Abstract
Purpose
This study seeks to adapt a guideline for end-of-life care in patients with cancer to be used by healthcare teams.
Methods
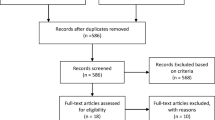
This methodological study was conducted by modifying the ADAPTE process and adding to it a qualitative study and consensus ratings by a multidisciplinary panel of experts. A qualitative study was thus performed to identify the end-of-life needs of patients with cancer. Then, the source guidelines and the results of the qualitative study were used to draft the initial version of the guideline, with 85 adaptation recommendations. A multidisciplinary panel of healthcare experts performed the external review of the recommendations based on the four criteria of relatedness, comprehensibility, usefulness, and feasibility and scored them on a scale of 1–9. The mean score of each recommendation was calculated, and the recommendations were classified into three categories: appropriate (mean score of 7–9), uncertain (mean score of 4–6.99), and inappropriate (mean score of 1–3.99).
Results
All the recommendations were approved, as they all had a mean score of 7 or higher, and were then categorized into 11 dimensions: communication management; participatory and evidence-based decision-making management; pain management; dyspnea management; nausea and vomiting management; anorexia and cachexia management; constipation management; death rattle management; management of delirium, anxiety, and restlessness; hydration management; and pharmacological considerations.
Conclusion
The adaptation of the guideline for end-of-life care in patients with cancer in Iran was performed by modifying the ADAPTE process with the participation of multidisciplinary stakeholders and based on the local needs.
Similar content being viewed by others
Data availability
N/A.
Code availability
N/A.
References
Graham ID, Harrison MB (2005) Evaluation and adaptation of clinical practice guidelines. Evid Based Nurs 8(3):68–72
Choi T-Y et al (2015) The quality of clinical practice guidelines in traditional medicine in Korea: appraisal using the AGREE II instrument. Implement Sci 10(1):104
Amer YS, Elzalabany MM, Omar TI, Ibrahim AG, Dowidar NL (2015) The ‘Adapted ADAPTE’: an approach to improve utilization of the ADAPTE guideline adaptation resource toolkit in the Alexandria Center for Evidence-Based Clinical Practice Guidelines. J Eval Clin Pract 21(6):1095–1106
Harrison MB, Graham ID, van den Hoek J, Dogherty EJ, Carley ME, Angus V (2013) Guideline adaptation and implementation planning: a prospective observational study. Implementation Sci 8(1):49
Chakraborty SP, Jones KM, Mazza D (2014) Adapting lung cancer symptom investigation and referral guidelines for general practitioners in Australia: reflections on the utility of the ADAPTE framework. J Eval Clin Pract 20(2):129–135
Fervers B, Burgers JS, Voellinger R, Brouwers M, Browman GP, Graham ID, Harrison MB, Latreille J, Mlika-Cabane N, Paquet L, Zitzelsberger L, Burnand B (2011) ADAPTE Collaboration. Guideline adaptation: an approach to enhance efficiency in guideline development and improve utilisation. BMJ Qual Saf 20(3):228–36
Attia A (2013) Adaptation of international evidence based clinical practice guidelines: the ADAPTE process. Middle East Fertil Soc J 18(2):123–126
Wang Z, Norris SL, Bero L (2018) The advantages and limitations of guideline adaptation frameworks. Implement Sci 13(1):72
Ayhan Baser D, Kahveci R, BaydarArtantas A, Yasar İ, Aksoy H, Koc EM, Kasim İ, Kunnamo I, Özkara A (2018) Exploring guideline adaptation strategy for Turkey: is “ADAPTE” feasible or does it need adaptation as well? J Eval Clin Pract 24(1):97–104
Chiorean EG, Nandakumar G, Fadelu T, Temin S, Alarcon-Rozas AE, Bejarano S, Croitoru AE, Grover S, Lohar PV, Odhiambo A, Park SH, Garcia ER, Teh C, Rose A, Zaki B, Chamberlin MD (2020) Treatment of patients with late-stage colorectal cancer: ASCO resource-stratified guideline. JCO Glob Oncol 6:414–438
Costas-Chavarri A, Nandakumar G, Temin S, Lopes G, Cervantes A, Cruz Correa M, Engineer R, Hamashima C, Ho GF, Huitzil FD, MalekzadehMoghani M, Sharara AI, Stern MC, Teh C, Vázquez Manjarrez SE, Verjee A, Yantiss R, Shah MA (2019) Treatment of patients with early-stage colorectal cancer: ASCO resource-stratified guideline. J Glob Oncol 5:1–19
Lovell M, Luckett T, Boyle F, Stubbs J, Phillips J, Davidson PM, Olver I, von Dincklage J, Agar M (2015) Adaptation of international guidelines on assessment and management of cancer pain for the Australian context. Asia Pac J Clin Oncol 11(2):170–177
Salarvand S, Hemati S, Adibi P, Taleghani F, Saleki M (2020) An innovative approach to clinical practice guideline adaptation in the nursing profession in a develo** country. Cancer Manag Res 12:2255–2264
Hoedl M, Schoberer D, Halfens RJG, Lohrmann C (2018) Adaptation of evidence-based guideline recommendations to address urinary incontinence in nursing home residents according to the ADAPTE-process. J Clin Nurs 27(15–16):2974–2983
Mack S, Hahn S, Palli C, Findling T, Lohrmann C (2019) Adaptation of clinical practice guideline recommendations in hospitals for people living with dementia and their caregivers. Worldviews on Evidence-Based Nursing 16(1):36–42
