Abstract
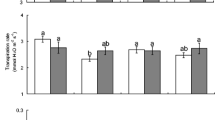
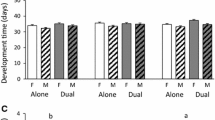
Plants growing under reduced water availability can affect insect herbivores differently, in some instances benefitting them. However, the forces mediating these positive impacts remain mostly unclear. To identify how water availability impacts plant quality and multi-trophic interactions, we conducted manipulative field studies with two populations of the specialist herbivore Pieris rapae, and its host plant, Rorippa indica. We found that P. rapae larvae experienced higher survival on R. indica growing under low water availability compared with plants grown under high water availability. Higher survival of eggs and larvae was related to the reduced abundance of other herbivores and natural enemies. Water availability had differential impacts on other members of the herbivore community by altering plant quality. Low water availability decreased the quality of R. indica to most herbivores, as indicated by reduced abundance in the field and decreased relative growth rate in laboratory feeding assays. In contrast, P. rapae larval performance was not affected by sympatric R. indica grown under different water availability. These results indicate that local P. rapae populations possess physiological adaptations to overcome fluctuations in host quality. Our findings illustrate that reduced water availability is beneficial to a specialist herbivore but detrimental to most other herbivores. Our work highlights the complex effects of the arthropod communities associated with plants in determining the impacts of water availability on insect herbivores.






Similar content being viewed by others
Data availability statement
Data in this manuscript are available in the Dryad Digital Repository: https://doi.org/10.5061/dryad.g1jwstqq0.
References
Anderegg WRL, Kane JM, Anderegg LDL (2013) Consequences of widespread tree mortality triggered by drought and temperature stress. Nat Clim Change 3:30–36. https://doi.org/10.1038/nclimate1635
Awmack CS, Leather SR (2002) Host plant quality and fecundity in herbivorous insects. Annu Rev Entomol 47:817–844. https://doi.org/10.1146/annurev.ento.47.091201.145300
Brooks ME, Kristensen K, van Benthem KJ, Magnusson A, Berg CW, Nielsen A, Skaug HJ, Machler M, Bolker BM (2017) glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. R J 9:378–400
Bunker DE, Carson WP (2005) Drought stress and tropical forest woody seedlings: effect on community structure and composition. J Ecol 93:794–806. https://doi.org/10.1111/j.1365-2745.2005.01019.x
Cáceres MD, Legendre P (2009) Associations between species and groups of sites: indices and statistical inference. Ecology 90:3566–3574. https://doi.org/10.1890/08-1823.1
Chung SH, Rosa C, Scully ED, Peiffer M, Tooker JF, Hoover K, Luthe DS, Felton GW (2013) Herbivore exploits orally secreted bacteria to suppress plant defenses. Proc Natl Acad Sci USA 110:15728–15733. https://doi.org/10.1073/pnas.1308867110
Copolovici L, Kännaste A, Remmel T, Niinemets Ü (2014) Volatile organic compound emissions from Alnus glutinosa under interacting drought and herbivory stresses. Environ Exp Bot 100:55–63. https://doi.org/10.1016/j.envexpbot.2013.12.011
Cronin JT, Abrahamson WG, Craig TP (2001) Temporal variation in herbivore host-plant preference and performance: constraints on host-plant adaptation. Oikos 93:312–320. https://doi.org/10.1034/j.1600-0706.2001.930214.x
Dambros CdS, da Silva VNV, Azevedo R, de Morais JW (2013) Road-associated edge effects in Amazonia change termite community composition by modifying environmental conditions. J Nat Conserv 21:279–285. https://doi.org/10.1016/j.jnc.2013.02.003
De Vos M, Van Oosten VR, Van Poecke RM, Van Pelt JA, Pozo MJ, Mueller MJ, Buchala AJ, Metraux JP, Van Loon LC, Dicke M, Pieterse CM (2005) Signal signature and transcriptome changes of Arabidopsis during pathogen and insect attack. Mol Plant Microbe Interact 18:923–937. https://doi.org/10.1094/MPMI-18-0923
English-Loeb G, Stout MJ, Duffey SS (1997) Drought stress in tomatoes: changes in plant chemistry and potential nonlinear consequences for insect herbivores. Oikos 79:456–468
Felton GW, Donato K, Del Vecchio RJ, Duffey SS (1989) Activation of plant foliar oxidases by insect feeding reduces nutritive quality of foliage for noctuid herbivores. J Chem Ecol 15:2667–2694. https://doi.org/10.1007/BF01014725
Fox J, Weisberg S (2018) An R companion to applied regression. Sage Publications, California
