lab Chip. https://doi.org/10.1039/c2lc40241f
He Y, Wu W, Zhang T, Fu J (2015) Micro structure fabrication with a simplified hot embossing method. RSC Adv. https://doi.org/10.1039/c5ra01410g
Herrmann N, Neubauer P, Birkholz M (2019) Spiral microfluidic devices for cell separation and sorting in bioprocesses. Biomicrofluidics 13. https://doi.org/10.1063/1.5125264
Hou HW, Warkiani ME, Khoo BL et al (2013) Isolation and retrieval of circulating tumor cells using centrifugal forces. Sci Rep. https://doi.org/10.1038/srep01259
Hou HW, Bhattacharyya RP, Hung DT, Han J (2015) Direct detection and drug-resistance profiling of bacteremias using inertial microfluidics. Lab Chip. https://doi.org/10.1039/c5lc00311c
Hou HW, Petchakup C, Tay HM et al (2016) Rapid and label-free microfluidic neutrophil purification and phenoty** in diabetes mellitus. Sci Rep. https://doi.org/10.1038/srep29410
Huang LR, Cox EC, Austin RH, Sturm JC (2004) Continuous particle separation through deterministic lateral displacement. Science 80-(304):987–990. https://doi.org/10.1126/science.1094567
Huang D, Shi X, Qian Y et al (2016) Rapid separation of human breast cancer cells from blood using a simple spiral channel device. Anal Methods. https://doi.org/10.1039/c6ay01077f
Johnston ID, McDonnell MB, Tan CKL et al (2014) Dean flow focusing and separation of small microspheres within a narrow size range. Microfluid Nanofluidics. https://doi.org/10.1007/s10404-013-1322-6
Kim GY, Han JI, Park JK (2018) Inertial microfluidics-based cell sorting. Biochip J 12:257–267. https://doi.org/10.1007/s13206-018-2401-2
Kulasinghe A, Tran THP, Blick T et al (2017) Enrichment of circulating head and neck tumour cells using spiral microfluidic technology. Sci Rep. https://doi.org/10.1038/srep42517
Kumar T, Ramachandraiah H, Iyengar SN et al (2021) High throughput viscoelastic particle focusing and separation in spiral microchannels. Sci Rep. https://doi.org/10.1038/s41598-021-88047-4
Kuntaegowdanahalli SS, Bhagat AAS, Kumar G, Papautsky I (2009) Inertial microfluidics for continuous particle separation in spiral microchannels. Lab Chip. https://doi.org/10.1039/b908271a
Lashkaripour A, Silva R, Densmore D (2018) Desktop micromilled microfluidics. Microfluid Nanofluidics. https://doi.org/10.1007/s10404-018-2048-2
Lee ML, Yao DJ (2018) The separation of microalgae using dean flow in a spiral microfluidic device. Inventions. https://doi.org/10.3390/inventions3030040
Lee W, Kwon D, Choi W et al (2015) Erratum: corrigendum: 3D-printed microfluidic device for the detection of pathogenic bacteria using size-based separation in helical channel with trapezoid cross-section. Scie Rep. https://doi.org/10.1038/srep07717
Lee JH, Lee SK, Kim JH, Park JH (2019) Separation of particles with bacterial size range using the control of sheath flow ratio in spiral microfluidic channel. Sensors Actuators, A Phys. https://doi.org/10.1016/j.sna.2018.12.047
Liu N, Petchakup C, Tay HM, et al (2019) Spiral inertial microfluidics for cell separation and biomedical applications. Appl Microfluid Syst Biol Med. https://doi.org/10.1007/978-981-13-6229-3_5
Mach AJ, Adeyiga OB, Di Carlo D (2013) Microfluidic sample preparation for diagnostic cytopathology. Lab Chip 13:1011–1026. https://doi.org/10.1039/c2lc41104k
Martel JM, Toner M (2014) Inertial focusing in microfluidics. Annu. Rev. Biomed. Eng. 16. https://doi.org/10.1146/annurev-bioeng-121813-120704
Martel JM, Toner M (2012) Inertial focusing dynamics in spiral microchannels. Phys Fluids. https://doi.org/10.1063/1.3681228
McGrath J, Jimenez M, Bridle H (2014) Deterministic lateral displacement for particle separation: a review. Lab Chip 14:4139–4158. https://doi.org/10.1039/c4lc00939h
Mehran A, Rostami P, Saidi MS et al (2021) High-throughput, label-free isolation of white blood cells from whole blood using parallel spiral microchannels with u-shaped cross-section. Biosensors. https://doi.org/10.3390/bios11110406
