Abstract
Objective
To investigate the value of 68 Ga-PSMA-11 positron emission tomography/computerized tomography (PET/CT) in evaluating lacrimal and salivary glands function.
Methods
Ten patients with pSS and 18 healthy volunteers were recruited in this study. All participants underwent 68 Ga-PSMA-11 PET/CT, and the patients with pSS performed salivary gland scintigraphy the next day. The maximum standardized uptake value (SUVmax), average of the standard uptake value (SUVavg), the average CT value (CTavg), and volume (V) in the region of interest (ROI) of each lacrimal and salivary gland were analyzed in68Ga-PSMA-11 PET/CT. The uptake ratio (UR) of the bilateral parotid gland and submandibular gland was calculated in salivary gland scintigraphy (SGS). Statistical analysis was processed by the SPSS software and the MedCalc software. A p-value of < 0.05 was considered as statistically significant.
Results
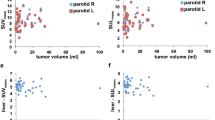
Almost all the parameters of pSS were significantly lower than those of the control group (p < 0.05). The left parotid gland (PG) UR was positive correlation with left PG SUVmax (r = 0.758, p = 0.011) and left PG SUVavg (r = 0.770, p = 0.009); the right PGUR was positive correlation with right PG SUVmax (r = 0.721, p = 0.019) and right PG SUVavg (r = 0.721, p = 0.019). The SUVmax and SUVavg of both sides of acrimal and salivary glands had area under the receiver operating curve values greater than 0.5.
Conclusions
68 Ga-PSMA-11 PET/CT can simultaneously enable the visualization of lacrimal glands and salivary glands and be used to evaluate the lacrimal and salivary glands function.
Key Points • We have firstly investigated the value of 68 Ga-PSMA-11 PET/CT in evaluating lacrimal and salivary glands function in patients with pSS 68 Ga-PSMA-11 PET/CT can simultaneously allow the visualization of lacrimal glands and salivary glands. • The results of the present study imply that 68 Ga-PSMA-11 PET/CT can be used to evaluate the lacrimal and salivary glands function in patients with pSS meanwhile. |



Similar content being viewed by others
Data availability
Not applicable.
Code availability
SPSS software (version 16.0; SPSS Inc., Chicago, IL, USA) and the MedCalc software (ver. 10.4.8; MedCalc Software, Mariakerke, Belgium).
References
Bayetto K, Logan RM (2010) Sjögren’s syndrome: a review of aetiology, pathogenesis, diagnosis and management. Aust Dent J 55(Suppl 1):39–47. https://doi.org/10.1111/j.1834-7819.2010.01197.x
Shen L, He J, Kramer JM, Bunya VY (2019) Sjögren’s syndrome: animal models, etiology, pathogenesis, clinical subtypes, and diagnosis. J Immunol Res 2019:8101503. https://doi.org/10.1155/2019/8101503
Manoussakis MN, Kapsogeorgou EK (2007) The role of epithelial cells in the pathogenesis of Sjögren’s syndrome. Clin Rev Allergy Immunol 32:225–230. https://doi.org/10.1007/s12016-007-8007-4
Mojsak MN, Rogowski F (2010) Application scintigraphy in evaluation of salivary gland function. Polski merkuriusz lekarski : organ Polskiego Towarzystwa Lekarskiego 28:214–219
Parisis D, Chivasso C, Perret J, Soyfoo MS, Delporte C (2020) Current state of knowledge on primary Sjögren’s syndrome, an autoimmune exocrinopathy. J Clin Med 9. https://doi.org/10.3390/jcm9072299
Pfob CH, Ziegler S, Graner FP, Köhner M, Schachoff S, Blechert B, Wester HJ, Scheidhauer K, Schwaiger M, Maurer T, Eiber M (2016) Biodistribution and radiation dosimetry of (68)Ga-PSMA HBED CC-a PSMA specific probe for PET imaging of prostate cancer. Eur J Nucl Med Mol Imaging 43:1962–1970. https://doi.org/10.1007/s00259-016-3424-3
Demirci E, Toklu T, Yeyin N, Ocak M, Alan-Selcuk N, Araman A, Kabasakal L (2018) Estimation of the organ absorbed doses and effective dose from 68Ga-PSMA-11 PET scan. Radiat Prot Dosimetry 182:518–524. https://doi.org/10.1093/rpd/ncy111
