Abstract
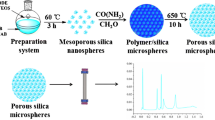
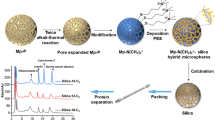
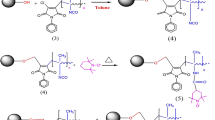
A process-simplified hard template approach was established to synthesize the monodisperse macroporous silica microspheres with homogeneous structures by twice alkali-thermal treatment and calcination routes. Porous vinyl-functionalized polysesquioxane microspheres (V-PMSQ) were synthesized through a hydrolyzation–polycondensation method and used as templates. The template particles with large aperture and high pore volume were obtained by adjusting the pH value and reaction time of the twice alkali-thermal reaction. After calcination, monodisperse silica microspheres with an average pore size of 30 nm, homogeneous pore structures, and narrow particle size distribution were fabricated, which can be directly used as chromatographic matrices without classification. After that, a new reversed-phase/strong anion-exchange (RP/SAX) mixed-mode stationary phase Sil-S-VOIM was prepared by bonding the 1-vinyl-3-octyl-imidazole ligands to the above silica microspheres through a “thiol-ene” click reaction. The performance of the Sil-S-VOIM column was evaluated by one acidic protein (transferrin) and two basic proteins (lysozyme, α-chymotrypsin) and compared to a single imidazole-modified Sil-S-VIM column and an octyl-modified Sil-C8 column, respectively. Due to the synergistic effect of electrostatic repulsion and hydrophobic interactions, baseline separations of the above proteins were observed only on the Sil-S-VOIM column, with resolutions of 2.55 and 2.01 between lysozyme and transferrin, and between transferrin and α-chymotrypsin, respectively, indicating good selectivity and separation ability compared with single-mode stationary phases. It was applied to the isolation of egg white samples with peaks identified by SDS-PAGE and MALDI-TOF–MS. The results showed that the selective retention and isolation of ovomucoid and ovotransferrin were successfully achieved, with yields of 78.8% and 67.2%, respectively. The protocol described in this work is simpler, faster, and has higher protein recovery. Overall, this new mixed-mode stationary phase provided a promising potential for the separation and determination of intact proteins.
Graphical Abstract




Similar content being viewed by others
References
Ashfaq A, Jahan K, Ul Islam R, Younis K (2022) Protein-based functional colloids and their potential applications in food: a review. LWT 154:112667. https://doi.org/10.1016/j.lwt.2021.112667
Liu ZY, Li HL, ** ZY, Li Y, Guo FF, He YZG, Liu XY, Section YNQ, Yuan LY, He FC, Li D (2022) Exploration of target spaces in the human genome for protein and peptide drugs. Genomics Proteomics Bioinf 20:780–794. https://doi.org/10.1016/j.gpb.2021.10.007
Stie MB, Kalouta K, Vetri V, Fodera V (2022) Protein materials as sustainable non- and minimally invasive strategies for biomedical applications. J Controlled Release 344:12–25. https://doi.org/10.1016/j.jconrel.2022.02.016
Li D, Liu C (2021) Hierarchical chemical determination of amyloid polymorphs in neurodegenerative disease. Nat Chem Biol 17:237–245. https://doi.org/10.1038/s41589-020-00708-z
Razi SM, Fahim H, Amirabadi S, Rashidinejad A (2023) An overview of the functional properties of egg white proteins and their application in the food industry. Food Hydrocoll 135:108183. https://doi.org/10.1016/j.foodhyd.2022.108183
Ma XJ, Liang R, Yang XT, Gou JK, Li Y, Lozano-Ojalvo DI (2020) Simultaneous separation of the four major allergens of hen egg white. J Chromatogr B 1152:122231. https://doi.org/10.1016/j.jchromb.2020.122231
