Abstract
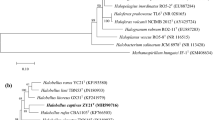
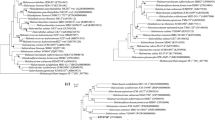
Two extremely halophilic archaea, isolates SYSU A00711T and SYSU A00630, were isolated from a sediment soil sample collected from the Aiding lake, China. Cells of these isolates were cocci, non-motile and stained Gram-negative. They grew optimally at 37 °C, with 20–22% NaCl (w/v) and at pH 7.5–8.0. Cells lysed in distilled water. Major polar lipids were phosphatidylglycerol, phosphatidylglycerol phosphate methyl ester, mannosyl glucosyl diether, sulfated mannosyl glucosyl diether, and two unidentified glycolipids. Pairwise sequence comparison revealed that isolates SYSU A00711T and SYSU A00630 were closely related to Halegenticoccus soli SYSU A9-0T (94.1 and 94.0% 16S rRNA gene sequence similarities; 94.0 and 94.2% rpoB' gene similarities, respectively). The overall genomic relatedness indices values between the two isolates and Halegenticocus soli SYSU A9-0 T were: AAI, both 79.6%; ANI, 84.6 and 84.5%; dDDH, 32.5 and 26.3%, respectively. Phylogenetic trees based on the 16S rRNA gene, rpoB' gene, and genome sequences demonstrated a robust clade of these two isolates with Halegenticoccus soli SYSU A9-0T. The DNA G + C contents of these two isolates are both 64.7% (genome method). Based on the differences in phenotypic, chemotaxonomic, and phylogenetic properties, isolates SYSU A00711T and SYSU A00630 are characterized to represent a novel species in the genus Halegenticoccus, for which the name Halegenticoccus tardaugens sp. nov. is proposed. The type strain of the species Halegenticoccus tardaugens is SYSU A00711T (= KCTC 4245T = CGMCC 1.15768T).




Similar content being viewed by others
Notes
The rpoB' gene sequences of isolates SYSU A00711T and SYSU A00630 have been deposited in the GenBank/EMBL/DDBJ with the accession numbers MN592642 and MN592641, respectively. The whole-genome shotgun project data of isolates SYSU A00711T and SYSU A00630 have been deposited at DDBJ/ENA/GenBank under the accessions SDIC00000000 and VCAT00000000, respectively
References
Burns DG, Janssen PH, Itoh T et al (2010) Halonotius pteroides gen. nov., sp. nov., an extremely halophilic archaeon recovered from a saltern crystallizer. Int J Syst Evol Microbiol 60(5):1196–1199. https://doi.org/10.1099/ijs.0.010017-0
Castresana J (2000) Selection of conserved blocks from multiple alignments for their use in phylogenetic analysis. Mol Biol Evol 17:540–552. https://doi.org/10.1093/oxfordjournals.molbev.a026334
Chen S, Liu HC, Zhou J, **ang H (2016) Haloparvum sedimenti gen. nov., sp. nov., a member of the family Haloferacaceae. Int J Syst Evol Microbiol 66:2327–2334. https://doi.org/10.1099/ijsem.0.001033
Cui HL, Lin ZY, Dong Y et al (2007) Halorubrum litoreum sp. nov., an extremely halophilic archaeon from a solar saltern. Int J Syst Evol Microbiol 57:2204–2206. https://doi.org/10.1099/ijs.0.65268-0
Cui HL, Li XY, Gao X et al (2010a) Halopelagius inordinatus gen. nov., sp. nov., a new member of the family Halobacteriaceae isolated from a marine solar saltern. Int J Syst Evol Microbiol 60:2089–2093. https://doi.org/10.1099/ijs.0.018598-0
Cui HL, Sun FF, Gao X et al (2010b) Haladaptatus litoreus sp. nov., an extremely halophilic archaeon from a marine solar saltern, and emended description of the genus Haladaptatus. Int J Syst Evol Microbiol 60:1085–1089. https://doi.org/10.1099/ijs.0015933-0
Durán-Visera A, Andrei AS, Ghai R et al (2019) New Halonotius species provide genomics-based insights into cobalamin synthesis in Haloarchaea. Front Microbiol 10:1928. https://doi.org/10.3389/fmicb.2019.01928
Dussault HP (1955) An improved technique for staining red halophilic bacteria. J Bacteriol 70:484–485. https://doi.org/10.1128/jb.70.4.484-485.1955
Edgar RC (2004) MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res 32:1792–1797. https://doi.org/10.1093/nar/gkh340