Rupp NJ, Umbricht CA, Pizzuto DA, Lenggenhager D, Töpfer A, Müller J, Muehlematter UJ, Ferraro DA, Messerli M, Morand GB, Huber GF, Eberli D, Schibli R, Müller C, Burger IA (2019) First clinicopathologic evidence of a non-PSMA-related uptake mechanism for (68)Ga-PSMA-11 in salivary glands. J Nucl Med 60:1270–1276. https://doi.org/10.2967/jnumed.118.222307
Shiboski CH, Shiboski SC, Seror R, Criswell LA, Labetoulle M, Lietman TM, Rasmussen A, Scofield H, Vitali C, Bowman SJ, Mariette X (2017) 2016 American College of Rheumatology/European League Against Rheumatism classification criteria for primary Sjögren’s syndrome: a consensus and data-driven methodology involving three international patient cohorts. Arthritis Rheumatol (Hoboken, NJ) 69:35–45. https://doi.org/10.1002/art.39859
Paschides CA, Kitsios G, Karakostas KX, Psillas C, Moutsopoulos HM (1989) Evaluation of tear break-up time, Schirmer’s-I test and rose bengal staining as confirmatory tests for keratoconjunctivitis sicca. Clin Exp Rheumatol 7:155–157
Klutmann S, Bohuslavizki KH, Kröger S, Bleckmann C, Brenner W, Mester J, Clausen M (1999) Quantitative salivary gland scintigraphy. J Nucl Med Technol 27:20–26
Kim HA, Yoon SH, Yoon JK, Lee SJ, Jo KS, Lee DH, Suh CH, An YS (2014) Salivary gland scintigraphy in Sjögren’s syndrome. Comparison of the diagnostic performance of visual and semiquantitative analysis. Nuklearmedizin 53:139–145. https://doi.org/10.3413/Nukmed-0630-13-10
Skopouli FN, Dafni U, Ioannidis JP, Moutsopoulos HM (2000) Clinical evolution, and morbidity and mortality of primary Sjögren’s syndrome. Semin Arthritis Rheum 29:296–304. https://doi.org/10.1016/s0049-0172(00)80016-5
Kassan SS, Moutsopoulos HM (2004) Clinical manifestations and early diagnosis of Sjögren syndrome. Arch Intern Med 164:1275–1284. https://doi.org/10.1001/archinte.164.12.1275
Guellec D, Cornec D, Jousse-Joulin S, Marhadour T, Marcorelles P, Pers JO, Saraux A, Devauchelle-Pensec V (2013) Diagnostic value of labial minor salivary gland biopsy for Sjögren’s syndrome: a systematic review. Autoimmun Rev 12:416–420. https://doi.org/10.1016/j.autrev.2012.08.001
Sun Z, Zhang Z, Fu K, Zhao Y, Liu D, Ma X (2012) Diagnostic accuracy of parotid CT for identifying Sjögren’s syndrome. Eur J Radiol 81:2702–2709. https://doi.org/10.1016/j.ejrad.2011.12.034
Parkin B, Chew JB, White VA, Garcia-Briones G, Chhanabhai M, Rootman J (2005) Lymphocytic infiltration and enlargement of the lacrimal glands: a new subtype of primary Sjögren’s syndrome? Ophthalmology 112:2040–2047. https://doi.org/10.1016/j.ophtha.2005.06.014
Acknowledgements
We gratefully thank the technicians in our department for their help.
Author information
Authors and Affiliations
Contributions
J-FL, L-PQ, Q-YW, X-HG, J-ZY, and Q-CW made substantial contributions to the conception and design, acquisition of data, and analysis and interpretation of data. L-JX and M-HC drafted and revised the manuscript critically for important intellectual content and approved the final manuscript for submission and publication.
Corresponding authors
Ethics declarations
Ethics approval
The study followed the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Consent to participate
Written informed consent was obtained from the patient for publication.
Consent for publication
Written informed consent was obtained from the patient for publication.
Conflicts of interest
All authors have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Jian-fang Li and Lu-** Qin equally contribute to this paper and should be considered as co-first authors.
Liang-jun **e and Mu-hua Cheng equally contribute to this paper and should be considered as cocorresponding author.
Rights and permissions
About this article
Cite this article
Li, Jf., Qin, Lp., Wu, Qy. et al. The value of 68 Ga-PSMA-11 positron emission tomography/computerized tomography in evaluating the lacrimal and salivary glands function. Clin Rheumatol 41, 1543–1550 (2022). https://doi.org/10.1007/s10067-021-06038-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-021-06038-2