Abstract
Background
Joint flexion to diminish the gap and avoid nerve grafts fell into disuse for decades, but recently attention for using this technique was regained. We report a case series of nerve suture under joint flexion, ultrasound monitoring, and physiotherapy. Our main objective was to determine how effective this multimodality treatment is.
Methods
A retrospective review of 8 patients treated with direct repair with joint flexion was done. Depending on the affected nerve, either the knee or the elbow was flexed intraoperatively to determine if direct suturing was possible. After surgery, the limb was held immobilized. Through serial ultrasounds and a physiotherapy program, the limb was fully extended. If a nerve repair rupture was observed, the patient was re-operated and grafts were used.
Results
Of the eight nerve sutures analyzed, four sustained a nerve rupture revealed by US at an early stage, while four did not show any sign of dehiscence. In the patients in whom the nerve suture was preserved, an early and very good response was observed. Ultrasound was 100% accurate at identifying nerve suture preservation. Early detection of nerve failure permitted early re-do surgery using grafts without flexion, ultimately determining good final results.
Conclusions
We observed a high rate of dehiscence in our group of patients treated with direct repair and joint flexion. We believe this was due to an incorrect use of the immobilization device, excessive movement, or a broken device. In opposition to this, we observed that applying direct nerve sutures and joint flexion offers unusually good and fast results. If this technique is employed, it is mandatory to closely monitor suture status with US, together with physiotherapy providing progressive, US-guided extension of the flexed joint. If nerve rupture occurs, the close monitoring dictated by this protocol should ensure the timely application of a successful graft repair.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In 1915, Delorme [4] became the first to describe using joint flexion to diminish the gap between two nerve stumps. His technique was popularized from the First World War through the 1930s, but fell into disuse afterwards. Its detractors maintained that, after the immobilization period, joint extension would place tension again at the suture site and secondary rupture would occur [6]. The later emergence of nerve grafts [14] contributed to the low popularity of Delorme’s technique, though it was never abandoned entirely, and recently has experienced increased use [1, 2]. Recently, we described modifying Delorme’s original technique to perform nerve suture under flexion in a pediatric patient, which included monitoring the nerve repair site with serial ultrasounds (US), a specific, intense physiotherapy program, and use of an articulated orthotic device to gradually extend the flexed joint under US control. Our full and complete protocol for progressive joint extension has already been published, if more precise information is required [20].
In the present paper, we describe a series of eight patients who underwent a direct nerve suture under joint flexion. Our objective was to determine how effective our multimodality treatment was at avoiding the use of grafts, and at obtaining faster and more complete functional nerve recovery. A secondary objective was to determine the sensitivity of US or, if required, magnetic resonance imaging (MRI), for identifying nerve dehiscence requiring either revision or replacement of the nerve suture, the second surgery using nerve grafts.
Methods and materials
Data from eight consecutives lacerating nerve sutures performed under joint flexion between January 2016 and December 2018 were collected prospectively. As an open injury followed by a nerve deficit suggests the presence of nerve section, the procedure was indicated as soon as possible.
For nerve gaps of 6 cm or shorter, the option of performing nerve repair under joint flexion was considered intraoperatively, based upon the length of the nerve gap. Before enrolling each patient, the pros and cons of this type of nerve repair were discussed with both the patient and/or, in pediatric cases, the patient’s parents. All ethics committee standards were respected. Though meticulously described in a previous article [20], a brief description of our nerve repair protocol using joint flexion follows.
After exploration, the diagnosis of complete nerve section was made, in all cases, using either direct visualization, nerve stimulation, or nerve action potentials (NAPs). Nerve stumps were trimmed until healthy tissue was observed, and the remaining nerve gap measured. Both nerve endings also were dissected, trying to leave as minimal a gap as possible. Depending on the affected nerve, either the knee or the elbow was flexed to determine if direct suturing was possible. If this was considered feasible, two initial equatorial 7.0 anchorage stiches were placed, while the suture was completed with separated nylon 9.0 stitches. Hip or shoulder mobilization was performed after the nerve repair and before wound closure, to ensure that these movements would not stretch the nerve post-operatively. After determining the minimum degree of joint flexion at which no tension was induced at the suture line, the limb was held immobilized throughout the remainder of surgery and during cast placement.
After a window was created in the cast 3 weeks after surgery, a baseline ultrasound (US) was performed to confirm nerve repair preservation. Two weeks later, the cast was removed and an articulated orthotic device applied, maintaining the joint (knee or elbow) in its initial degree of flexion. From that moment onward, an intense physiotherapy program was initiated to recover joint flexion and prevent joint stiffness; however, extension was not permitted beyond the critical point, and just joint flexion and not-maximal extension were trained. A second ultrasound was obtained 3 weeks later to confirm not only nerve suture preservation but also vascular flow at the repair site and nerve gliding under joint flexion. If all three previously mentioned requirements were confirmed, from that point onward, always under strict US guidance, extension gradually was increased by altering the degrees of orthotic device flexion in increments of 10° every 3 weeks. This procedure (physiotherapy ultrasound- ≤ 10° joint extension guided by ultrasound) was repeated until either (1) complete joint extension with nerve suture preservation was accomplish or (2) repair site rupture was observed, in which case repeat exploration and repair were indicated, this time using nerve grafts to bridge the gap. In all cases, our physiotherapy program was followed to completeness.
Results
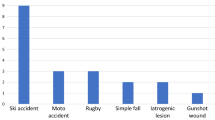
Table 1 provides individual patient demographic and clinical data, and summarizes each patient’s clinical course after treatment. No patients were lost to follow-up. One patient presented with an upper limb nerve injury (radial nerve, #5), while the rest (n = 7) had suffered a lower limb injury, either of the entire sciatic nerve (n = 2) or just its peroneal component (n = 5). All patients were male, and ranged in age from 6 to 42 years old (mean = 21.6 years). Nerve gaps deemed amenable to repair ranged from 2.5 to 6.0 cm (mean 4.8 cm) and the range of joint flexion required for tension-free nerve repair ranged from 30° to 90° (mean = 50°).
Patient #3. A A 12-year-old boy suffered a domestic lacerating injury that completely sectioned his right peroneal nerve at the popliteal fossa, 4 cm distal to the sciatic nerve bifurcation. A foot drop was immediately observed. During surgical exploration, a 4-cm gap was repaired using a direct nerve suture with his knee in 45° flexion, a degree of flexion then maintained with a cast. B An articulated knee orthotic was placed 4 weeks after the surgery, maintaining the same degrees of articular flexion as with the cast. At that time, the nerve suture was evaluated with ultrasound, and was found to be intact. C MRI acquired in coronal plane with reconstruction of the common peroneal nerve showing the proximal (small arrow) and distal (long arrow) pathway, with dehiscence of the neurorraphy and a significant thinning of the common peroneal nerve (arrowhead). D Surgical re-exploration was indicated, and suture rupture detected. E The new gap was 5 cm after trimming the nerve stumps (1 cm longer than during the primary repair). F Sural nerve grafts were employed to bridge the gap. G Two years later, showing M4 recovery of peroneal right function
Patient #4. A 31-year-old male suffered a lacerating injury with a knife to the posterior surface of his distal thigh. A A sciatic nerve gap of 6 cm was identified. B The gap was closed with a direct nerve suture with his knee in 90° of flexion. Fifteen months post-operatively, the patient had recovered foot sensation, as well as plantar and dorsal flexion
Patient #5. A 6-year-old child suffered a lacerating injury to the left arm and elbow after falling from a horse and hitting a wire fence. A A 4-cm nerve gap in the radial nerve was found after trimming the nerve stumps, and a direct suture placed kee** the elbow flexed at 90° for 2 months in a cast. After several follow-up, ultrasounds showed that the repair was intact; progressive elbow extension was performed under US guidance, supervised by his physiotherapist. B The first signs of nerve recovery were present at 3 months, with complete restoration of radial nerve function documented 6 months after the accident