Bower JE, Bak K, Berger A, Breitbart W, Escalante CP, Ganz PA, Schnipper HH, Lacchetti C, Ligibel JA, Lyman GH, Ogaily MS, Pirl WF, Jacobsen PB (2014) American Society of Clinical Oncology. Screening, assessment, and management of fatigue in adult survivors of cancer: an American Society of Clinical oncology clinical practice guideline adaptation. J Clin Oncol 32(17):1840–50
Andersen BL, DeRubeis RJ, Berman BS, Gruman J, Champion VL, Massie MJ, Holland JC, Partridge AH, Bak K, Somerfield MR, Rowland JH (2014) American Society of Clinical Oncology. Screening, assessment, and care of anxiety and depressive symptoms in adults with cancer: an American Society of Clinical Oncology guideline adaptation. J Clin Oncol 32(15):1605–19
The ADAPTE Collaboration. The ADAPTE process: resource toolkit for guideline adaptation. Version 2.0. Guideline international network. 2009. Available from: http://www.g-i-n.net. Accessed 25 Feb 2020
Lee GH, McGrath C, Yiu CK (2016) Develo** clinical practice guidelines for caries prevention and management for pre-school children through the ADAPTE process and Delphi consensus. Health Res Policy Syst 14(1):44
National Clinical Guideline Centre (2015) Care of dying adults in the last days of life. NICE guideline NG31. https://www.nice.org.uk/guidance/ng31
NCCN Clinical Practice Guidelines in Oncology (2016) Palliative care version 1. https://www.nccn.org/
McCusker M, Ceronsky L, Crone C, Epstein H, Greene B, Halvorson J, Kephart K, Mallen E, Nosan B, Rohr M, Rosenberg E, Ruff R, Schlecht K, Setterlund L. (2013) Institute for clinical systems improvement. Palliative Care for Adults
Registered Nurses’ Association of Ontario (2011) End-of-life care during the last days and hours. Registered Nurses’ Association of Ontario, Toronto
National Institute for Clinical Excellence (2004) Improving supportive and palliative care for adults with cancer. Cancer service guideline [CSG4]. https://www.nice.org.uk/guidance/csg4
Guideline And Audit Implementation Network (2013) Guidelines for palliative and end of life care in nursing homes and residential care homes. https://rqia.org.uk/
Missouri Department of Health and Senior Services and Missouri End of Life Coalition’s End of Life in the Nursing Home Task Force (2003) Guidelines for end of life care in long-term care facilities. http://health.mo.gov/safety/showmelongtermcare/pdf/EndofLifeManual.pdf/
National Consensus Project for Quality Palliative Care (2013) Clinical practice guidelines for quality palliative care. https://www.nationalcoalitionhpc.org/
The AGREE Research Trust (2013) Appraisal of guidelines for research and evaluation II. AGREE II Instrument. www.agreetrust.org. Accessed 6 Aug 2015
Irajpour A, Hashemi M, Taleghani F (2020) The quality of guidelines on the end-of-life care: a systematic quality appraisal using AGREE II instrument. Support Care Cancer 28(4):1555–1561
Elo S, Kyngäs H (2008) The qualitative content analysis process. J Adv Nurs 62(1):107–115
Hashemi M, Irajpour A, Taleghani F (2018) Caregivers needing care: the unmet needs of the family caregivers of end-of-life cancer patients. Support Care Cancer 26(3):759–766
Speziale HS, Streubert HJ, Carpenter DR (2011) Qualitative research in nursing: advancing the humanistic imperative. Lippincott Williams & Wilkins, Philadelphia
Fitch K, Bernstein SJ, Aguilar MD, Burnand B, LaCalle JR (2001) The RAND/UCLA appropriateness method user’s manual. Rand Corp Santa Monica CA
Abidi S, Mian I, Garcia-Ortega I, Lecomte T, Raedler T, Jackson K, Jackson K, Pringsheim T, Addington D (2017) Canadian guidelines for the pharmacological treatment of schizophrenia spectrum and other psychotic disorders in children and youth. Can J Psychiatry 62(9):635–647
Funding
The present study was conducted with the fund support of the Vice-Chancellor in Research of Isfahan University of Medical sciences (Project number: 394536).
Author information
Authors and Affiliations
Contributions
Study conception and design: Alireza Irajpour, Fariba Taleghani.
Acquisition of data: Maryam Hashemi.
Analysis and interpretation of data: Maryam Hashemi, Alireza Irajpour.
Drafting of manuscript: Maryam Hashemi.
Critical revision of manuscript: Alireza Irajpour, Fariba Taleghani.
Read and approval of the final manuscript: Alireza Irajpour, Fariba Taleghani, Maryam Hashemi.
Corresponding author
Ethics declarations
Ethics approval
This study was approved by the Vice-Chancellor in Research Affairs and Ethics Committee (code: 394536), Isfahan university of medical sciences, Iran. This study was carried out in accordance with the principles of the Declaration of Helsinki. The participants were briefed on the goals and methods used in the research and signed informed written consent forms. All the information provided by the participants remained confidential. All the references used in the study were cited.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent for publication
Informed consent was obtained from the participants and all authors approved the final version of the manuscript.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Irajpour, A., Hashemi, M. & Taleghani, F. Clinical practice guideline for end-of-life care in patients with cancer: a modified ADAPTE process. Support Care Cancer 30, 2497–2505 (2022). https://doi.org/10.1007/s00520-021-06558-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-021-06558-2