Garssen AG, Verhoeven JTA, Soons MB (2014) Effects of climate-induced increases in summer drought on riparian plant species: a meta-analysis. Freshw Biol 59:1052–1063. https://doi.org/10.1111/fwb.12328
Gripenberg S, Morriën E, Cudmore A, Salminen J-P, Roslin T (2007) Resource selection by female moths in a heterogeneous environment: what is a poor girl to do? J Anim Ecol 76:854–865. https://doi.org/10.1111/j.1365-2656.2007.01261.x
Gripenberg S, Mayhew PJ, Parnell M, Roslin T (2010) A meta-analysis of preference–performance relationships in phytophagous insects. Ecol Lett 13:383–393. https://doi.org/10.1111/j.1461-0248.2009.01433.x
Hartig F (2017) DHARMa: residual diagnostics for hierarchical (multi-level/mixed) regression models. R package version 0.1 5:5
Huang TC (1996) Flora of Taiwan, vol 2, 2nd edn. Taipei, Editorial Committee of the Flora of Taiwan, p 766
Huang J, Nuessly GS, McAuslane HJ, Nagata RT (2003) Effect of screening methods on expression of romaine lettuce resistance to adult banded cucumber beetle, Diabrotica balteata (Coleoptera: Chrysomelidae). Fla Entomol 86(194–198):195
Jamieson MA, Trowbridge AM, Raffa KF, Lindroth RL (2012) Consequences of climate warming and altered precipitation patterns for plant–insect and multitrophic interactions. Plant Physiol 160:1719–1727. https://doi.org/10.1104/pp.112.206524
Jisaburō Ōi FGM, Walker EH (1965) Flora of Japan. Smithsonian Institution, Washington
Jones HG (2007) Monitoring plant and soil water status: established and novel methods revisited and their relevance to studies of drought tolerance. J Exp Bot 58:119–130. https://doi.org/10.1093/jxb/erl118
Kroes A, Van Loon JJA, Dicke M (2015) Density-dependent interference of aphids with caterpillar-induced defenses in Arabidopsis: involvement of phytohormones and ttranscription factors. Plant Cell Physiol 56:98–106. https://doi.org/10.1093/pcp/pcu150
Kuussaari M, Singer M, Hanski I (2000) Local specialization and landscape-level influence on host use in an herbivorous insect. Ecology 81:2177–2187. https://doi.org/10.2307/177106
Langsrud Ø (2003) ANOVA for unbalanced data: use type II instead of type III sums of squares. Stat Comput 13:163–167. https://doi.org/10.1023/A:1023260610025
Li Y, Dicke M, Harvey JA, Gols R (2014) Intra-specific variation in wild Brassica oleracea for aphid-induced plant responses and consequences for caterpillar–parasitoid interactions. Oecologia 174:853–862. https://doi.org/10.1007/s00442-013-2805-6
Mattson WJ, Haack RA (1987) The role of drought in outbreaks of plant-eating insects. Bioscience 37:110–118. https://doi.org/10.2307/1310365
Murren CJ, Alt CHS, Kohler C, Sancho G (2020) Natural variation on whole-plant form in the wild is influenced by multivariate soil nutrient characteristics: natural selection acts on root traits. Am J Bot. https://doi.org/10.1002/ajb2.1420
National Research Council (2011) Advancing the science of climate change. National Academies Press, Washington
Ockendon N, Baker DJ, Carr JA, White EC, Almond REA, Amano T, Bertram E, Bradbury RB, Bradley C, Butchart SHM, Doswald N, Foden W, Gill DJC, Green RE, Sutherland WJ, Tanner EVJ, Pearce-Higgins JW (2014) Mechanisms underpinning climatic impacts on natural populations: altered species interactions are more important than direct effects. Glob Change Biol 20:2221–2229. https://doi.org/10.1111/gcb.12559
Oksanen J, Blanchet FG, Kindt R, Legendre P, O’hara R, Simpson GL, Solymos P, Stevens MHH, Wagner H (2010) Vegan: community ecology package. R package version 1.17-4
Orre GUS, Wratten SD, Jonsson M, Hale RJ (2010) Effects of an herbivore-induced plant volatile on arthropods from three trophic levels in brassicas. Biol Control 53:62–67. https://doi.org/10.1016/j.biocontrol.2009.10.010
Peiffer M, Felton GW (2005) The host plant as a factor in the synthesis and secretion of salivary glucose oxidase in larval Helicoverpa zea. Arch Insect Biochem Physiol 58:106–113. https://doi.org/10.1002/arch.20034
Pichersky E, Gershenzon J (2002) The formation and function of plant volatiles: perfumes for pollinator attraction and defense. Curr Opin Plant Biol 5:237–243. https://doi.org/10.1016/s1369-5266(02)00251-0
Pickett STA, Cadenasso ML, Grove JM, Boone CG, Groffman PM, Irwin E, Kaushal SS, Marshall V, McGrath BP, Nilon CH, Pouyat RV, Szlavecz K, Troy A, Warren P (2011) Urban ecological systems: scientific foundations and a decade of progress. J Environ Manag 92:331–362. https://doi.org/10.1016/j.jenvman.2010.08.022
