Abstract
Background
Accumulating evidence shows an overabundance of Fusobacterium nucleatum in colorectal tumor tissues. However, the correlation between the absolute copy number of F. nucleatum in colorectal cancer tissues and colorectal cancer progression is unclear from previous reports. Therefore, we performed a study to compare the abundance of F. nucleatum in colorectal tissues with clinicopathologic and molecular features of colorectal cancer.
Methods
We collected 100 colorectal cancer tissues and 72 matched normal-appearing mucosal tissues. Absolute copy numbers of F. nucleatum were measured by droplet digital PCR.
Results
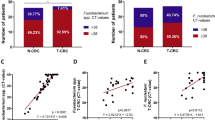
The detection rates of F. nucleatum were 63.9% (46/72) in normal-appearing mucosal tissues and 75.0% (75/100) in CRC tissue samples. The median copy number of F. nucleatum was 0.4/ng DNA in the normal-appearing colorectal mucosa in patients with colorectal cancer and 1.9/ng DNA in the colorectal cancer tissues (P = 0.0031). F. nucleatum copy numbers in stage IV colorectal cancer tissues were significantly higher than those in the normal-appearing mucosa in patients with colorectal cancer (P = 0.0016). The abundance of F. nucleatum in colorectal cancer tissues correlated with tumor size and KRAS mutation and was significantly associated with shorter overall survival times; this trend was notable in the patients with stage IV colorectal cancer. Focusing on normal-appearing mucosa in the patients with colorectal cancer, the F. nucleatum copy number was significantly higher in the patients with stage IV rather than stages I–III.
Conclusion
These results suggest that determining F. nucleatum levels may help predict clinical outcomes in colorectal cancer patients. Further confirmatory studies using independent datasets are required to confirm our findings.





Similar content being viewed by others
Abbreviations
- CRC:
-
Colorectal cancer
- Fn:
-
Fusobacterium nucleatum
- ROC:
-
Receiver-operating characteristic
References
Rubinstein MR, Wang X, Liu W, et al. Fusobacterium nucleatum promotes colorectal carcinogenesis by modulating E-cadherin/beta-catenin signaling via its FadA adhesin. Cell Host Microbe. 2013;14:195–206.
Flanagan L, Schmid J, Ebert M, et al. Fusobacterium nucleatum associates with stages of colorectal neoplasia development, colorectal cancer and disease outcome. Eur J Clin Microbiol Infect Dis. 2014;33:1381–90.
Tahara T, Yamamoto E, Suzuki H, et al. Fusobacterium in colonic flora and molecular features of colorectal carcinoma. Cancer Res. 2014;74:1311–8.
Ito M, Kanno S, Nosho K, et al. Association of Fusobacterium nucleatum with clinical and molecular features in colorectal serrated pathway. Int J Cancer. 2015;137:1258–68.
Abed J, Emgard JE, Zamir G, et al. Fap2 mediates Fusobacterium nucleatum colorectal adenocarcinoma enrichment by binding to tumor-expressed Gal-GalNAc. Cell Host Microbe. 2016;20:215–25.
Li YY, Ge QX, Cao J, et al. Association of Fusobacterium nucleatum infection with colorectal cancer in Chinese patients. World J Gastroenterol. 2016;22:3227–33.
Michaud DS. Role of bacterial infections in pancreatic cancer. Carcinogenesis. 2013;34:2193–7.
Michaud DS, Izard J, Wilhelm-Benartzi CS, et al. Plasma antibodies to oral bacteria and risk of pancreatic cancer in a large European prospective cohort study. Gut. 2013;62:1764–70.
Signat B, Roques C, Poulet P, et al. Fusobacterium nucleatum in periodontal health and disease. Curr Issues Mol Biol. 2011;13:25–36.
Ohkusa T, Okayasu I, Ogihara T, et al. Induction of experimental ulcerative colitis by Fusobacterium varium isolated from colonic mucosa of patients with ulcerative colitis. Gut. 2003;52:79–83.
Minami M, Ando T, Okamoto A, et al. Seroprevalence of Fusobacterium varium in ulcerative colitis patients in Japan. FEMS Immunol Med Microbiol. 2009;56:67–72.
Strauss J, Kaplan GG, Beck PL, et al. Invasive potential of gut mucosa-derived Fusobacterium nucleatum positively correlates with IBD status of the host. Inflamm Bowel Dis. 2011;17:1971–8.
Brook I, Frazier EH. Microbiological analysis of pancreatic abscess. Clin Infect Dis. 1996;22:384–5.
Shahani L, Khardori N. Fusobacterium necrophorum–beyond Lemierres syndrome. BMJ Case Rep. 2011.
Yoneda M, Kato S, Mawatari H, et al. Liver abscess caused by periodontal bacterial infection with Fusobacterium necrophorum. Hepatol Res. 2011;41:194–6.
