Abstract
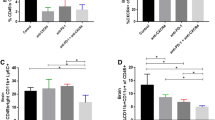
Although chemoimmunotherapy often lengthens glioblastoma (GBM) survival, early relapses remain problematic as immunosuppressive M2 macrophages (Mϕ) that function via inhibitory cytokine and PD-L1 production cause immunotherapy resistance. Here, we detail anti-PD-L1 antibody effects on the tumor microenvironment, including Mϕ infiltration, using a temozolomide (TMZ)-treated glioma model. In addition, we tested combinations of anti-PD-L1 antibody and the M2Mϕ inhibitor IPI-549 on tumor growth. We simulated late TMZ treatment or relapse stage, persistent GBM cells by generating TMZ-resistant TS (TMZRTS) cells. M2Mϕ-associated cytokine production and PD-L1 expression in these cells were investigated. TMZRTS cells were then subcutaneously implanted into C57BL/6 mice to determine the effectiveness of an anti-PD-L1 antibody and/or IPI-549 treatment on infiltration of CD163-positive Mϕ, usually considered as an M2Mϕ marker into tumor tissues. CD163 expression in samples from human GBM patients were also evaluated. CD163-positive Mϕ heavily infiltrated TMZRS tumor tissues after in vivo anti-PD-L1 antibody treatment. Tumor growth was strongly inhibited by anti-PD-L1 antibody and IPI-549 combination therapy. Anti-PD-L1 antibody treatment significantly reduced infiltration of CD163-positive Mϕ into tumors, while combined PD-L1 antibody and IPI-549 therapy remarkably inhibited tumor growth. These therapies may be useful for recurrent or chronic GBM after TMZ treatment, but clinical safety and effectiveness studies are needed.





Similar content being viewed by others
Abbreviations
- FRT:
-
Fractionated radiotherapy
- GBM:
-
Glioblastoma multiforme
- IHC:
-
Immunohistochemistry
- MGMT:
-
O6-Methylguanine-DNA methyltransferase
- PD-1:
-
Programmed cell death 1
- PD-L1:
-
PD-1 ligand
- TIL:
-
Tumor-infiltrating lymphocytes
- Mϕ:
-
Macrophage
- TMZ:
-
Temozolomide
References
Stupp R, Mason WP, van den Bent MJ et al (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352:987–996
Hodi FS, O'Day SJ, McDermott DF et al (2010) Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 363(8):711–723
Borghaei H, Paz-Ares L, Horn L et al (2015) Nivolumab versus docetaxel in advanced nonsquamous non-small-cell lung cancer. N Engl J Med 373:1627–1639
Garon EB, Rizvi NA, Hui R et al (2015) Pembrolizumab for the treatment of non-small-cell lung cancer. N Engl J Med 372:2018–2028
Ansell SM, Lesokhin AM, Borrello I et al (2015) PD-1 blockade with nivolumab in relapsed or refractory Hodgkin's lymphoma. N Engl J Med. 372(4):311–319
Kang YK, Boku N, Satoh T et al (2017) Nivolumab in patients with advanced gastric or gastro-oesophageal junction cancer refractory to, or intolerant of, at least two previous chemotherapy regimens (ONO-4538-12, ATTRACTION-2): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 390(10111):2461–2471
Powles T, Durán I, van der Heijden MS et al (2018) Atezolizumab versus chemotherapy in patients with platinum-treated locally advanced or metastatic urothelial carcinoma (IMvigor211): a multicentre, open-label, phase 3 randomised controlled trial. Lancet 391:748–757
Cohen EEW, Soulières D, Tourneau C, KEYNOTE-040 investigators et al (2019) Pembrolizumab versus methotrexate, docetaxel, or cetuximab for recurrent or metastatic head-and-neck squamous cell carcinoma (KEYNOTE-040): a randomised, open-label, phase 3 study. Lancet 393(10167):156–167
Omuro A, Vlahovic G, Lim M et al (2018) Nivolumab with or without ipilimumab in patients with recurrent glioblastoma: results from exploratory phase I cohorts of CheckMate 143. Neuro Oncol. 20(5):674–686
Zhao J, Chen AX, Gartrell RD et al (2019) Immune and genomic correlates of response to anti-PD-1 immunotherapy in glioblastoma. Nat Med. 25(3):462–469
