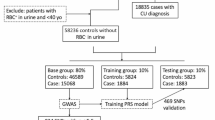
Abstract
In this study, we investigated the association of ACAN variants with otosclerosis, a frequent cause of hearing loss among young adults. We sequenced the coding, 5′-UTR and 3′-UTR regions of ACAN in 1497 unrelated otosclerosis cases and 1437 matched controls from six different subpopulations. The association between variants in ACAN and the disease risk was tested through single variant and gene-based association tests. After correction for multiple testing, 14 variants were significantly associated with otosclerosis, ten of which represented independent association signals. Eight variants showed a consistent association across all subpopulations. Allelic odds ratios of the variants identified four predisposing and ten protective variants. Gene-based tests showed an association of very rare variants in the 3′-UTR with the phenotype. The associated exonic variants are all located in the CS domain of ACAN and include both protective and predisposing variants with a broad spectrum of effect sizes and population frequencies. This includes variants with strong effect size and low frequency, typical for monogenic diseases, to low effect size variants with high frequency, characteristic for common complex traits. This single-gene allelic spectrum with both protective and predisposing alleles is unique in the field of complex diseases. In conclusion, these findings are a significant advancement to the understanding of the etiology of otosclerosis.




Similar content being viewed by others
Availability of data and material
The datasets generated and/or analyzed during the current study are available in the European Genome-phenome Archive (EGA; EGAS00001004398).
Code availability
Not applicable.
References
Allin KH, Hansen T, Pedersen OB (2014) The genome and diabetes. Ugeskr Laeger 176(23):2176–2179
Aspberg A (2012) The different roles of aggrecan interaction domains. J Histochem Cytochem 60:987–996. https://doi.org/10.1369/0022155412464376
Bast TH (1933) Development of the otic capsule: II. The origin, development and significance of the fissula ante fenestram and its relation to otosclerotic foci. Arch Otolaryngol 18:1–20. https://doi.org/10.1001/archotol.1933.03580060005001
Bel Hadj Ali I, Thys M, Beltaief N, Schrauwen I, Hilgert N, Vanderstraeten K, Dieltjens N, Mnif E, Hachicha S, Besbes G, Ben Arab S, Van Camp G (2008) A new locus for otosclerosis, OTSC8, maps to the pericentromeric region of chromosome 9. Hum Genet 123:267–272. https://doi.org/10.1007/s00439-008-0470-3
Boyle EA, O’Roak BJ, Martin BK, Kumar A, Shendure J (2014) MIPgen: optimized modeling and design of molecular inversion probes for targeted resequencing. Bioinformatics (oxford, England) 30:2670–2672. https://doi.org/10.1093/bioinformatics/btu353
Brown DJ, Kim TB, Petty EM, Downs CA, Martin DM, Strouse PJ, Moroi SE, Milunsky JM, Lesperance MM (2002) Autosomal dominant stapes ankylosis with broad thumbs and toes, hyperopia, and skeletal anomalies is caused by heterozygous nonsense and frameshift mutations in NOG, the gene encoding noggin. Am J Hum Genet 71:618–624. https://doi.org/10.1086/342067
Brownstein Z, Goldfarb A, Levi H, Frydman M, Avraham KB (2006) Chromosomal map** and phenotypic characterization of hereditary otosclerosis linked to the OTSC4 locus. Arch Otolaryngol Head Neck Surg 132:416–424. https://doi.org/10.1001/archotol.132.4.416
Chapman SC (2011) Can you hear me now? Understanding vertebrate middle ear development. Front Biosci (landmark Edition) 16:1675–1692. https://doi.org/10.2741/3813
Chen W, Campbell CA, Green GE, Van Den Bogaert K, Komodikis C, Manolidis LS, Aconomou E, Kyamides Y, Christodoulou K, Faghel C, Giguére CM, Alford RL, Manolidis S, Van Camp G, Smith RJH (2002) Linkage of otosclerosis to a third locus (OTSC3) on human chromosome 6p21.3-22.3. J Med Genet 39:473–477. https://doi.org/10.1136/jmg.39.7.473
