Abstract
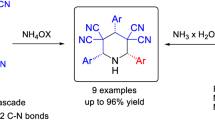
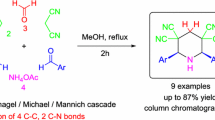
A pseudo-five-component reaction for one-pot synthesis of polysubstituted 2-piperidinones from two equivalent aromatic aldehydes, nitromethane, ammonium acetate, and dialkyl malonates is reported. Interestingly, the formation of products was highly stereoselective. Two different stereochemical classes of polysubstituted 2-piperidinones were provided depending on the substituent position of the aromatic aldehydes.
Graphical abstract



Similar content being viewed by others
References
Mateeva NN, Winfield LL, Redda KK (2005) Curr Med Chem 12:551
Källstrm S, Leino R (2008) Bioorg Med Chem 16:601
Elbein DA, Molyneux R (1987) In: Palletier SW (ed) Alkaloids: chemical and biological perspectives, vol 57. Wiley, New York
O’Hagan D (2000) Nat Prod Rep 17:435
Mortari MR, Schwartz ENF, Schwartz CA, Pires OR Jr, Santos MM, Bloch C Jr, Sebben A (2004) Toxicon 43:303
Chen FE, Huang J (2005) Chem Rev 105:4671
Woodward RB, Bader FE, Bickel H, Frey AJ, Kierstead RW (1958) Tetrahedron 110:5925
Schlitter E (1965) In: Manske RHF (ed) The alkaloids: chemistry and physiology, vol VIII. Academic, New York, p 287
Magnus P, Fielding MR, Wells C, Lynch V (2002) Tetrahedron Lett 43:947
Young IS, Kerr MA (2007) J Am Chem Soc 129:1465
Ono K, Nakagawa M, Nishida A (2004) Angew Chem Int Ed 43:2020
Nagata T, Nakagawa M, Nishida A (2003) J Am Chem Soc 125:7484
Nara S, Tanaka R, Eishima J, Hara M, Takahashi Y, Otaki S, Foglesong RJ, Hughes PF, Turkington S, Kanda Y (2003) J Med Chem 46:2467
Beliah V, Jeyaraman R, Chandrasekaran L (1983) Chem Rev 83:379
Pinder AR (1992) Nat Prod Rep 9:17
Felpin FX, Lebreton J (2004) Tetrahedron 60:10127
Rubiralta M, Giralt E, Diez A (1991) Structure, preparation, reactivity, and synthetic application of piperidine and its derivatives. Elsevier, Amsterdam, p 346
Strack WD (1990) In: Brossi A (ed) The alkaloids, vol 39. Academic, San Diego
Choi IS, Song KS, Hong J, Lee CO, Jung JH (2002) Bull Korean Chem Soc 23:497
Fisyuk AS, Bundel YG (1999) Chem Heterocycl Compd 35:125
Xu F, Corley E, Zacuto M, Conlon DA, Pipik B, Humphrey G, Murry J, Tschaen D (2010) J Org Chem 75:1343
Deiters A, Pettersson M, Martin SF (2006) J Org Chem 71:6547
Garner P, Kaniskan HU, Keyari CM, Weerasinghe L (2011) J Org Chem 76:5283
Tu YS, Duh TH, Tseng CY, Lin YT, Lo YH, Hu YL, Chen CH, Chien CM, Yang SH, Lin SR, Yang SC, Wu MJ (2009) Bio Med Chem 17:7412
Husson HP, Royer J (1999) Chem Soc Rev 28:383
Bates RW, Kanicha SE (2002) Tetrahedron 58:5957
Felpin FX, Lebreton J (2003) Eur J Org Chem 3693
Bailey PD, Millwood PA, Smith PD (1998) Chem Commun 633
Buffat MGP (2004) Tetrahedron 60:1701
Weintraub PM, Sabol JS, Kane JM, Borcherding DR (2003) Tetrahedron 59:2953
Escolano C, Amat M, Bosch J (2006) Chem Eur J 12:8198
Sainte F, Serckx-Poncin B, Frisque-Hesbain AM, Ghosez L (1982) J Am Chem Soc 104:1428
Bayard P, Ghosez L (1988) Tetrahedron Lett 29:6115
Sustmann R, Sicking W, Lamy-Schelkens H, Ghosez L (1991) Tetrahedron Lett 32:1401
Singh V, Yadav GP, Maulik PR, Batra S (2006) Tetrahedron 62:8731
Burdzhiev NT, Stanoeva ER (2006) Tetrahedron 62:8318
Davies SG, Roberts PM, Smith AD (2007) Org Biomol Chem 5:1405
Upadhyay PK, Kumar P (2010) Synthesis 2512
Kumar P, Louie J (2011) Angew Chem Int Ed 50:10768
Katritzky AR, Luo Z, Fang Y, Feng D, Ghi (2000) J Chem Soc Perkin Trans 2:1375
Tejedor D, Garcia-Tellado F (2007) Chem Soc Rev 36:484