Pieterse C, Van der Does D, Zamioudis C, Leon-Reyes A, Van Wees S, Schekman R (2012) Hormonal modulation of plant immunity. Annu Rev Cell Dev Biol 28:489–521. https://doi.org/10.1146/annurev-cellbio-092910-154055
R Core Team (2017) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Schmelz EA, Alborn HT, Tumlinson JH (2001) The influence of intact-plant and excised-leaf bioassay designs on volicitin- and jasmonic acid-induced sesquiterpene volatile release in Zea mays. Planta 214:171–179. https://doi.org/10.1007/s004250100603
Schulze E (1986) Whole-plant responses to drought. Funct Plant Biol 13:127–141. https://doi.org/10.1071/PP9860127
Scott ER, Li X, Kfoury N, Morimoto J, Han W-Y, Ahmed S, Cash SB, Griffin TS, Stepp JR, Robbat A, Orians CM (2018) Interactive effects of drought severity and simulated herbivory on tea (Camellia sinensis) volatile and non-volatile metabolites. Exp Bot Environ. https://doi.org/10.1016/j.envexpbot.2018.10.025
Soler R, Badenes-Pérez FR, Broekgaarden C, Zheng S-J, David A, Boland W, Dicke M (2012) Plant-mediated facilitation between a leaf-feeding and a phloem-feeding insect in a brassicaceous plant: from insect performance to gene transcription. Funct Ecol 26:156–166. https://doi.org/10.1111/j.1365-2435.2011.01902.x
Tariq M, Wright DJ, Bruce TJ, Staley JT (2013) Drought and root herbivory interact to alter the response of above-ground parasitoids to aphid infested plants and associated plant volatile signals. PLoS ONE 8:e69013. https://doi.org/10.1371/journal.pone.0069013
Thompson JN (1988) Evolutionary ecology of the relationship between oviposition preference and performance of offspring in phytophagous insects. Entomol Exp Appl 47:3–14. https://doi.org/10.1111/j.1570-7458.1988.tb02275.x
Trotter R, Cobb N, Whitham T (2008) Arthropod community diversity and trophic structure: a comparison between extremes of plant stress. Ecol Entomol 33:1–11. https://doi.org/10.1111/j.1365-2311.2007.00941.x
Venables WN, Ripley BD (2013) Modern applied statistics with S-PLUS. Springer Science & Business Media, Berlin
Wang J, Peiffer M, Hoover K, Rosa C, Zeng R, Felton GW (2017) Helicoverpa zea gut-associated bacteria indirectly induce defenses in tomato by triggering a salivary elicitor(s). New Phytol 214:1294–1306. https://doi.org/10.1111/nph.14429
Waring GL, Cobb NS (1992) The impact of plant stress on herbivore population dynamics. Insect-Plant Interact 4:167–226
White TCR (1969) An index to measure weather-induced stress of trees associated with outbreaks of psyllids in Australia. Ecology 50:905–909. https://doi.org/10.2307/1933707
White TC (1974) A hypothesis to explain outbreaks of looper caterpillars, with special reference to populations of Selidosema suavis in a plantation of Pinus radiata in New Zealand. Oecologia 16:279–301. https://doi.org/10.1007/BF00344738
White TCR (2009) Plant vigour versus plant stress: a false dichotomy. Oikos 118:807–808. https://doi.org/10.1111/j.1600-0706.2009.17495.x
Zuur AF, Ieno EN, Walker NJ, Saveliev AA, Smith GM (2009) Mixed effects models and extensions in ecology with R. Springer Science & Business Media, Berlin
Zuur AF, Ieno EN, Elphick CS (2010) A protocol for data exploration to avoid common statistical problems. Methods Ecol Evol 1:3–14. https://doi.org/10.1111/j.2041-210X.2009.00001.x
Acknowledgements
We thank the International Agriculture and Development Graduate Program (College of Agricultural Sciences, Pennsylvania State University, INTAD competitive grant) for providing monetary support and feedback on experimental design. We thank Chi-Shun Chang, Yi-Zhang Wang, Han-Rong Li, Kazumu Kuramitsu, and Kai Han for collecting data. Special thanks to Dr. Yooichi Kainoh for arranging space and logistics of experiments in Japan, Dr. Ching-Wen Tan and Wei-Ting Chen for helpful information on the experiments in Taiwan, and Dr. Charles Mason, Dr. Asher Jones, and Jagdeep Singh Sidhu for feedbacks on the manuscript.
Author information
Authors and Affiliations
Contributions
PAL, KN, CKH, WPC, and GWF conceived the ideas, and designed the methodology. PAL and JAO analyzed the data. PAL, CML, and CHS collected the data. PAL, CML, JAO, KN, CKH, WPC, and GWF wrote the manuscript.
Corresponding author
Additional information
Communicated by Merijn Kant.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lin, PA., Liu, CM., Ou, JA. et al. Changes in arthropod community but not plant quality benefit a specialist herbivore on plants under reduced water availability. Oecologia 195, 383–396 (2021). https://doi.org/10.1007/s00442-020-04845-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-020-04845-z