Felsenstein J (1981) Evolutionary trees from DNA sequences: a maximum likelihood approach. J Mol Evol 17:368–376. https://doi.org/10.1007/bf01734359
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39:783–791. https://doi.org/10.1111/j.1558-5646.1985.tb00420.x
Fitch WM (1971) Toward defining the course of evolution: minimum change for a specific tree topology. Syst Zool 20:406–416. https://doi.org/10.1093/sysbio/20.4.406
Gupta RS, Naushad S, Baker S (2015) Phylogenomic analyses and molecular signatures for the class Halobacteria and its two major clades: a proposal for division of the class Halobacteria into an emended order Halobacteriales and two new orders, Haloferacales ord. nov. and Natrialbales ord. nov., containing the novel families Haloferacaceae fam. nov. and Natrialbaceae fam. nov. Int J Syst Evol Microbiol 65:1050–1069. https://doi.org/10.1099/ijs.0.070136-0
Gupta RS, Naushad S, Fabros R, Adeolu M (2016a) A phylogenomic reappraisal of family-level divisions within the class Halobacteria: proposal to divide the order Halobacteriales into the families Halobacteriaceae, Haloarculaceae fam. nov., and Halococcaceae fam. nov., and the order Haloferacales into the families, Haloferacaceae and Halorubraceae fam nov. Antonie van Leeuwenhoek 109:565–587. https://doi.org/10.1007/s10482-016-0660-2
Gupta RS, Naushad S, Fabros R, Adeolu M (2016b) Erratum to: A phylogenomic reappraisal of family-level divisions within the class Halobacteria: proposal to divide the order Halobacteriales into the families Halobacteriaceae, Haloarculaceae fam. nov., and Halococcaceae fam. nov., and the order Haloferacales into the families, Haloferacaceae and Halorubraceae fam nov. Antonie van Leeuwenhoek 109:1521–1523. https://doi.org/10.1007/s10482-016-0765-7
Gutierrez MC, Castillo AM, Kamekura M, Ventosa A (2008) Haloterrigena salina sp. nov., an extremely halophilic archaeon isolated from a salt lake. Int J Syst Evol Microbiol 58:2880–2884. https://doi.org/10.1099/ijs.0.2008/001602-0
Harrison PG, Strulo B (2000) SPADES—a process algebra for discrete event simulation. J Logic Comput 10:3–42. https://doi.org/10.1093/logcom/10.1.3
Kates M (2010) Techniques of lipidology: isolation, analysis, and identification of lipids, 3rd edn. NewportSomerville
Kimura M (1980) A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16:111–120. https://doi.org/10.1007/bf01731581
Kumar S, Stecher G, Li M et al (2018) MEGA X: molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol 35:1547–1549. https://doi.org/10.1093/molbev/msy096
Lee I, Chalita M, Ha SM et al (2017) ContEst16S: an algorithm that identifies contaminated prokaryotic genomes using 16S RNA gene sequences. Int J Syst Evol Microbiol 67:2053–2057. https://doi.org/10.1099/ijsem.0.001872
Letunic I, Bork P (2021) Interactive tree of life (iTOL) v5: an online tool for phylogenetic tree display and annotation. Nucleic Acids Res 49(W1):W293–W296. https://doi.org/10.1093/nar/gkab301
Liu BB, Zhao WY, Chu X et al (2014) Haladaptatus pallidirubidus sp. nov., a halophilic archaeon isolated from saline soil samples in Yunnan and **njiang, China. Antonie van Leeuwenhoek 106:901–910. https://doi.org/10.1007/s10482-014-0259-4
Liu BB, Narsing Rao MP, Yin XQ et al (2019) Description of Halegenticoccus soli gen. nov., sp. nov., a halophilic archaeon isolated from a soil sample of Ebi lake. Extremophiles 23:521–528. https://doi.org/10.1007/s00792-019-01104-9
Meier-Kolthoff JP, Auch AF, Klenk HP, Göker M (2013) Genome sequence-based species delimitation with confidence intervals and improved distance functions. BMC Bioinform 14(1):1–14. https://doi.org/10.1186/1471-2105-14-60
Minegishi H, Kamekura M, Itoh T et al (2010) Further refinement of the phylogeny of the Halobacteriaceae based on the fulL-length RNA polymerase subunit B’ (rpoB´) gene. Int J Syst Evol Microbiol 60:2398–2408. https://doi.org/10.1099/ijs.0.017160-0
Oren A, Ventosa A, Grant WD (1997) Proposed minimal standards for description of new taxa in the order Halobacteriales. Int J Syst Bacteriol 47:233–238. https://doi.org/10.1099/00207713-47-1-233