Meng L, Gao S, Wei D et al (2021) Effects of cross-sectional geometry on flow characteristics in spiral separators. Sep Sci Technol. https://doi.org/10.1080/01496395.2020.1853169
Mihandoust A, Bazaz SR, Maleki-Jirsaraei N et al (2020) High-throughput particle concentration using complex cross-section microchannels. Micromachines. https://doi.org/10.3390/MI11040440
Mohamed Yousuff C, Hamid B, NH, Kamal Basha IH, Wei Ho ET, (2017) Output channel design for collecting closely-spaced particle streams from spiral inertial separation devices. AIP Adv. https://doi.org/10.1063/1.4986617
Mukherjee P, Wang X, Zhou J, Papautsky I (2019) Single stream inertial focusing in low aspect-ratio triangular microchannels. Lab Chip. https://doi.org/10.1039/c8lc00973b
Nivedita N, Papautsky I (2013) Continuous separation of blood cells in spiral microfluidic devices. Biomicrofluidics. https://doi.org/10.1063/1.4819275
Nivedita N, Garg N, Lee AP, Papautsky I (2017a) A high throughput microfluidic platform for size-selective enrichment of cell populations in tissue and blood samples. Analyst. https://doi.org/10.1039/c7an00290d
Nivedita N, Ligrani P, Papautsky I (2017b) Dean flow dynamics in low-aspect ratio spiral microchannels. Sci Rep. https://doi.org/10.1038/srep44072
Ookawara S, Higashi R, Street D, Ogawa K (2004) Feasibility study on concentration of slurry and classification of contained particles by microchannel. Chem Eng J. https://doi.org/10.1016/j.cej.2003.11.008
Pramuanjaroenkij A, Bunta J, Thiangpadung J, et al (2018) The development of lab-on-a-chip fabricated from two molds. In: IOP conference series: materials science and engineering. https://doi.org/10.1088/1757-899X/297/1/012023
Praveenkumar S, Sridhar PA, Lingaraja D, Dinesh RG (2018) Design and computational modeling of spiral microfluidic channel for sorting and separating the biomolecules. Curr Signal Transduct Ther 15:252–257. https://doi.org/10.2174/1574362413666181029110920
Rafeie M, Hosseinzadeh S, Huang J et al (2019) New insights into the physics of inertial microfluidics in curved microchannels II. Biomicrofluidics, Adding an additive rule to understand complex cross-sections. https://doi.org/10.1063/1.5109012
Ramya S, Lingaraja D, Ram GD et al (2021) Microfluidic circulating tumour cell sorter using deterministic lateral displacement. In: 2021 IEEE international conference on distributed computing, VLSI, electrical circuits and robotics, DISCOVER 2021: proceedings. https://doi.org/10.1109/DISCOVER52564.2021.9663577
Raoufi MA, Mashhadian A, Niazmand H et al (2019) Experimental and numerical study of elasto-inertial focusing in straight channels. Biomicrofluidics. https://doi.org/10.1063/1.5093345
Raoufi MA, RazaviBazaz S, Niazmand H et al (2020) Fabrication of unconventional inertial microfluidic channels using wax 3D printing. Soft Matter. https://doi.org/10.1039/c9sm02067e
Ravula SK, Branch DW, James CD et al (2008) A microfluidic system combining acoustic and dielectrophoretic particle preconcentration and focusing. Sensors Actuators, B Chem 130:645–652. https://doi.org/10.1016/j.snb.2007.10.024
Rogers CI, Qaderi K, Woolley AT, Nordin GP (2015) 3D printed microfluidic devices with integrated valves. Biomicrofluidics. https://doi.org/10.1063/1.4905840
Russom A, Gupta AK, Nagrath S et al (2009) Differential inertial focusing of particles in curved low-aspect-ratio microchannels. New J Phys. https://doi.org/10.1088/1367-2630/11/7/075025
Rzhevskiy AS, Bazaz SR, Ding L et al (2020) Rapid and label-free isolation of tumour cells from the urine of patients with localised prostate cancer using inertial microfluidics. Cancers (Basel). https://doi.org/10.3390/cancers12010081
Saffman PG (1965) The lift on a small sphere in a slow shear flow. J Fluid Mech. https://doi.org/10.1017/S0022112065000824
