Abstract
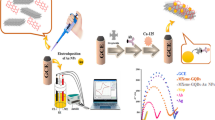
We report on the modification of a graphene paste electrode with gold nanoparticles (AuNPs) and a Nafion-L-cysteine composite film, and how this electrode can serve as a platform for the construction of a novel electrochemical immunosensor for the detection of hepatitis B surface antigen (HBsAg). To obtain the immunosensor, an antibody against HBsAg was immobilized on the surface of the electrode, and this process was followed by cyclic voltammetry and electrochemical impedance spectroscopy. The peak currents of a hexacyanoferrate redox system decreased on formation of the antibody-antigen complex on the surface of the electrode. Then increased electrochemical response is thought to result from a combination of beneficial effects including the biocompatibility and large surface area of the AuNPs, the high conductivity of the graphene paste electrode, the synergistic effects of composite film, and the increased quantity of HBsAb adsorbed on the electrode surface. The differential pulse voltammetric responses of the hexacyanoferrate redox pair are proportional to the concentration of HBsAg in the range from 0.5–800 ng mL−1, and the detection limit is 0.1 ng mL−1 (at an S/N of 3). The immunosensor is sensitive and stable.

We report on the modification of a graphene paste electrode with gold nanoparticles and a Nafion-L-cysteine composite film, and how this electrode can serve as a platform for the construction of a novel electrochemical immunosensor for the detection of hepatitis B surface antigen. The immunosensor is sensitive and stable.






Similar content being viewed by others
References
Gitlin N (1997) Hepatitis B: diagnosis, prevention, and treatment. Clin Chem 43:1500
Badur S, Akgun A (2001) Diagnosis of hepatitis B infections and monitoring of treatment. J Clin Virol 21:229
Tong P, Tang SR, He Y, Shao YH, Zhang L, Chen GN (2011) Label-free immunosensing of microcystin-LR using a gold electrode modified with gold nanoparticles. Microchim Acta 173:299
Yang MH, Li H, Javadi A, Gong SQ (2010) Multifunctional mesoporous silica nanoparticles as labels for the preparation of ultrasensitive electrochemical immunosensors. Biomaterials 31:3281
Zhou L, Ou LJ, Chu X, Shen GL, Yu RQ (2007) Aptamer-based rolling circle amplification: a platform for electrochemical detection of protein. Anal Chem 79:7492
Liu B, Lu LS, Li Q, **e GM (2011) Disposable electrochemical immunosensor for myeloperoxidase based on the indium tin oxide electrode modified with an ionic liquid composite film containing gold nanoparticles, poly(o-phenylenediamine) and carbon nanotubes. Microchim Acta 173:513
Wang J, Meng WY, Zheng XF, Liu SL, Li GX (2009) Combination of aptamer with gold nanoparticles for electrochemical signal amplification: application to sensitive detection of platelet-derived growth factor. Biosens Bioelectron 24:1598
Shan C, Yang H, Han D, Zhang Q, Ivaska A, Niu L (2010) Graphene/AuNPs/chitosan nanocomposites film for glucose biosensing. Biosens Bioelectron 25:1070
Gan T, Hu SS (2011) Electrochemical sensors based on graphene materials. Microchim Acta 175:1
Zhou M, Zhai YM, Dong SJ (2009) Electrochemical sensing and biosensing platform based on chemically reduced graphene oxide. Anal Chem 81:5603
Yan L, Yang WK, Fan MQ, Liu A (2011) A Sensitive Label-free Amperometric CEA Immunosensor Based on Graphene-Nafion Nanocomposite Film as an Enhanced Sensing Platform. Anal Sci 27:727
Sun JY, Huang KJ, Zhao SF, Fan Y, Wu ZW (2011) Direct electrochemistry and electrocatalysis of hemoglobin on chitosan-room temperature ionic liquid-TiO2-graphene nanocomposite film modified electrode. Biochem 82:125
Zhou KF, Zhu YH, Yang XL, Li CZ (2010) Electrocatalytic Oxidation of Glucose by the Glucose Oxidase Immobilized in Graphene-Au-Nafion Biocomposite. Electroanalysis 22:259
