Abstract
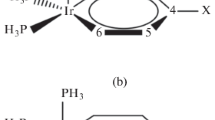
This study was conducted to investigate the effect of various substituents on the Cr=C bond in the [(OC)5Cr=C(OEt)-para-C6H4X] complexes using B3LYP*-based quantum mechanical calculations. In this respect, the study evaluates the influence of electron withdrawing and donating groups on the Cr=C bond distances and topological properties and correlates the calculated parameters with the Hammett and Brown constants for the para-substituted (σp and σ +p , respectively) functional groups. Also, the frontier orbital analysis was used to show the electronic structure of complexes and the percentage composition in terms of the defined groups of frontier orbitals was evaluated. To obtain insight into the physical nature of Cr=C bond bonds, we extensively used energy decomposition analysis and Bader’s Quantum Theory of Atoms-in-Molecules (QTAIM). With this aim, in addition to examining the bond critical points properties, we apply Pendás’ Interacting Quantum Atoms (IQA) scheme, which enables the rigorous and physical study of Cr=C bonds in these complexes.
Graphical abstract





Similar content being viewed by others
References
Taxak N, Patel B, Bharatam PV (2013) Inorg Chem 52:5097
Bourissou D, Guerret O, Gabbaï FP, Bertrand G (2000) Chem Rev 100:39
Korotkikh NI, Raenko GF, Pekhtereva TM, Shvaika OP, Cowley AH, Jones JN (2006) Russ J Org Chem 42:1822
Fischer EO, Kreis G, Kreiter CG, Müller J, Huttner G, Lorenz H (1973) Angew Chem Int Ed 12:564
Depooeter CKR (1981) Chem Rev 81:447
Wilkinson SG, Stone FGA, Abel EW (1982) Comprehensive organometallic chemistry, vol 8. Pergamon Press, Oxford, p 40
Herndon JW (2013) Coord Chem Rev 257:2899
Re RED, Hopkins MD (2005) Coord Chem Rev 249:1396
Herndon JW (2003) Coord Chem Rev 243:3
Mindiola DJ (2006) Acc Chem Res 39:813
Cordiner RL, Gugger PA, Hill AF, Willis AC (2009) Organometallics 28:6632
Colebatch L, Hill AF, Shang R, Willis AC (2010) Organometallics 29:6482
Herndon JW (2014) Coord Chem Rev 272:48
Herndon JW (2015) Coord Chem Rev 286:30
Ghiasi R (2015) J Theor Comput Chem 14:1550022
Ghiasi R, Mokarram EE (2011) Russ J Phys Chem A 875:1174
Ghiasi R, Abdolmohammadi S, Moslemizadeh S (2015) J Chin Chem Soc 62:898
Ghobadi H, Ghiasi R, Jamehbozorgi S (2017) J Chin Chem Soc 64:522
Shamami MK, Ghiasi R, Asli MD (2017) J Chin Chem Soc 64:522
Shamami MK, Ghiasi R (2017) J Chin Chem Soc 64:651
Sierra MA (2000) Chem Rev 100:3591
Meijere AD, Schirmer H, Duetsch M (2000) Angew Chem Int Ed 39:3964
Barluenga J, Santamarıa J, Tomas M (2004) Chem Rev 104:2259
Gomez-Gallego M, Mancheno MJ, Sierra MA (2005) Acc Chem Res 38:44
Sierra MA, Gomez-Gallego M, Martınez-Alvarez R (2007) Chem Eur J 13:736
Lage ML, Fernandez I, Mancheno MJ, Sierra MA (2008) Inorg Chem 47:5253
Stockmann A, Kurzawa J, Fritz N, Acar N, Schneider S, Daub J, Engl R, Clark T (2002) J Phys Chem A 106:7958
Ottonelli M, Piccardo M, Duce D, Thea S, Dellepiane G (2012) J Phys Chem A 116:611
Cheng Y-H, Fang Y, Zhao X, Liu L, Guo Q-X (2002) Bull Chem Soc Jpn 75:1715
Pichierri F (2017) Theor Chem Acc 136:114
Remya GS, Suresh CH (2016) Phys Chem Chem Phys 18:20615
Szatylowicz H, Jezuita A, Siodła T, Varaksin KS, Domanski MA, Ejsmont K, Krygowski TM (2017) ACS Omega 2:7163
Ghiasi R, Zamani A (2017) J Chin Chem Soc 64:1340
Ghiasi R, Pasdar H, Fereidoni S (2016) Russ J Inorg Chem 61:327
Ghiasi R, Heydarbeighi A (2016) Russ J Inorg Chem 61:985
Ghiasi R, Pasdar H, Irajizadeh F (2015) J Chil Chem Soc 60:2740
Peikari A, Ghiasi R, Pasdar H (2015) Russ J Phys Chem A 89:250
Ghiasi R, Amini E (2015) J Struct Chem 56:1483
Fashami MZ, Ghiasi R (2015) J Struct Chem 56:1474
Ghiasi R, Boshak A (2013) J Mex Chem Soc 57:8
Pasdar H, Ghiasi R (2009) Main Group Chem 8:143
Egorochkin AN, Kuznetsova OV, Khamaletdinova NM, Domratcheva-Lvova LG (2018) Inorg Chim Acta 471:148
Anane H, Houssame SE, Guerraze AE, Guermoune A, Boutalib A, Jarid A, Nebot-Gil I, Tomás F (2008) Cent Eur J Chem 6:400
Denning DM, Falvey DE (2017) J Org Chem 82:1552
Hammett LP (1937) J Am Chem Soc 59:96
Hansch C, Leo A, Taft RW (1991) Chem Rev 91:165
Gázquez J, Cedillo A, Vela A (2007) J Phys Chem A 11:1966
Chattaraj P, Chakraborty A, Giri S (2009) J Phys Chem A 113:10068
Cremer D, Kraka E (1984) Croat Chem Acta 57:1259
Espinosa E, Alkorta I, Elguero J, Molins E (2002) J Chem Phys 117:5529
Blanco M, Pandás AM, Mckee WC (2014) J Comput Chem 35:1499
Pendas AM, Blanco MA, Francisco E (2007) J Comput Chem 28:161
Blanco MA, Pendas AM, Francisco E (2005) J Chem Theory Comput 1:1096
Francisco E, Pendas AM, Blanco MAA (2006) J Chem Theory Comput 2:90
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalman G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA Jr, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian 09, revision A02. Gaussian Inc, Wallingford
Krishnan R, Binkley JS, Seeger R, Pople JA (1980) J Chem Phys 72:650
Wachters AJH (1970) J Chem Phys 52:1033
Hay PJ (1977) J Chem Phys 66:4377
McLean AD, Chandler GS (1980) J Chem Phys 72:5639
Rappoport D, Furche F (2010) J Chem Phys 133:134105
Parr RG, Yang W (1989) Density-function theory of atoms and molecules. Oxford University Press, Oxford
O’Boyle NM, Tenderholt AL, Langner KM (2008) J Comp Chem 29:839
Lu T, Chen F (2012) J Mol Gr Model 38:314
Lu T, Chen F (2012) J Comp Chem 33:580
Keith TA (2013) AIMAll Version 13.11.04. TK Gristmill Software, Overland Park, KS
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ghiasi, R., Saraf, S.H. & Pasdar, H. Influences of the substituents on the Cr=C bond in [(OC)5Cr=C(OEt)-para-C6H4X] complexes: quantum Theory of Atoms in Molecules, Energy Decomposition Analysis, and Interacting Quantum Atoms. Monatsh Chem 149, 2167–2174 (2018). https://doi.org/10.1007/s00706-018-2299-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-018-2299-3