Sarkar A, Hou HW, Mahan AE et al (2016) Multiplexed affinity-based separation of proteins and cells using inertial microfluidics. Sci Rep. https://doi.org/10.1038/srep23589
Schaap A, Dumon J, den Toonder J (2016) Sorting algal cells by morphology in spiral microchannels using inertial microfluidics. Microfluid Nanofluidics. https://doi.org/10.1007/s10404-016-1787-1
Schets FM, Van Wijnen JH, Schijven JF et al (2008) Monitoring of waterborne pathogens in surface waters in Amsterdam, the Netherlands, and the potential health risk associated with exposure to Cryptosporidium and Giardia in these waters. Appl Environ Microbiol. https://doi.org/10.1128/AEM.01609-07
Segré G, Silberberg A (1961) Radial particle displacements in poiseuille flow of suspensions. Nature. https://doi.org/10.1038/189209a0
Segré G, Silberberg A (1962) Behaviour of macroscopic rigid spheres in Poiseuille flow: part 2. Experimental results and interpretation. J Fluid Mech. https://doi.org/10.1017/S0022112062001111
Shallan AI, Smejkal P, Corban M et al (2014) Cost-effective three-dimensional printing of visibly transparent microchips within minutes. Anal Chem. https://doi.org/10.1021/ac4041857
Shen S, Tian C, Li T et al (2017) Spiral microchannel with ordered micro-obstacles for continuous and highly-efficient particle separation. Lab Chip. https://doi.org/10.1039/c7lc00691h
Shiriny A, Bayareh M (2021) Inertial focusing of CTCs in a novel spiral microchannel. Chem Eng Sci. https://doi.org/10.1016/j.ces.2020.116102
Sollier E, Murray C, Maoddi P, Di Carlo D (2011) Rapid prototy** polymers for microfluidic devices and high pressure injections. Lab Chip 11. https://doi.org/10.1039/c1lc20514e
Sun J, Li M, Liu C et al (2012) Double spiral microchannel for label-free tumor cell separation and enrichment. Lab Chip 12:3952–3960. https://doi.org/10.1039/c2lc40679a
Sun J, Liu C, Li M et al (2013) Size-based hydrodynamic rare tumor cell separation in curved microfluidic channels. Biomicrofluidics. https://doi.org/10.1063/1.4774311
Tabatabaei SA, Zabetian Targhi M, Javaherchian J, Yaghoubi M (2022) Basic concepts of biological microparticles isolation by inertia spiral microchannels in simple terms: a review. J Micromech Microeng 32. https://doi.org/10.1088/1361-6439/ac388c
Tabatabaei SA, ZabetianTarghi M (2021) Design and experimental investigation of a novel spiral microfluidic chip to separate wide size range of micro-particles aimed at cell separation. Proc Inst Mech Eng Part H J Eng Med. https://doi.org/10.1177/09544119211029753
Tang W, Zhu S, Jiang D et al (2020) Channel innovations for inertial microfluidics. Lab Chip 20:3485–3502. https://doi.org/10.1039/d0lc00714e
Tay HM, Kharel S, Dalan R et al (2017) Rapid purification of sub-micrometer particles for enhanced drug release and microvesicles isolation. NPG Asia Mater. https://doi.org/10.1038/am.2017.175
Thanormsridetchai A, Ketpun D, Srituravanich W et al (2017) Focusing and sorting of multiple-sized beads and cells using low-aspect-ratio spiral microchannels. J Mech Sci Technol 31:5397–5405. https://doi.org/10.1007/s12206-017-1034-z
Tuerk C, Gold L (1990) Systematic evolution of ligands by exponential enrichment: RNA ligands to bacteriophage T4 DNA polymerase. Science. https://doi.org/10.1126/science.2200121
Urrios A, Parra-Cabrera C, Bhattacharjee N et al (2016) 3D-printing of transparent bio-microfluidic devices in PEG-DA. Lab Chip. https://doi.org/10.1039/c6lc00153j
Vona G, Sabile A, Louha M et al (2000) Isolation by size of epithelial tumor cells: a new method for the immunomorphological and molecular characterization of circulating tumor cells. Am J Pathol. https://doi.org/10.1016/S0002-9440(10)64706-2
Wang Z, Zhe J (2011) Recent advances in particle and droplet manipulation for lab-on-a-chip devices based on surface acoustic waves. Lab Chip 11:1280–1285. https://doi.org/10.1039/c0lc00527d