Zhu J, Bienayme H (eds) (2005) Multicomponent reactions. Wiley, Weinheim
Orru RVA, Hulme C, Oddon G, Schmitt P (2000) Chem Eur J 6:3321
Domling A, Ugi I (2000) Angew Chem Int Ed 39:3168
Blackwell HE (2006) Curr Opin Chem Biol 10:203
Domling A (2006) Chem Rev 106:17
Brauer S, Almstetter M, Antuch W, Behnke D, Taube R, Furer P, Hess S (2005) J Comb Chem 7:218
Toure BB, Hall DG (2009) Chem Rev 109:4439
Nandaluru PR, Bodwell GJ (2012) Org Lett 14:310
Jiang B, Rajale T, Wever W, Tu S, Li G (2010) Chem Asian J 5:2318
Kiruthika SE, Lakshmi NV, Banu BR, Perumal PT (2011) Tetrahedron Lett 52:6508
Ruijter E, Scheffelaar R, Orru RVA (2011) Angew Chem Int Ed 50:6234
Schreiber SL (2000) Science 287:1964
Groenendaal B, Ruijter E, Orru RVA (2008) Chem Commun 5474
Ismabery N, Lavila R (2008) Chem Eur J 14:8444
Sunderhaus JD, Martin SF (2009) Chem Eur J 15:1300
Jiang B, Shi F, Tu SJ (2010) Curr Org Chem 14:357
Tietze LF, Kinzel T, Brazel CC (2009) Acc Chem Res 42:367
Gonzalez-Zamora E, Fayol A, Bois-Choussy M, Chiaroni A, Zhu J (2001) Chem Commun 1684
Bossharth E, Desbordes P, Monteiro N, Balme G (2003) Org Lett 5:2441
Tour BB, Hoveyda HR, Tailor J, Ulaczyk-Lesanko A, Hall DG (2003) Chem Eur J 9:466
Fayol A, Zhu J (2004) Org Lett 6:115
**ao D, Wang L, Feng X (2005) Synlett 1531
Clarke PA, Zaytzev AV, Whitwood AC (2007) Tetrahedron Lett 48:5209
Godineau E, Landais Y (2007) J Am Chem Soc 129:12662
Clarke PA, Zaytzev AV, Whitwood AC, Wilson C (2008) Org Lett 10:2877
Jakubec P, Helliwell M, Dixon DJ (2008) Org Lett 10:4267
Sarkar N, Banerjee A, Nelson SG (2008) J Am Chem Soc 130:9222
Takasu K, Shindoh N, Tokuyama H, Ihara M (2006) Tetrahedron 62:11900
Bhagwatheeswaran H, Gaur SP, Jain PC (1976) Synthesis 615
Toure BB, Hoveyda HR, Tailor J, Ulaczyk-Lesanko A, Hall DG (2003) Chem Eur J 9:466
Zhu W, Mena M, Jnoff E, Sun N, Pasau P, Ghosez L (2009) Angew Chem Int Ed 48:5880
Ono N (2001) The nitro group in organic synthesis. Wiley, Weinheim
Altug C, Burnett AK, Caner E, Durust Y, Elliott MC, Glanville RPJ, Guy C, Westwell A (2011) Tetrahedron 67:9522
Michaud D, Hamelin J, Texier-Boullet F, Toupet L (2002) Tetrahedron 58:5865
Kshirsagar SW, Patil NR, Samant SD (2010) Tetrahedron Lett 51:2924
Elinson MN, Ilovaisky AI, Merkulova VM, Belyakov PA, Chizhov AO, Nikishin GI (2010) Tetrahedron 66:4043
Adamo MFA, Duffy EF, Donati D, Sarti-Fantoni P (2007) Tetrahedron 63:2047
Liu H, Zhou Z, Sun Q, Li Y, Li Y, Liu J, Yan P, Wang D, Wang C (2012) ACS Comb Sci 14:366
Haasnoot CAG, DeLeeuw FAAM, Altona A (1980) Tetrahedron 36:2783
Acknowledgments
Financial support of this research by the National Natural Science Foundation of China (NNSFC 21173181) is gratefully acknowledged by the authors. This project was funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
706_2012_912_MOESM1_ESM.pdf
Supplementary Material Full characterization data and copies of the 1H and 13C NMR spectra of all compounds as well as crystallographic data for 4a, 4c, and 4y can be found in the Supplementary Material available online. (PDF 4913 kb)
Rights and permissions
About this article
Cite this article
Liu, H., Sun, Q., Zhou, Z. et al. One-pot synthesis of polysubstituted 2-piperidinones from aromatic aldehydes, nitromethane, ammonium acetate, and dialkyl malonates. Monatsh Chem 144, 1031–1041 (2013). https://doi.org/10.1007/s00706-012-0912-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-012-0912-4