Abstract
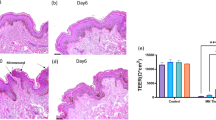
Cultured skin has been used extensively for testing therapeutic drugs because it replicates the physical and biochemical properties of whole skin. However, traditional static culture cannot fully maintain cell viability and skin morphology because of the limitations involved with nutrient transmission. Here, we develop a new dynamic perfusion platform for skin culture and compare it with a static culture device. Rat skins were cultured in either static or dynamic condition for 0, 3, 6, 9 and 12 days. H&E, periodic acid–Schiff (PAS) and picrosirius red (PSR) staining were used for skin morphology detection, immunostaining against cytokeratin 10 (CK10) for differentiation detection, immunostaining against proliferating cell nuclear antigen (PCNA) for cell proliferation detection and TUNEL staining for apoptosis detection. After culturing for 12 days, the epidermis, basement membrane, hair follicles and connective tissue were disrupted in the static group, whereas these features were preserved in the dynamic group. Moreover, compared to the static group, proliferation in the epidermis and hair follicles was significantly improved and apoptosis in dermis was significantly decreased in the dynamic group. These findings suggest that our device is effective for extending the culture period of rat skin to maintain its characteristics and viability in vitro.






Similar content being viewed by others
References
Akarca SO, Yavasoglu A, Aysegul U, Fatih O, Yilmaz-Dilsiz O, Timur K, Huseyin A (2012) Investigation on the effects of experimental STZ-induced diabetic rat model on basal membrane structures and gap junctions of skin. Int J Diabetes Dev C 32:82–89
Atac B, Wagner I, Horland R, Lauster R, Marx U, Tonevitsky AG, Azar RP, Lindner G (2013) Skin and hair on-a-chip: in vitro skin models versus ex vivo tissue maintenance with dynamic perfusion. Lab Chip 13:3555–3561
Black AF, Hudon V, Damour O, Germain L, Auger FA (1999) A novel approach for studying angiogenesis: a human skin equivalent with a capillary-like network. Cell Biol Toxicol 15:81–90
Diederichs S, Roker S, Marten D, Peterbauer A, Scheper T, van Griensven M, Kasper C (2009) Dynamic cultivation of human mesenchymal stem cells in a rotating bed bioreactor system based on the Z RP platform. Biotechnol Prog 25:1762–1771
El Maghraby GM, Barry BW, Williams AC (2008) Liposomes and skin: from drug delivery to model membranes. Eur J Pharm Sci 34:203–222
Festa E, Fretz J, Berry R, Schmidt B, Rodeheffer M, Horowitz M, Horsley V (2011) Adipocyte lineage cells contribute to the skin stem cell niche to drive hair cycling. Cell 146:761–771
Hanley K, Jiang Y, Elias PM, Feingold KR, Williams ML (1997) Acceleration of barrier ontogenesis in vitro through air exposure. Pediatr Res 41:293–299
Jaasma MJ, Plunkett NA, O'Brien FJ (2008) Design and validation of a dynamic flow perfusion bioreactor for use with compliant tissue engineering scaffolds. J Biotechnol 133:490–496
Kamiya T, Shirai A, Kawashima S, Sato S, Tamaoki T (1998) Hair follicle elongation in organ culture of skin from newborn and adult mice. J Dermatol Sci 17:54–60
Korin N, Bransky A, Dinnar U, Levenberg S (2009) Periodic “flow-stop” perfusion microchannel bioreactors for mammalian and human embryonic stem cell long-term culture. Biomed Microdevices 11:87–94
Ladd MR, Lee SJ, Atala A, Yoo JJ (2009) Bioreactor maintained living skin matrix. Tissue Eng A 15:861–868
Li L, Paus R, Margolis LB, Hoffman RM (1992) Hair growth in vitro from histocultured skin. In Vitro Cell Dev Biol 28A:479–481
Lindner G, Horland R, Wagner I, Atac B, Lauster R (2011) De novo formation and ultra-structural characterization of a fiber-producing human hair follicle equivalent in vitro. J Biotechnol 152:108–112
Metcalfe AD, Ferguson MW (2007) Tissue engineering of replacement skin: the crossroads of biomaterials, wound healing, embryonic development, stem cells and regeneration. J R Soc Interface 4:413–437