Wang J, Lu W, Tang C et al (2015) Label-free isolation and mRNA detection of circulating tumor cells from patients with metastatic lung cancer for disease diagnosis and monitoring therapeutic efficacy. Anal Chem. https://doi.org/10.1021/acs.analchem.5b03484
Wang X, Liedert C, Liedert R, Papautsky I (2016) A disposable, roll-to-roll hot-embossed inertial microfluidic device for size-based sorting of microbeads and cells. Lab Chip. https://doi.org/10.1039/c6lc00215c
Warkiani ME, Guan G, Luan KB et al (2014a) Slanted spiral microfluidics for the ultra-fast, label-free isolation of circulating tumor cells. Lab Chip. https://doi.org/10.1039/c3lc50617g
Warkiani ME, Khoo BL, Tan DSW et al (2014b) An ultra-high-throughput spiral microfluidic biochip for the enrichment of circulating tumor cells. Analyst. https://doi.org/10.1039/c4an00355a
Warkiani ME, Tay AKP, Guan G, Han J (2015a) Membrane-less microfiltration using inertial microfluidics. Sci Rep. https://doi.org/10.1038/srep11018
Warkiani ME, Tay AKP, Khoo BL et al (2015b) Malaria detection using inertial microfluidics. Lab Chip. https://doi.org/10.1039/c4lc01058b
Whitesides GM (2006) The origins and the future of microfluidics. Nature 442:368–373. https://doi.org/10.1038/nature05058
Whitesides GM, Ostuni E, Takayama S, et al (2001) Soft lithography in biology and biochemistry. Annu. Rev. Biomed. Eng. 3. https://doi.org/10.1146/annurev.bioeng.3.1.335
Wu L, Guan G, Hou HW et al (2012) Separation of leukocytes from blood using spiral channel with trapezoid cross-section. Anal Chem. https://doi.org/10.1021/ac302085y
**ang N, Ni Z (2015) High-throughput blood cell focusing and plasma isolation using spiral inertial microfluidic devices. Biomed Microdevices. https://doi.org/10.1007/s10544-015-0018-y
**ang N, Chen K, Sun D et al (2013) Quantitative characterization of the focusing process and dynamic behavior of differently sized microparticles in a spiral microchannel. Microfluid Nanofluidics. https://doi.org/10.1007/s10404-012-1025-4
Xu X, Huang X, Sun J, et al (2021) Recent progress of inertial microfluidic-based cell separation. Analyst 146. https://doi.org/10.1039/d1an01160j
Yager P, Edwards T, Fu E, et al (2006) Microfluidic diagnostic technologies for global public health. Nature 442. https://doi.org/10.1038/nature05064
Yamada M, Nakashima M, Seki M (2004) Pinched flow fractionation: continuous size separation of particles utilizing a laminar flow profile in a pinched microchannel. Anal Chem 76:5465–5471. https://doi.org/10.1021/ac049863r
Yeo DC, Wiraja C, Zhou Y et al (2015) Interference-free micro/nanoparticle cell engineering by use of high-throughput microfluidic separation. ACS Appl Mater Interfaces. https://doi.org/10.1021/acsami.5b06167
Zabaglo L, Ormerod MG, Parton M et al (2003) Cell filtration-laser scanning cytometry for the characterisation of circulating breast cancer cells. Cytom Part A. https://doi.org/10.1002/cyto.a.10071
Zhang J, Yan S, Yuan D et al (2016) Fundamentals and applications of inertial microfluidics: a review. Lab Chip 16:10–34. https://doi.org/10.1039/c5lc01159k
Zhang Y, Zheng T, Wang L, et al (2021) From passive to active sorting in microfluidics: A review. Rev Adv Mater Sci 60. https://doi.org/10.1515/rams-2020-0044
Zhao Q, Yuan D, Zhang J, Li W (2020) A review of secondary flow in inertial microfluidics. Micromachines 11. https://doi.org/10.3390/MI11050461
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The corresponding author declares that there is no conflict of interest in submitting this paper on behalf of the other authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ramya, S., Kumar, S.P., Ram, G.D. et al. A short review of spiral microfluidic devices with distinct cross-sectional geometries. Microfluid Nanofluid 26, 95 (2022). https://doi.org/10.1007/s10404-022-02593-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10404-022-02593-5