Shen X, Zhao B (2018) Efficacy of PD-1 or PD-L1 inhibitors and PD-L1 expression status in cancer: meta-analysis. BMJ 362:k3529
Feng YY, Griffith OL, Griffith M (2017) Clinical implications of neoepitope landscapes for adult and pediatric cancers. Genome Med 9(1):77
Rosenberg JE, Hoffman-Censits J, Powles T et al (2016) Atezolizumab in patients with locally advanced and metastatic urothelial carcinoma who have progressed following treatment with platinum-based chemotherapy: a single-arm, multicentre, phase 2 trial. Lancet 387(10031):1909–1920
Yarchoan M, Hopkins A, Jaffee EM (2017) Tumor mutational burdenand response rate to PD-1 inhibition. N Engl J Med 377(25):2500–2501
Howitt BE, Shukla SA, Sholl LM et al (2015) Association of polymerase e-mutated and microsatellite-instable endometrial cancers with neoantigen load, number of tumor-infiltrating lymphocytes, and expression of PD-1 and PD-L1. JAMA Oncol. 1(9):1319–1323
Overman MJ, McDermott R, Leach JL et al (2017) Nivolumab in patients with metastatic DNA mismatch repair-deficient or microsatellite instability-high colorectal cancer (CheckMate 142): an open-label, multicentre, phase 2 study. Lancet Oncol. 18(9):1182–1191
Le DT, Durham JN, Smith KN et al (2017) Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade. Science 357(6349):409–413
Chae YK, Anker JF, Bais P et al (2017) Mutations in DNA repair genes are associated with increased neo-antigen load and activated T cell infiltration in lung adenocarcinoma. Oncotarget 9(8):7949–7960
Prelaj A, Tay R, Ferrara R et al (2019) Predictive biomarkers of response for immune checkpoint inhibitors in non-small-cell lung cancer. Eur J Cancer. 106:144–159
Miyazaki T, Ishikawa E, Matsuda M et al (2017) Assessment of PD-1 positive cells on initial and secondary resected tumor specimens of newly diagnosed glioblastoma and its implications on patient outcome. J Neurooncol. 133(2):277–285
Osuka S, Van Meir EG (2017) Overcoming therapeutic resistance in glioblastoma: the way forward. J Clin Invest. 127(2):415–426
Jhaveri N, Chen TC, Hofman FM (2016) Tumor vasculature and glioma stem cells: contributions to glioma progression. Cancer Lett. 380(2):545–551
Silver DJ, Sinyuk M, Vogelbaum MA et al (2016) The intersection of cancer, cancer stem cells, and the immune system: therapeutic opportunities. Neuro Oncol. 18(2):153–159
Chen Z, Feng X, Herting CJ et al (2017) Cellular and molecular identity of tumor-associated macrophages in glioblastoma. Cancer Res. 77(9):2266–2278
Chen Z, Hambardzumyan D (2018) Immune microenvironment in glioblastoma subtypes. Front Immunol. 9:1004
Nusblat LM, Carroll MJ, Roth CM (2017) Crosstalk between M2 macrophages and glioma stem cells. Cell Oncol. 40(5):471–482
Evans CA, Liu T, Lescarbeau A et al (2016) Discovery of a Selective Phosphoinositide-3-Kinase (PI3K)-γ Inhibitor (IPI-549) as an immuno-oncology clinical candidate. ACS Med Chem Lett. 7(9):862–867
Hartley GP, Chow L, Ammons DT et al (2018) Programmed cell death ligand 1 (PD-L1) signaling regulates macrophage proliferation and activation. Cancer Immunol Res. 6(10):1260–1273
Sampetrean O, Saga I, Nakanishi M et al (2011) Invasion precedes tumor mass formation in a malignant brain tumor model of genetically modified neural stem cells. Neoplasia 13:784–791
Hegi ME, Diserens AC, Gorlia T et al (2005) MGMT gene silencing and benefit from temozolomide in glioblastoma. N Engl J Med. 352(10):997–1003
Cloughesy TF, Mochizuki AY, Orpilla JR et al (2019) Neoadjuvant anti-PD-1 immunotherapy promotes a survival benefit with intratumoral and systemic immune responses in recurrent glioblastoma. Nat Med. 25(3):477–486