Zhuo Y, Yuan R, Chai YQ, Tang DP, Zhang Y, Wang N, Li XL, Zhu Q (2005) A reagentless amperometric immunosensor based on gold nanoparticles/thionine/Nafion-membrane-modified gold electrode for determination of [alpha]-1-fetoprotein. Electrochem Commun 7:355
Li XL, Yuan R, Chai YQ, Zhang LY, Zhuo Y, Zhang Y (2006) Amperometric immunosensor based on toluidine blue/nano-Au through electrostatic interaction for determination of carcinoembryonic antigen. J Biotechnol 123:356
Valentini F, Amine A, Orlanducci S, Terranova ML, Palleschi G (2003) Carbon nanotube purification: preparation and characterization of carbon nanotube paste electrodes. Anal Chem 75:5413
Wang J, Musameh M, Mo JW (2006) Acid stability of carbon paste enzyme electrodes. Anal Chem 78:7044
Wang SF, **ong HY, Zeng QX (2007) Design of carbon paste biosensors based on the mixture of ionic liquid and paraffin oil as a binder for high performance and stabilization. Electrochem Commun 9:807
Hummers W, Offeman R (1958) RE Preparation of graphitic oxide. J Am Chem Soc 80:1339
Jiang Y, Zhao H, Zhu NN, Lin YQ, Yu P, Mao LQ (2008) A simple assay for direct colorimetric visualization of trinitrotoluene at Picomolar levels using gold nanoparticles. Angew Chem Int Ed 47:8601
Brownson DAC, Banks CE (2011) Graphene electrochemistry: surfactants inherent to graphene inhibit metal analysis. Electrochem Commun 13:111
Wang J, Cao Y, Xu YY, Li GX (2009) Colorimetric multiplexed immunoassay for sequential detection of tumor markers. Biosens Bioelectron 25:532
Yuan R, Tang D, Chai Y, Zhong X, Liu Y, Dai J (2004) Ultrasensitive potentiometric immunosensor based on SA and OCA techniques for immobilization of HBsAb with colloidal Au and polyvinyl butyral as matrixes. Langmuir 20:7240
Tang D, Yuan R, Chai Y, Zhong X, Liu Y, Dai J (2004) A novel immunosensor based on immobilization of hepatitis B surface antibody on platinum electrode modified colloidal gold and polyvinyl butyral as matrices via electrochemical impedance spectroscopy. Bioelectrochem 65:15
Liang R, Peng H, Qiu J (2008) Fabrication, characterization, and application of potentiometric immunosensor based on biocompatible and controllable three-dimensional porous chitosan membranes. J Colloid Interface Sci 320:125
Zhuo Y, Yuan R, Chai Y, Li X, Zhang Y, Zhu Q, Wang N (2005) An amperometric immunosensor based on immobilization of hepatitis B surface antibody on gold electrode modified gold nanoparticles and horseradish peroxidase. Anal Chim Acta 548:205
Tang DY, **a BY (2008) Electrochemical immune bioassay for the antigen-antibody interaction based on [Fe (CN) 6] 4-/3-and [AuCl 4]-ions-derivated biomimetic interface. Ionics 14:329
Fu XH, Pu L, Wang JY, Zhong ZY (2010) Ionic liquids-doped organic–inorganic hybrid film for electrochemical immunoassay for hepatitis B surface antigen. Ionics 16:51
Qiu JD, Huang H, Liang RP (2011) Biocompatible and label-free amperometric immunosensor for hepatitis B surface antigen using a sensing film composed of poly(allylamine)-branched ferrocene and gold nanoparticles. Microchim Acta 174:97
Acknowledgments
This work was supported by the National Natural Science Foundation of China (20805040), Program for Science & Technology Innovation Talents in Universities of Henan Province (2010HASTIT025), Excellent Youth Foundation of He’nan Scientific Committee (104100510020).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Huang, KJ., Li, J., Liu, YM. et al. Disposable immunoassay for hepatitis B surface antigen based on a graphene paste electrode functionalized with gold nanoparticles and a Nafion-cysteine conjugate. Microchim Acta 177, 419–426 (2012). https://doi.org/10.1007/s00604-012-0805-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00604-012-0805-6