Athavale NV, Leitch DG, Cowling P. Liver abscesses due to Fusobacterium spp that mimic malignant metastatic liver disease. Eur J Clin Microbiol Infect Dis. 2002;21:884–6.
Kostic AD, Gevers D, Pedamallu CS, et al. Genomic analysis identifies association of Fusobacterium with colorectal carcinoma. Genome Res. 2012;22:292–8.
Castellarin M, Warren RL, Freeman JD, et al. Fusobacterium nucleatum infection is prevalent in human colorectal carcinoma. Genome Res. 2012;22:299–306.
Kostic AD, Chun E, Robertson L, et al. Fusobacterium nucleatum potentiates intestinal tumorigenesis and modulates the tumor-immune microenvironment. Cell Host Microbe. 2013;14:207–15.
Han YW, Ikegami A, Rajanna C, et al. Identification and characterization of a novel adhesin unique to oral fusobacteria. J Bacteriol. 2005;187:5330–40.
Xu M, Yamada M, Li M, et al. FadA from Fusobacterium nucleatum utilizes both secreted and nonsecreted forms for functional oligomerization for attachment and invasion of host cells. J Biol Chem. 2007;282:25000–9.
Ikegami A, Chung P, Han YW. Complementation of the fadA mutation in Fusobacterium nucleatum demonstrates that the surface-exposed adhesin promotes cellular invasion and placental colonization. Infect Immun. 2009;77:3075–9.
Human Microbiome Project C. Structure, function and diversity of the healthy human microbiome. Nature. 2012;486:207–14.
Dewhirst FE, Chen T, Izard J, et al. The human oral microbiome. J Bacteriol. 2010;192:5002–17.
Faust K, Sathirapongsasuti JF, Izard J, et al. Microbial co-occurrence relationships in the human microbiome. PLoS Comput Biol. 2012;8:e1002606.
Ashare A, Stanford C, Hancock P, et al. Chronic liver disease impairs bacterial clearance in a human model of induced bacteremia. Clin Transl Sci. 2009;2:199–205.
Hindson BJ, Ness KD, Masquelier DA, et al. High-throughput droplet digital PCR system for absolute quantitation of DNA copy number. Anal Chem. 2011;83:8604–10.
Sobin LH, Gospodarowicz MK, Wittekind C, editors. TNM classification of malignant tumours. 7th ed. Oxford: Wiley; 2009.
Suehiro Y, Sakai K, Nishioka M, et al. Highly sensitive stool DNA testing of Fusobacterium nucleatum as a marker for detection of colorectal tumours in a Japanese population. Ann Clin Biochem. 2017;54:86–91.
Martin FE, Nadkarni MA, Jacques NA, et al. Quantitative microbiological study of human carious dentine by culture and real-time PCR: association of anaerobes with histopathological changes in chronic pulpitis. J Clin Microbiol. 2002;40:1698–704.
Suehiro Y, Wong CW, Chirieac LR, et al. Epigenetic-genetic interactions in the APC/WNT, RAS/RAF, and P53 pathways in colorectal carcinoma. Clin Cancer Res. 2008;14:2560–9.
Yang Y, Weng W, Peng J, et al. Fusobacterium nucleatum increases proliferation of colorectal cancer cells and tumor development in mice by activating toll-like receptor 4 signaling to nuclear factor-kappaB, and up-regulating expression of microRNA-21. Gastroenterology. 2017;152(851–66):e24.
Mima K, Nishihara R, Qian ZR, et al. Fusobacterium nucleatum in colorectal carcinoma tissue and patient prognosis. Gut. 2016;65:1973–80.
Stillwell AP, Ho YH, Veitch C. Systematic review of prognostic factors related to overall survival in patients with stage IV colorectal cancer and unresectable metastases. World J Surg. 2011;35:684–92.
Mima K, Sukawa Y, Nishihara R, et al. Fusobacterium nucleatum and T cells in colorectal carcinoma. JAMA Oncol. 2015;1:653–61.
Galon J, Mlecnik B, Bindea G, et al. Towards the introduction of the ‘Immunoscore’ in the classification of malignant tumours. J Pathol. 2014;232:199–209.
Tuttle RS, Strubel NA, Mourad J, et al. A non-lectin-like mechanism by which Fusobacterium nucleatum 10953 adheres to and activates human lymphocytes. Oral Microbiol Immunol. 1992;7:78–83.
Acknowledgements
We are very grateful to Ms. Naoko Okayama and Mr. Hidekazu Mizuno of Yamaguchi University Hospital for their invaluable help in analyzing the data. This study was supported by JSPS KAKENHI Grant no. 25460687.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest.
Rights and permissions
About this article
Cite this article
Yamaoka, Y., Suehiro, Y., Hashimoto, S. et al. Fusobacterium nucleatum as a prognostic marker of colorectal cancer in a Japanese population. J Gastroenterol 53, 517–524 (2018). https://doi.org/10.1007/s00535-017-1382-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-017-1382-6