Schalper KA, Rodriguez-Ruiz ME, Diez-Valle R et al (2019) Neoadjuvant nivolumab modifies the tumor immune microenvironment in resectable glioblastoma. Nat Med. 25(3):470–476
Ishikawa E, Tsuboi K, Yamamoto T et al (2007) Clinical trial of autologous formalin-fixed tumor vaccine for glioblastoma multiforme patients. Cancer Sci. 98(8):1226–1233
Muragaki Y, Maruyama T, Iseki H et al (2011) Phase I/IIa trial of autologous formalin-fixed tumor vaccine concomitant with fractionated radiotherapy for newly diagnosed glioblastoma. J Neurosurg. 115(2):248–255
Ishikawa E, Muragaki Y, Yamamoto T et al (2014) Phase I/IIa trial of fractionated radiotherapy, temozolomide, and autologous formalin-fixed tumor vaccine for newly diagnosed glioblastoma. J Neurosurg. 121(3):543–553
Ishikawa E, Yamamoto T, Matsumura A (2017) Prospect of immunotherapy for glioblastoma: tumor vaccine, immune checkpoint inhibitors and combination therapy. Neurol Med Chir (Tokyo). 57(7):321–330
Blando J, Sharma A, Higa MG et al (2019) Comparison of immune infiltrates in melanoma and pancreatic cancer highlights VISTA as a potential target in pancreatic cancer. Proc Natl Acad Sci U S A. 116(5):1692–1697
Kaneda MM, Messer KS, Ralainirina N et al (2016) PI3Kγ is a molecular switch that controls immune suppression. Nature 539(7629):437–442
De Henau O, Rausch M, Winkler D et al (2016) Overcoming resistance to checkpoint blockade therapy by targeting PI3Kγ in myeloid cells. Nature 539(7629):443–447
Barros MH, Hauck F, Dreyer JH et al (2013) Macrophage polarisation: an immunohistochemical approach for identifying M1 and M2 macrophages. PLoS ONE 8(11):e80908
Fujimura T, Kambayashi Y, Fujisawa Y et al (2018) Tumor-associated macrophages: therapeutic targets for skin cancer. Front Oncol 8:3
Xu C, Chen YP, Du XJ et al (2018) Comparative safety of immune checkpoint inhibitors in cancer: systematic review and network meta-analysis. BMJ 363:k4226
Acknowledgements
The authors thank Dr. Alexander Zaboronok, Department of Neurosurgery, for critical revision. The authors would also like to thank Dr. Bryan J. Mathis of the Medical English Communications Center of the Faculty of Medicine, University of Tsukuba, for language revision.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
This study was supported by a Grant-in-Aid for Scientific Research (Research No. 18K08962) from the Japanese Ministry of Education, Culture, Sports, Science and Technology (MEXT) and the project for promotion of practical applications of advanced medical technologies in Tsukuba University Hospital.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10014_2020_357_MOESM1_ESM.tif
Supplemental Figure 1 The mRNA expression levels of DNA mismatch repair genes (MLH1, PMS2, MSH2, MSH3, MSH6) in TS and TMZRTS cells. (n = 3, mean ± SD) (TIF 48 kb)
10014_2020_357_MOESM2_ESM.tif
Supplemental Figure 2 CD163 mRNA expression in GBM patients and outcomes A. Datasets of CD163 mRNA expression in GBM and normal brain tissues using GlioVis. B. Survival outcome of GBM patients with high and low CD163 mRNA expression (TIF 112 kb)
10014_2020_357_MOESM3_ESM.tif
Supplemental Table S-Table 1 GBM patient characteristics, treatment histories and pathological status determined by immunohistochemistry (IHC) (TIF 85 kb)
Rights and permissions
About this article
Cite this article
Miyazaki, T., Ishikawa, E., Matsuda, M. et al. Infiltration of CD163-positive macrophages in glioma tissues after treatment with anti-PD-L1 antibody and role of PI3Kγ inhibitor as a combination therapy with anti-PD-L1 antibody in in vivo model using temozolomide-resistant murine glioma-initiating cells. Brain Tumor Pathol 37, 41–49 (2020). https://doi.org/10.1007/s10014-020-00357-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10014-020-00357-z