Montan I, Schiestl C, Schneider J, Pontiggia L, Luginbühl J, Biedermann T, Böttcher-Haberzeth S, Braziulis E, Meuli M, Reichmann E (2010) Formation of human capillaries in vitro: the engineering of prevascularized matrices. Tissue Eng A 16:269–282
Neves AA, Medcalf N, Brindle KM (2005) Influence of stirring-induced mixing on cell proliferation and extracellular matrix deposition in meniscal cartilage constructs based on polyethylene terephthalate scaffolds. Biomaterials 26:4828–4836
Nishi M, Matsumoto R, Dong J, Uemura T (2013) Engineered bone tissue associated with vascularization utilizing a rotating wall vessel bioreactor. J Biomed Mater Res A 101:421–427
Parenteau NL, Bilbo P, Nolte CJ, Mason VS, Rosenberg M (1992) The organotypic culture of human skin keratinocytes and fibroblasts to achieve form and function. Cytotechnology 9:163–171
Paus R (1991) Hair growth inhibition by heparin in mice: a model system for studying the modulation of epithelial cell growth by glycosaminoglycans? Br J Dermatol 124:415–422
Paus R, Luftl M, Czarnetzki BM (1994) Nerve growth factor modulates keratinocyte proliferation in murine skin organ culture. Br J Dermatol 130:174–180
Piola M, Soncini M, Cantini M, Sadr N, Ferrario G, Fiore GB (2013) Design and functional testing of a multichamber perfusion platform for three-dimensional scaffolds. TheScientificWorldJournal 2013:123974
Ratcliffe A, Niklason LE (2002) Bioreactors and bioprocessing for tissue engineering. Ann N Y Acad Sci 961:210–215
Sanchez WY, Prow TW, Sanchez WH, Grice JE, Roberts MS (2010) Analysis of the metabolic deterioration of ex vivo skin from ischemic necrosis through the imaging of intracellular NAD(P)H by multiphoton tomography and fluorescence lifetime imaging microscopy. J Biomed Opt 15:046008
Schmelzer E, Gerlach JC (2016) Multicompartmental hollow-fiber-based bioreactors for dynamic three-dimensional perfusion culture. Methods Mol Biol 1502:1–19
Sun T, Norton D, Haycock JW, Ryan AJ, MacNeil S (2006) Development of a closed bioreactor system for culture of tissue-engineered skin at an air–liquid interface. Tissue Eng A 11:1824–1831
Sutherland RM, Sordat B, Bamat J, Gabbert H, Bourrat B, Mueller-Klieser W (1986) Oxygenation and differentiation in multicellular spheroids of human colon carcinoma. Cancer Res 46:5320–5329
Vunjak-Novakovic G, Freed LE, Biron RJ, Langer R (1996) Effects of mixing on the composition and morphology of tissue-engineered cartilage. AIChE J 42:850–860
Wagner I, Materne EM, Brincker S, Sussbier U, Fradrich C, Busek M, Sonntag F, Sakharov DA, Trushkin EV, Tonevitsky AG, Lauster R, Marx U (2013) A dynamic multi-organ-chip for long-term cultivation and substance testing proven by 3D human liver and skin tissue co-culture. Lab Chip 13:3538–3547
Wilson C, Cotsarelis G, Wei Z-G, Fryer E (1994) Cells within the bulge region of mouse hair follicle transiently proliferate during early anagen: heterogeneity and functional differences of various hair cycles. Differentiation 55:127–136
Yanjia L, Jianli J (2009) Culture and application of skin organ model in vitroin skin transplantation field. CRTER 13:919–923
Funding
This work was supported by grants from the National Natural Science Foundation of China (Grant No. 31570975, 81602782) and the Natural Science Foundation of Hubei Province (Grant No. 2016CFB348).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All studies were approved by the Ethic Committee of Da** Hospital and conducted according to guidelines of the Experimental Animal Care and Use Committee at Army Medical University.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Yan, H., Tang, H., Qiu, W. et al. A new dynamic culture device suitable for rat skin culture. Cell Tissue Res 375, 723–731 (2019). https://doi.org/10.1007/s00441-018-2945-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-018-2945-4