Qiu XX, Zhao ML, Han D et al (2013) Taxonomic study of the genera Halogeometricum and Halosarcina; transfer of Halosarcina limi and Halosarcina pallida to the genus Halogeometricum as Halogeometricum limi comb. nov. and Halogeometricum pallidum comb. nov., respectively. Int J Syst Evol Microbiol 63:3915–3919. https://doi.org/10.1099/ijs.0.055038-0
Richter M, Rosselló-Móra R (2009) Shifting the genomic gold standard for the prokaryotic species definition. Proc Natl Acad Sci USA 106:19126–19131. https://doi.org/10.1073/pnas.0906412106
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425. https://doi.org/10.1093/oxfordjournalsmolbev.a040454
Salam N, Jiao JY, Zhang XT, Li WJ (2019) Update on the classification of higher ranks in the phylum Actinobacteria. Int J Syst Evol Microbiol 70(2):1331–1355. https://doi.org/10.1099/ijsem.0.003920
Thompson JD, Gibson TJ, Plewniak F et al (1997) The CLUSTAL-X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res 25:4876–4882. https://doi.org/10.1093/nar/25.24.4876
Trifinopoulos J, Nguyen LT, von Haeseler A, Minh BQ (2016) W-IQ-TREE: a fast online phylogenetic tool for maximum likelihood analysis. Nucleic Acids Res 44:W232–W235. https://doi.org/10.1093/nar/gkw256
Wayne LG, Brenner DJ, Colwell RR et al (1987) Report of the ad hoc committee on reconciliation of approaches to bacterial systematics. Int J Syst Bacteriol 37:463–464. https://doi.org/10.1099/00207713-37-4-463
Wu M, Scott AJ (2012) Phylogenomic analysis of bacterial and archaeal sequences with AMPHORA2. Bioinformatics 28:1033–1034. https://doi.org/10.1093/bioinformatics/bts079
Xu Q, Cui HL, Meng F (2019) Haloprofundus halophilus sp. nov., isolated from the saline soil of Tarim Basin. Antonie van Leeuwenhoek 112(4):553–559. https://doi.org/10.1007/s10482-018-1186-6
Yin XQ, Liu BB, Chu X et al (2017) Saliphagus infecundisoli gen. nov., sp. nov., an extremely halophilic archaeon isolated from a saline soil. Int J Syst Evol Microbiol 67:4154–4160. https://doi.org/10.1099/ijsem.0.002270
Yoon SH, Ha SM, Kwon S et al (2017a) Introducing EzBioCloud: a taxonomically united database of 16S rRNA gene sequences and whole-genome assemblies. Int J Syst Evol Microbiol 67:1613–1617. https://doi.org/10.1099/ijsem.0.001755
Yoon SH, Ha SM, Lim J et al (2017b) A large-scale evaluation of algorithms to calculate average nucleotide identity. Antonie Van Leeuwenhoek 110:1281–1286. https://doi.org/10.1007/s10482-017-0844-4
Zhang G, Gu J, Zhang R et al (2017) Haloprofundus marisrubri gen. nov., sp. nov., an extremely halophilic archaeon isolated from a brine–seawater interface. Int J Syst Evol Microbiol 67:9–16. https://doi.org/10.1099/ijsem.0.001559
Acknowledgements
The authors are grateful to Prof. Yu-Guang Zhou (CGMCC, China) for providing the reference strains. We are also grateful to Mr. Yuan-Guo **e (Sun Yat-sen University, China) for help in generating the phylogenomic tree, and Mr. Zheng-Han Lian and Dr. Jian-Yu Jiao (Sun Yat-sen University, China) for drawing the AAI heatmap.
Funding
This research was supported by the National Natural Science Foundation of China (31800001, 31850410475), Key Scientific Research Project of Colleges and Universities in Henan Province (20A180019), Key Technologies R&D Program of Henan Province (212102310232), Science and Technology Development Special Fund Program of Guangdong Province (2017A030310206) and Key Technologies R&D Program of Nanyang city (KJGG079).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Additional information
Communicated by A. Oren.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, BB., Salam, N., Cheng, S. et al. Halegenticoccus tardaugens sp. nov., an extremely halophilic archaeon isolated from a saline soil. Extremophiles 25, 483–492 (2021). https://doi.org/10.1007/s00792-021-01243-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00792-021-01243-y