Chen W, Meyer NC, McKenna MJ, Pfister M, McBride DJ Jr, Fukushima K, Thys M, Camp GV, Smith RJ (2007) Single-nucleotide polymorphisms in the COL1A1 regulatory regions are associated with otosclerosis. Clin Genet 71:406–414. https://doi.org/10.1111/j.1399-0004.2007.00794.x
Clark K, Sowers MR, Wallace RB, Jannausch ML, Lemke J, Anderson CV (1995) Age-related hearing loss and bone mass in a population of rural women aged 60 to 85 years. Ann Epidemiol 5:8–14
Consortium UK, Walter K, Min JL, Huang J, Crooks L, Memari Y, McCarthy S, Perry JRB, Xu C, Futema M, Lawson D, Iotchkova V, Schiffels S, Hendricks AE, Danecek P, Li R, Floyd J, Wain LV, Barroso I, Humphries SE, Hurles ME, Zeggini E, Barrett JC, Plagnol V, Richards JB, Greenwood CMT, Timpson NJ, Durbin R, Soranzo N (2015) The UK10K project identifies rare variants in health and disease. Nature 526:82–90. https://doi.org/10.1038/nature14962
Dawson SJ, Crompton M, Ziff J, Mowat A, Lavy JA, Saeed SR (2018) Progress in identifying genes underlying familial Otosclerosis using a Whole Exome sequencing approach. Mol Biol Hear Deafness (abstract)
Declau F, Van Spaendonck M, Timmermans JP, Michaels L, Liang J, Qiu JP, Van de Heyning P (2001) Prevalence of otosclerosis in an unselected series of temporal bones. Otol Neurotol 22:596–602
Ficker M, Powles N, Warr N, Pirvola U, Maconochie M (2004) Analysis of genes from inner ear developmental-stage cDNA subtraction reveals molecular regionalization of the otic capsule. Dev Biol 268:7–23. https://doi.org/10.1016/j.ydbio.2003.11.023
Fukuhara Y, Cho SY, Miyazaki O, Hattori A, Seo J-H, Mashima R, Kosuga M, Fukami M, ** D-K, Okuyama T, Nishimura G (2019) The second report on spondyloepimetaphyseal dysplasia, aggrecan type: a milder phenotype than originally reported. Clin Dysmorphol 28:26–29. https://doi.org/10.1097/MCD.0000000000000241
Gordon MA (1989) The genetics of otosclerosis: a review. Am J Otol 10:426–438
Hiatt JB, Pritchard CC, Salipante SJ, O’Roak BJ, Shendure J (2013) Single molecule molecular inversion probes for targeted, high-accuracy detection of low-frequency variation. Genome Res 23:843–854. https://doi.org/10.1101/gr.147686.112
Hindorff LA, Gillanders EM, Manolio TA (2011) Genetic architecture of cancer and other complex diseases: lessons learned and future directions. Carcinogenesis 32:945–954. https://doi.org/10.1093/carcin/bgr056
Hoffmann TJ, Keats BJ, Yoshikawa N, Schaefer C, Risch N, Lustig LR (2016) A large genome-wide association study of age-related hearing impairment using electronic health records. PLoS Genet 12:e1006371. https://doi.org/10.1371/journal.pgen.1006371
Karczewski KJ, Francioli LC, Tiao G, Cummings BB, Alföldi J, Wang Q, Collins RL, Laricchia KM, Ganna A, Birnbaum DP, Gauthier LD, Brand H, Solomonson M, Watts NA, Rhodes D, Singer-Berk M, England EM, Seaby EG, Kosmicki JA, Walters RK, Tashman K, Farjoun Y, Banks E, Poterba T, Wang A, Seed C, Whiffin N, Chong JX, Samocha KE, Pierce-Hoffman E, Zappala Z, O’Donnell-Luria AH, Minikel EV, Weisburd B, Lek M, Ware JS, Vittal C, Armean IM, Bergelson L, Cibulskis K, Connolly KM, Covarrubias M, Donnelly S, Ferriera S, Gabriel S, Gentry J, Gupta N, Jeandet T, Kaplan D, Llanwarne C, Munshi R, Novod S, Petrillo N, Roazen D, Ruano-Rubio V, Saltzman A, Schleicher M, Soto J, Tibbetts K, Tolonen C, Wade G, Talkowski ME, Neale BM, Daly MJ, MacArthur DG (2019) Variation across 141,456 human exomes and genomes reveals the spectrum of loss-of-function intolerance across human protein-coding genes. bioRxiv. https://doi.org/10.1101/531210
Khalfallah A, Schrauwen I, Mnejja M, HadjKacem H, Dhouib L, Mosrati MA, Hakim B, Lahmar I, Charfeddine I, Driss N, Ayadi H, Ghorbel A, Van Camp G, Masmoudi S (2011) Association of COL1A1 and TGFB1 polymorphisms with otosclerosis in a Tunisian population. Ann Hum Genet 75:598–604. https://doi.org/10.1111/j.1469-1809.2011.00665.x