Interestingly, half of our patients (n = 4) sustained with a nerve rupture detected at the second ultrasound that required secondary nerve repair using nerve grafts and therefore avoiding joint flexion. In all four cases, US revealed dehiscence at an early stage (cases #3, 4, 6, and 7, mean time = 5 weeks, range 3–8). In the remaining four patients, in whom the nerve suture was preserved and confirmed by US, an early and very good response (M4 in all cases, starting at 3 to 8 months, mean = 4.25 months) was observed. The adopted imaging method was 100% accurate at identifying nerve suture preservation. Of note, in case #8, peroneal nerve-dependent muscle contraction was observed on US at 2.5 months, 2 weeks before any contraction was observed clinically. The early detection of nerve failure permitted early re-do surgery with grafts (cases #3, 4, and 7) or a tendon transfer (case #6) and, ultimately, obtaining good results in all four patients in whom nerve suture rupture was identified (M4 in two patients, M3 in one, and a tendon transfer in one). Tibialis tendon transfer was indicated in case #6 because the COVID-19 pandemic prevented nerve grafting in a timely enough manner.
The mean follow-up duration was 27.6 months (range = 12–48). In the four patients who experienced suture rupture, either incorrect use of the immobilization device, excessive movement (i.e., accidentally falling), or a broken device was considered the probable cause of dehiscence.
Discussion
This is, to the best of our knowledge, the largest reported series in which modern imaging and adequate physiotherapy were combined to document the results of nerve suturing performed using joint flexion as a way to avoid utilizing nerve grafts to bridge the gap when repairing an injured nerve. Of course, a failure rate of 50% is high, and raises the question: should we keep on using this type of nerve repair?
Suture failure avoidance is perhaps the key-point when using the joint flexion technique for nerve suture. As mentioned in the results, problems related to the misuse of the immobilization device or doing violent movements by the patients should be avoided in order to lower the rate of nerve suture dehiscence to the maximum, or eventually to even zero.
Our series clearly shows the pros and cons that should be considered in the future in order to maintain or abandon this technique.
The first advantage is that, if successful, avoiding nerve grafts generally yields a better result than using a graft, not just in our series but in the literature. The outcomes of nerve grafting are poorer than those of direct sutures, whether the suture is a primary (up to 7 days after trauma) or secondary suture (thereafter) [21]. Obviously, this is because the growing axon needs to transverse just one nerve suture, that being the distal stump of the nerve itself, instead of sutures at both ends of a graft. It has been calculated that the delay required by axons to transverse a suture is approximately 30 days in transected nerves [11, 13, 15]. Hence, direct nerve sutures are associated with quicker arrival of nerve impulses to the target muscle and, consequently, less denervation time and a faster and better final result, relative to classical graft repairs. This is especially important when sensory and motor targets are located at considerable distances from the site of injury, as in sciatic nerve injuries in the buttock or thigh [13]. Also, the regeneration of axons in a mixed nerve into inappropriate pathways is a major contributing factor to poor functional recovery, and this is probably more likely the case with nerve grafts than with direct sutures [3, 11, 13, 15].
Positive results of nerve sutures performed using joint flexion are reported in the literature. Oberlin reported sutures being applied during knee flexion for sciatic nerve repair in a series of 12 patients, with the majority experiencing grade 3 or 4 muscle recovery in flexor, extensor, and fibular muscles [16], while Pfister and Oberlin demonstrated their utility in cadaveric models [18]. Such good results have been replicated by Döring et al. and by Mathieu et al., who have both reported small series of direct sciatic nerve repairs under knee flexion [5, 13]. The favorable results of these series and our own cases are encouraging, compared to the less-satisfactory outcomes reported in previously published series involving nerve grafts [8,9,10,11, 19]. However, since larger series and randomized clinical trials remain elusive, the efficacy and safety of this technique have yet to be proven. In the end, the effects of nerve stretching remain an enigma [12].
A second encouraging finding in our series is that US was so accurate (100% sensitive and specific) for distinguishing nerve suture rupture from preservation, which implies that timely secondary nerve repair with grafts can be assured, providing a good window of time for acceptable recovery of neurological function, even in patients in whom the initial procedure fails (albeit, with outcomes perhaps not quite as good as via direct repair). This was, indeed, observed in the three out of four patients in our series who were able to undergo a secondary surgical procedure in a timely manner, two of whom also ultimately experienced M4-level recovery. The fourth patient with failed direct nerve suture also obtained a good (M4) result with a tendon transfer. Ultrasound is considered the best technique to evaluate the surgical repair of peripheral nerves, particularly with large and superficial nerves [17].
On the other hand, a 50% rate of success with nerve repair is definitely not as high as anyone would like to see. The post-operative care that suturing during joint flexion requires also is much more complex than that generally necessary following classic nerve grafting and requires a highly compliant patient. In fact, all instances of nerve dehiscence in our series were attributed either to a failed orthotic or to inadequate adherence to the prescribed treatment. Recently, another clever—albeit more invasive—way to avoid the problems that we observed in our series has been proposed, entailing bone shortening and using external fixators to slowly overcome bone defects without introducing tension to nerves (Allan Belzberg—personal communication). Other “nerve lengthening “ techniques have recently been described in rats, providing further promising strategies in the mid-term future [7].
Another drawback of the technique we have described is that prolonged immobilization normally produces joint stiffness, which is well tolerated in children and young adults, but might be less suitable for elderly or even middle-aged patients. This said, our oldest patient was 42 years old at the time of surgery and did not experience this problem.
In any case, prior to performing nerve repair under flexion, detailed discussion with the patient and/or the patient’s parent/caregivers is required, explaining both the advantages and disadvantages of this approach and the compliance that is needed both from them and from the therapeutic team, if a nerve suture under flexion technique is to be used. It is quite important that the patient and its family understand the importance. In our opinion, this technique could be used in well-selected patients, as its final results are quite good, probably much better than those achieved with classical graft nerve repair. The higher cost of post-operative care also should be explained [20]. Ultimately, full compliance with all aspects of the post-operative protocol is necessary if nerve rupture at the suture site is to be avoided. If such compliance cannot be ascertained, some other approach may be more prudent.
Conclusions
Our main conclusion is that applying nerve sutures during joint flexion to avoid using grafts has a high rate of nerve dehiscence, but offers unusually good and fast results, in terms of neurological outcomes, when it is successful. If this approach is selected for use, it is mandatory that ultrasound also be employed to closely monitor suture status, together with physiotherapy providing progressive, incremental, US-guided extension of the flexed joint to protect the neurorrhaphy site. If nerve rupture occurs, the close monitoring dictated by this protocol should ensure the timely application of successful graft repair. A high degree of compliance is required, both from patients/parents and from the therapeutic team, if nerve dehiscence rates are to be avoided below what is published herein. Further studies with larger numbers of patients and variations in immobilization techniques could provide more definitive conclusions in the future about this technique’s role in nerve repair surgery.
References
Abou-Al-Shaar H, Yoon N, Mahan MA (2018) Surgical repair of sciatic nerve traumatic rupture: technical considerations and approaches. Neurosurg Focus 44(VideoSuppl1):V3
Addas BMJ, Midha R (2013) Técnica quirúrgica. Reparación del defecto: ¿Injertos nerviosos o tubos artificiales? M Socolovsky M Siqueira M Malessy Eds Introd. Cir. Los Nerv. Periféricos. Journal, Argentina, pp 101–113
Al-Majed AA, Neumann CM, Brushart TM, Gordon T (2000) Brief electrical stimulation promotes the speed and accuracy of motor axonal regeneration. J Neurosci Off J Soc Neurosci 20(7):2602–2608
Delorme E (1915) The treatment of gunshot wounds of nerves. Brit Med J 1(2837):853–855
Döring R, Ciritsis B, Giesen T, Simmen H-P (2012) Giovanoli P (2012) Direct nerve suture and knee immobilization in 90° flexion as a technique for treatment of common peroneal, tibial and sural nerve injuries in complex knee trauma. J Surg Case Rep 12:rjs019
Highet WB, Sanders FK (1943) The effects of stretching nerves after suture. Brit J Surg 30:355–369
Howarth HM, Kadoor A, Salem R, Nicolds B, Adachi S, Kanaris A, Lovering RM, Brown JM, Shah SB (2019) Nerve lengthening and subsequent end-to-end repair yield more favourable outcomes compared with autograft repair of rat sciatic nerve defects. J Tissue Eng Regen Med 13(12):2266–2278
Kallio PK, Vastamäki M (1993) An analysis of the results of late reconstruction of 132 median nerves. J Hand Surg Edinb Scotl 18(1):97–105
Kim DH, Murovic JA, Tiel R, Kline DG (2004) Management and outcomes in 353 surgically treated sciatic nerve lesions. J Neurosurg 101(1):8–17
Lee SK, Wolfe SW (2000) Peripheral nerve injury and repair. J Am Acad Orthop Surg 8(4):243–252
Mackinnon S (2015) Nerve surgery, 1st edn. Thieme Medical Publishers Inc., New York
Mahan MA (2019) Nerve stretching: a history of tension. J Neurosurg 132(1):252–259
Mathieu L, Pfister G, Murison JC, Oberlin C, Belkheyar Z (2019) Missile injury of the sciatic nerve: observational study supporting early exploration and direct suture with flexed knee. Mil Med 184(11–12):e937–e944
Millesi H (1967) Nerve transplantation for reconstruction of peripheral nerves injured by the use of the microsurgical technic. Minerva Chir 22(17):950–951
Millesi H, Schmidhammer R (2007) How to improve the results of peripheral nerve surgery. Springer
Oberlin C, Rantissi M (2011) Gunshot injuries to the nerves. Chir Main 30(3):176–182
Peer S, Bodner G (2008) High-resolution sonography of the peripheral nervous system, 2nd, revised. Springer, Frankfurt
Pfister G, Ghabi A, de Carbonnières A, Oberlin C, Belkheyar Z, Mathieu L (2020) Direct suturing of sciatic nerve defects in high-degree knee flexion: an experimental study. World Neurosurg 133:e288–e292
Roganovic Z (2005) Missile-caused complete lesions of the peroneal nerve and peroneal division of the sciatic nerve: results of 157 repairs. Neurosurgery 57(6):1201–1212 (discussion 1201-1212)
Socolovsky M, Bataglia D, Barousse R, Robla-Costales J (2018) Use of ultrasound and targeted physiotherapy in the management of a nerve suture performed under joint flexion. Acta Neurochir (Wien) 160(8):1597–1601
Yang M, Rawson JL, Zhang EW, Arnold PB, Lineaweaver W, Zhang F (2011) Comparisons of outcomes from repair of median nerve and ulnar nerve defect with nerve graft and tubulization: a meta-analysis. J Reconstr Microsurg 27(8):451–460
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
All research procedures described in this paper were in accordance with the ethical standards of the institutional and/or national research committee (University of Buenos Aires School of Medicine) and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Consent to participate
Informed consent was obtained from all patients enrolled in the current study.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Comments
This paper by Mariano Socolovsky and colleagues highlights the use of direct nerve repair (instead of more common nerve grafting) when surgeons are faced with a sizeable nerve gap. These authors flexed the knee/elbow joint to gain length and resolve the nerve gap; performed a direct repair without tension in that position; protected the limb post-operatively by immobilizing it in flexion; and imaged the patients with ultrasound (US) as the limb was gradually straightened over time. The advantages of performing a nerve repair across a single suture line rather than reconstructing across 2 suture lines include the possibility of an earlier, better outcome without the morbidity of nerve grafts. This technique of direct nerve repair with joint immobilization has been performed for more than a century, but has been revitalized in the past decade. A recent publication with an embedded video nicely illustrates intraoperatively the impressive amount of nerve excursion with passive joint flexion (1). A parallel approach to making up sizeable nerve gaps has been applied by orthopedic surgeons for years—bony shortening by itself or combined with sequential lengthening (2) (note the expanding practice of distraction osteogenesis has led to useful information about nerve elongation). Each of these techniques has obvious challenges and opportunities.
This case series provides additional follow-up on this technique and algorithm which were previously presented initially in a case report (3). The addition of US to monitor the repairs post-operatively expands our knowledge about this subject. Four of 8 patients were found to have a suture line rupture approximately 1 month after repair, noted on the 2nd US study during the straightening phase of the protection protocol (following a normal 1st US study). Without imaging, the rupture would not have been detected and the primary repair by definition would have failed. In contrast, these 4 patients underwent further reconstruction (3 with timely nerve grafting and 1 by tendon transfer by itself done due to a delay imposed by the COVID pandemic).
The final results in all patients of this paper are admirable: those that had direct repair and those that underwent secondary nerve grafting/tendon transfer. All patients regained M3 or 4 dorsiflexion/wrist extension. While many patients were young and the repairs were done relatively distally (closer to the muscle end organs), good results with peroneal nerve repairs/grafts in any circumstance are difficult to achieve.
This paper obviously is not intended to answer the question about whether the approach of direct repair (with a flexed joint) is superior to nerve grafting. Still, despite the small series, there was some evidence of an earlier, better response in the direct suture group. Further studies will need to help optimize the repair: i.e., timing of surgery, maximal nerve gap, type and number of sutures, use of fibrin glue, total time of and method of immobilization (e.g., casting, splinting, locked brace, external fixation) not to mention the time of protected mobilization and the rate and angle of straightening.
Documentation of the high failure rate of the direct repair is extremely valuable. The use of post-operative US in this setting in the surveillance of suture line integrity will become part of my practice. Suture line rupture likely explains some of the cases with poor results reported by others with this technique. Importantly, this complication is correctable if addressed in a timely fashion.
This paper underscores the advantages of cross-fertilization of ideas and strategies to improve nerve regeneration across fields. A multidisciplinary team might benefit from a biomedical engineer: to translate the nerve tensionmeter from the research lab to the operating room. Kudos to my friends and colleagues who are stretching our field to its limits!
Robert J. Spinner,.
Rochester, MN, USA.
References
1. Abou-Al-Shaar H, Yoon N, Mahan, MA. Surgical repair of sciatic nerve traumatic rupture: technical considerations and approaches. Neurosurg Focus 2018 Jan; 44(Video Suppl1):V3.
2. Belzberg AJ, Osggod G. Novel approach to nerve repair and neural regeneration. Spine Summit 2022, Las Vegas, NV, Feb 26, 2022.
3. Socolovsky M, Bataglia D, Barousse R, Robla-Costales J. Use of ultrasound and targeted physiotherapy in the management of a nerve suture performed under joint flexion. Acta Neurochir (Wien) 2018;160(8):1597–1601.
This article is part of the Topical Collection on Peripheral Nerves
Rights and permissions
About this article
Cite this article
Socolovsky, M., Bataglia, D., Barousse, R. et al. Use of ultrasound and targeted physiotherapy to manage nerve sutures placed under joint flexion: a case series. Acta Neurochir 164, 1329–1336 (2022). https://doi.org/10.1007/s00701-022-05195-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-022-05195-w