Abeyrathne E, Lee HY, Ahn DU (2013) Egg white proteins and their potential use in food processing or as nutraceutical and pharmaceutical agents-a review. Poultry Sci 92:3292–3299. https://doi.org/10.3382/ps.2013-03391
Huang H, Dong X, Sun Y, Shi Q (2023) Biomimetic affinity chromatography for antibody purification: host cell protein binding and impurity removal. J Chromatogr A 1707:464305. https://doi.org/10.1016/j.chroma.2023.464305
Mohsin AZ, Sukor R, Selamat J, Hussin ASM, Ismail IH, Jambari NN, Jonet A (2020) A highly selective two-way purification method using liquid chromatography for isolating alpha(S2)-casein from goat milk of five different breeds. J Chromatogr B 1160:122380. https://doi.org/10.1016/j.jchromb.2020.122380
Staub A, Zurlino D, Rudaz S, Veuthey JL, Guillarme D (2011) Analysis of peptides and proteins using sub-2 μm fully porous and sub 3-μm shell particles. J Chromatogr A 1218:8903–8914. https://doi.org/10.1016/j.chroma.2011.07.051
Kothalawala SG, Jiao JQ, Speight R, Song H, Yang YN, Zhang J (2022) Pore architecture influences the enzyme immobilization performance of mesoporous silica nanospheres. Microporous Mesoporous Mater 338:111963. https://doi.org/10.1016/j.micromeso.2022.111963
Sulman AM, Haskell AK, Tikhonov BB, Grebennikova OV, Sidorov AI, Bronstein LM, Matveeva VG (2022) Larger pores dramatically enhance activity of an immobilized enzyme in mesoporous magnetic silica. Microporous Mesoporous Mater 341:112092. https://doi.org/10.1016/j.micromeso.2022.112092
Li BJ, Zou XY, Zhao YB, Sun L, Li SL (2013) Biofunctionalization of silica microspheres for protein separation. Mater Sci Eng C 33:2595–2600. https://doi.org/10.1016/j.msec.2013.02.030
Chen JW, Zhu LL, Ren LB, Teng C, Wang Y, Jiang BW, He J (2018) Fabrication of monodisperse porous silica microspheres with a tunable particle size and pore size for protein separation. ACS Appl Bio Mater 1:604–612. https://doi.org/10.1021/acsabm.8b00088
Fait F, Steinbach JC, Kandelbauer A, Mayer HA (2023) Impact of porosity and surface functionalization of hard templates on the preparation of mesoporous silica microspheres. Microporous Mesoporous Mater. 351:112482. https://doi.org/10.1016/j.micromeso.2023.112482
Fekete S, Veuthey JL, Guillarme D (2012) New trends in reversed-phase liquid chromatographic separations of therapeutic peptides and proteins: theory and applications. J Pharm Biomed Anal 69:9–27. https://doi.org/10.1016/j.jpba.2012.03.024
Hayrapetyan SS, Khachatryan HG (2005) Wide-porosity silicas for high-performance liquid chromatography. Acta Chromatogr 15:162–172. https://www.researchgate.net/publication/267264740_Wide-porosity_silicas_for_high-performance_liquid_chromatography
Cheng LP, Cai JF, Ke YX (2019) Synthesis of large-pore silica microspheres using dodecylamine as a catalyst, template and porogen agent. J Inorg Organomet Polym Mater 29:1417–1421. https://doi.org/10.1007/s10904-019-01086-3
Belbekhouche S, Poostforooshan J, Shaban M, Ferrara B, Alphonse V, Cascone I, Bousserrhine N, Courty J, Weber AP (2020) Fabrication of large pore mesoporous silica microspheres by salt-assisted spray-drying method for enhanced antibacterial activity and pancreatic cancer treatment. Int J Pharm 590:119930. https://doi.org/10.1016/j.ijpharm.2020.119930
Wei JX, Shi ZG, Chen F, Feng YQ, Guo QZ (2009) Synthesis of penetrable macroporous silica spheres for high-performance liquid chromatography. J Chromatogr A 1216:7388–7393. https://doi.org/10.1016/j.chroma.2009.04.066