Kiani C, Chen L, Wu YJ, Yee AJ, Yang BB (2002) Structure and function of aggrecan. Cell Res 12:19–32. https://doi.org/10.1038/sj.cr.7290106
Lauing KL, Cortes M, Domowicz MS, Henry JG, Baria AT, Schwartz NB (2014) Aggrecan is required for growth plate cytoarchitecture and differentiation. Dev Biol 396:224–236. https://doi.org/10.1016/j.ydbio.2014.10.005
Mangino M, Flex E, Capon F, Sangiuolo F, Carraro E, Gualandi F, Mazzoli M, Martini A, Novelli G, Dallapiccola B (2001) Map** of a new autosomal dominant nonsyndromic hearing loss locus (DFNA30) to chromosome 15q25-26. Eur J Hum Genet 9:667–671. https://doi.org/10.1038/sj.ejhg.5200707
Manolio TA, Collins FS, Cox NJ, Goldstein DB, Hindorff LA, Hunter DJ, McCarthy MI, Ramos EM, Cardon LR, Chakravarti A, Cho JH, Guttmacher AE, Kong A, Kruglyak L, Mardis E, Rotimi CN, Slatkin M, Valle D, Whittemore AS, Boehnke M, Clark AG, Eichler EE, Gibson G, Haines JL, Mackay TFC, McCarroll SA, Visscher PM (2009) Finding the missing heritability of complex diseases. Nature 461:747–753. https://doi.org/10.1038/nature08494
Mariella E, Marotta F, Grassi E, Gilotto S, Provero P (2019) The length of the expressed 3′ UTR is an intermediate molecular phenotype linking genetic variants to complex diseases. Front Genet 10:714–714. https://doi.org/10.3389/fgene.2019.00714
McKenna MJ, Kristiansen AG, Bartley ML, Rogus JJ, Haines JL (1998) Association of COL1A1 and otosclerosis: evidence for a shared genetic etiology with mild osteogenesis imperfecta. Am J Otol 19:604–610
Milroy CM, Michaels L (1990) Pathology of the otic capsule. J Laryngol Otol 104:83–90. https://doi.org/10.1017/s0022215100111946
Moumoulidis I, Axon P, Baguley D, Reid E (2007) A review on the genetics of otosclerosis. Clin Otolaryngol 32:239–247. https://doi.org/10.1111/j.1365-2273.2007.01475.x
Nilsson O, Guo MH, Dunbar N, Popovic J, Flynn D, Jacobsen C, Lui JC, Hirschhorn JN, Baron J, Dauber A (2014) Short stature, accelerated bone maturation, and early growth cessation due to heterozygous aggrecan mutations. J Clin Endocrinol Metab 99:E1510–E1518. https://doi.org/10.1210/jc.2014-1332
O’Roak BJ, Vives L, Fu W, Egertson JD, Stanaway IB, Phelps IG, Carvill G, Kumar A, Lee C, Ankenman K, Munson J, Hiatt JB, Turner EH, Levy R, O’Day DR, Krumm N, Coe BP, Martin BK, Borenstein E, Nickerson DA, Mefford HC, Doherty D, Akey JM, Bernier R, Eichler EE, Shendure J (2012) Multiplex targeted sequencing identifies recurrently mutated genes in autism spectrum disorders. Science (new York, N.y.) 338:1619–1622. https://doi.org/10.1126/science.1227764
Ovadia M, Parker CH, Lash JW (1980) Changing patterns of proteoglycan synthesis during chondrogenic differentiation. J Embryol Exp Morphol 56:59–70
Priyadarshi S, Ray CS, Biswal NC, Nayak SR, Panda KC, Desai A, Ramchander PV (2015) Genetic association and altered gene expression of osteoprotegerin in otosclerosis patients. Ann Hum Genet 79:225–237. https://doi.org/10.1111/ahg.12118
Quesnel AM, Ishai R, McKenna MJ (2018) Otosclerosis: temporal bone pathology. Otolaryngol Clin N Am 51:291–303. https://doi.org/10.1016/j.otc.2017.11.001
Rämö JT, Kiiskinen T, Karjalainen J, Krebs K, Kurki M, Havulinna AS, Hämäläinen E, Häppölä P, Hautakangas H, Karczewski KJ, Kanai M, Mägi R, Palta P, Esko T, Metspalu A, Pirinen M, Ripatti S, Milani L, Mäkitie A, Daly MJ, Palotie A (2020) Genome-wide screen of otosclerosis in population biobanks: 18 loci and shared heritability with skeletal structure. medRxiv. https://doi.org/10.1101/2020.11.15.20227868
Ruoslahti E, Yamaguchi Y (1991) Proteoglycans as modulators of growth factor activities. Cell 64:867–869. https://doi.org/10.1016/0092-8674(91)90308-l
San Lucas FA, Wang G, Scheet P, Peng B (2012) Integrated annotation and analysis of genetic variants from next-generation sequencing studies with variant tools. Bioinformatics 28:421–422. https://doi.org/10.1093/bioinformatics/btr667
Schrauwen I, Thys M, Vanderstraeten K, