Abstract
Purpose
Modafinil has been reported to benefit a subgroup of patients suffering severe fatigue while undergoing chemotherapy. Docetaxel is associated with fatigue that may lead to premature therapy withdrawal. We investigated whether modafinil could reduce fatigue during docetaxel chemotherapy.
Methods
This multicenter, randomized, double-blind, placebo-controlled study evaluated the efficacy of modafinil in patients with metastatic prostate or breast cancer undergoing docetaxel chemotherapy (every 21 days; minimum dose 50 mg/m2). At the start of their third or subsequent chemotherapy cycle, patients with significant docetaxel-associated fatigue were randomized to receive concurrent modafinil 200 mg/day or placebo for 15 days (“treatment periods” (TP)). Docetaxel was continued for up to four further cycles. Fatigue was evaluated with the fatigue component of the MD Anderson Symptom Inventory (MDASI). The primary endpoint was cumulative MDASI area under the curve (AUC) during the first 7 days of study medication during TP1 and TP2.
Results
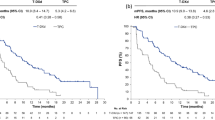
Evaluable data were available from 83 patients (65 with prostate cancer). There was no statistically significant difference between the two treatment arms for the primary endpoint (MSADI AUC3–10 35.9 vs 39.6; 95 % confidence interval −8.9, 1.4; P = 0.15). Overall toxicity was comparable between treatment groups; however, the incidence of grade ≤2 nausea and vomiting was higher in the modafinil arm (45.4 vs 25 %).
Conclusions
Assessing and managing chemotherapy-related fatigue remains a major challenge. There was a lack of difference between the two arms in the planned primary endpoint. However, there was a modest but consistent trend towards improvement of docetaxel-related fatigue in those treated with modafinil. Based on the study findings, modafinil for the treatment of fatigue associated with docetaxel chemotherapy elicits modest improvements. Larger, longer term, randomized, controlled studies are required to clarify the exact role of modafinil in the treatment of docetaxel-related fatigue.



Similar content being viewed by others
References
Hofman M, Ryan JL, Figueroa-Moseley CD, Jean-Pierre P, Morrow GR (2007) Cancer-related fatigue: the scale of the problem. Oncologist 12(Suppl 1):4–10
Bower JE, Lamkin DM (2013) Inflammation and cancer-related fatigue: mechanisms, contributing factors, and treatment implications. Brain Behav Immun 30:S48–S57
Miller AH (2009) Norman cousins lecture. mechanisms of cytokine-induced behavioral changes: psychoneuroimmunology at the translational interface. Brain Behav Immun 23(2):149–158
Ryan JL, Carroll JK, Ryan EP, Mustian KM, Fiscella K, Morrow GR (2007) Mechanisms of cancer-related fatigue. Oncologist 12(Suppl 1):22–34
Iop A, Manfredi AM, Bonura S (2004) Fatigue in cancer patients receiving chemotherapy: an analysis of published studies. Ann Oncol 15(5):712–720
Stone PC, Minton O (2008) Cancer-related fatigue. Eur J Cancer 44(8):1097–1104
Jacobsen PB, Donovan KA, Vadaparampil ST, Small BJ (2007) Systematic review and meta-analysis of psychological and activity-based interventions for cancer-related fatigue. Health Psychol 26(6):660–667
Minton O, Richardson A, Sharpe M, Hotopf M, Stone PC (2011) Psychostimulants for the management of cancer-related fatigue: a systematic review and meta-analysis. J Pain Symptom Manage 41(4):761–767
Minton O, Richardson A, Sharpe M, Hotopf M, Stone P (2010) Drug therapy for the management of cancer-related fatigue. Cochrane Database Syst Rev 7:CD006704
Minton O, Richardson A, Sharpe M, Hotopf M, Stone P (2008) A systematic review and meta-analysis of the pharmacological treatment of cancer-related fatigue. J Natl Cancer Inst 100(16):1155–1166
Golicki D, Bala MM, Niewada M, Wierzbicka A (2010) Modafinil for narcolepsy: systematic review and meta-analysis. Med Sci Monit 16(8):RA177–RA186
Brown JN, Howard CA, Kemp DW (2010) Modafinil for the treatment of multiple sclerosis-related fatigue. Ann Pharmacother 44(6):1098–1103
Saavedra-Velez C, Yusim A, Anbarasan D, Lindenmayer JP (2009) Modafinil as an adjunctive treatment of sedation, negative symptoms, and cognition in schizophrenia: a critical review. J Clin Psychiatry 70(1):104–112
Kumar R (2008) Approved and investigational uses of modafinil : an evidence-based review. Drugs 68(13):1803–1839
Wirz S, Nadstawek J, Kuhn KU, Vater S, Junker U, Wartenberg HC (2010) Modafinil for the treatment of cancer-related fatigue : an intervention study. Schmerz 24(6):587–595
Kaleita TA, Wellisch DK, Graham CA, Steh B, Nghiemphu P, Ford JM et al (2006) Pilot study of modafinil for treatment of neurobehavioral dysfunction and fatigue in adults patients with brain tumors. J Clin Oncol (Meeting Abstracts) 24(Suppl 18):1503
Spathis A, Dhillan R, Booden D, Forbes K, Vrotsou K, Fife K (2009) Modafinil for the treatment of fatigue in lung cancer: a pilot study. Palliat Med 23(4):325–331
Blackhall L, Petroni G, Shu J, Baum L, Farace E (2009) A pilot study evaluating the safety and efficacy of modafinal for cancer-related fatigue. J Palliat Med 12(5):433–439
Morrow GR, Ryan JL, Kohli S, et al. (2006) Modafinil (Provigil®) for persistent post-treatment fatigue: an open label study of 82 women with breast cancer. Abstracts of the 18th MASCC International Symposium, Toronto; Supportive Care in Cancer; 583–687
Jean-Pierre P, Morrow GR, Roscoe JA, Heckler C, Mohile S, Janelsins M et al (2010) A phase 3 randomized, placebo-controlled, double-blind, clinical trial of the effect of modafinil on cancer-related fatigue among 631 patients receiving chemotherapy: a University of Rochester Cancer Center Community Clinical Oncology Program Research base study. Cancer 116(14):3513–3520
Cleeland CS, Mendoza TR, Wang XS, Chou C, Harle MT, Morrissey M et al (2000) Assessing symptom distress in cancer patients: the M.D. Anderson Symptom Inventory. Cancer 89(7):1634–1646
Hadzi-Pavlovic D, Hickie IB, Wilson AJ, Davenport TA, Lloyd AR, Wakefield D (2000) Screening for prolonged fatigue syndromes: validation of the SOFA scale. Soc Psychiatry Psychiatr Epidemiol 35(10):471–479
Tannock IF, de Wit R, Berry WR, Horti J, Pluzanska A, Chi KN et al (2004) Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N Engl J Med 351(15):1502–1512
Brown H, Prescott R (eds) (2006) Applied mixed models in medicine, 2nd edn. John Wiley and Sons, New Jersey
Sloan JA, Cella D, Hays RD (2005) Clinical significance of patient-reported questionnaire data: another step toward consensus. J Clin Epidemiol 58(12):1217–1219
Barton DL, Liu H, Dakhil SR et al (2012) Phase III evaluation of American ginseng (Panax quinquefolius) to improve cancer-related fatigue: NCCTG trial N07C2. J Clin Oncol 30(Suppl):9001, Abstract
Yennurajalingam S, Frisbee-Hume S, Palmer JL, Gado-Guay MO, Bull J, Phan AT et al (2013) Reduction of cancer-related fatigue with dexamethasone: a double-blind, randomized, placebo-controlled trial in patients with advanced cancer. J Clin Oncol 31(25):3076–3082
Stankoff B, Waubant E, Confavreux C, Edan G, Debouverie M, Rumbach L et al (2005) Modafinil for fatigue in MS: a randomized placebo-controlled double-blind study. Neurology 64(7):1139–1143
Fife K, Spathis A, Dutton S, et al. (2013) A multicenter, randomized, double-blinded, placebo-controlled trial of modafinil for lung cancer-related fatigue: dose response and patient satisfaction data. J Clin Oncol 31[Suppl]
Jean-Pierre P, Figueroa-Moseley CD, Kohli S, Fiscella K, Palesh OG, Morrow GR (2007) Assessment of cancer-related fatigue: implications for clinical diagnosis and treatment. Oncologist 12(Suppl 1):11–21
Acknowledgments
We thank the patients and their families who took part in this study. We greatly appreciate the contributions of the clinicians and their staff from the hospitals from around Australia that were involved in this study, although not all were able to recruit patients. NSW: Prince of Wales Hospital, Bankstown - Lidcombe Hospital, Southern Medical Day Care, Sydney Haematology & Oncology Clinical Trials Unit, St George Hospital, St Vincent's Hospital, Coffs Harbour Base Hospital, Armidale Hospital, Tamworth Hospital, Lingard Private Hospital, The Tweed Hospital, Southern Highlands Cancer Centre, Manning Rural Referral Hospital; ACT: The Canberra Hospital; VIC: Box Hill Hospital, Monash Medical Centre, Frankston Hospital, Frankston Private Hospital, John Fawkner Private Hospital, Ballarat Oncology & Haematology Services; TAS: Launceston General Hospital; SA: Royal Adelaide Hospital, The Queen Elizabeth Hospital, Calvary Hospital North Adelaide and QLD: Gold Coast Hospital.
Funding and financial disclosures
This work has been carried out with financial support from Sanofi Australia Pty Ltd. The sponsor was involved in the design and conduct of the study, provided logistical support during the trial, funded statistical and editorial assistance, and reviewed the final draft before submission. The study Steering Committee (EJH, PLDeS, GMM, and AL) designed the trial protocol and contributed to the design of the statistical analysis plan. This study is registered at ClinicalTrials.gov, NCT00917748. Statistical analyses were performed externally (Caro-Anne Badcock, Statistical Revelations Pty Ltd). The decision to submit the report for publication was made by the Steering Committee, who drafted then finalized the report with the help of a medical writer (Hazel Palmer, Scius Solutions Pty Ltd). The sponsor funded statistical and editorial assistance and reviewed the final draft before submission.
Conflict of interest
Phillip Parente is currently conducting research sponsored by Sanofi Australia. Tal Rapke is an employee of Sanofi Australia Pty Ltd. Elizabeth Hovey, Gavin Marx, Paul deSouza, and Phillip Parente have each sat on an advisory board for Sanofi oncology products. All remaining authors have declared no conflict of interest.
Author information
Authors and Affiliations
Consortia
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 22 kb)
Rights and permissions
About this article
Cite this article
Hovey, E., de Souza, P., Marx, G. et al. Phase III, randomized, double-blind, placebo-controlled study of modafinil for fatigue in patients treated with docetaxel-based chemotherapy. Support Care Cancer 22, 1233–1242 (2014). https://doi.org/10.1007/s00520-013-2076-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-013-2076-0