Marques AC, Vale M (2021) Macroporosity control by phase separation in sol-gel derived monoliths and microspheres. Materials 14:4247. https://doi.org/10.3390/ma14154247
Haynes T, Bougnouch O, Dubois V, Hermans S (2020) Preparation of mesoporous silica nanocapsules with a high specific surface area by hard and soft dual templating approach: application to biomass valorization catalysis. Microporous Mesoporous Mater 306:110400. https://doi.org/10.1016/j.micromeso.2020.110400
**g Y, Guo X, Qi CD, Chen L (2022) Fabrication of silica microspheres for HPLC packing with narrow particle size distribution and different pore sizes by hard template method for protein separation. Chromatographia 85:985–995. https://doi.org/10.1007/s10337-022-04200-9
Bai J, Zhu Q, Tang C, Liu J, Yi Y, Bai Q (2022) Synthesis and application of 5 μm monodisperse porous silica microspheres with controllable pore size using polymeric microspheres as templates for the separation of small solutes and proteins by high-performance liquid chromatography. J Chromatogr A 1675:463165. https://doi.org/10.1016/j.chroma.2022.463165
Tankrathok A, Daduang S, Patramanon R, Araki T, Thammasirirak S (2009) Purification process for the preparation and characterizations of hen egg white ovalbumin, lysozyme, ovotransferrin, and ovomucoid. Prep Biochem Biotechnol 39:380–399. https://doi.org/10.1080/10826060903209646
Omana DA, Wang JP, Wu JP (2010) Co-extraction of egg white proteins using ion-exchange chromatography from ovomucin-removed egg whites. J Chromatogr B 878:1771–1776. https://doi.org/10.1016/j.jchromb.2010.04.037
Santarelli X, Cabanne C (2019) Mixed mode chromatography: a novel way toward new selectivity. Curr Protein Pept Sci 20:14–21. https://doi.org/10.2174/1389203718666171024121137
Huo ZX, Chen L (2020) Base-deactivated and alkaline-resistant chromatographic stationary phase based on functionalized polymethylsilsesquioxane microspheres. J Sep Sci 43:389–397. https://doi.org/10.1002/jssc.201900634
Huo ZX, Wan QH, Chen L (2020) Energy-efficient and environment-friendly method to prepare monodispersed silica stationary phases for simultaneous separation of compound drugs. J Chromatogr A 1618:460866. https://doi.org/10.1016/j.chroma.2020.460866
Li JL, Huo ZX, Chen L, Wan QH (2017) Mercaptopropyl functionalized polymethylsilsesquioxane microspheres prepared by co-condensation method as organosilica-based chromatographic packings. Chromatographia 80:1287–1297. https://doi.org/10.1007/s10337-017-3349-4
Soliven A, Dennis GR, Guiochon G, Hilder EF, Haddad PR, Shalliker RA (2010) Cyano bonded silica monolith-development of an in situ modification method for analytical scale columns. J Chromatogr A 1217:6085–6091. https://doi.org/10.1016/j.chroma.2010.07.052
Chen M, Chen L (2018) A novel high hydrothermal stability amino-functionalized stationary phase prepared by a vapour deposition method. Anal Methods 10:1538–1546. https://doi.org/10.1039/c8ay00133b
Geng F, Huang Q, Wu XF, Ren GD, Shan YY, ** GF, Ma MH (2012) Co-purification of chicken egg white proteins using polyethylene glycol precipitation and anion-exchange chromatography. Sep Purif Technol 96:75–80. https://doi.org/10.1016/j.seppur.2012.05.021
Funding
This work was supported by the Tian** Graduate Student Research and Innovation Project (2021YJSB194).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Guo, X., Bai, H. & Chen, L. Imidazole-octyl mixed-mode stationary phase based on macroporous silica for the purification of ovomucoid and ovotransferrin. Microchim Acta 190, 404 (2023). https://doi.org/10.1007/s00604-023-05986-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-023-05986-7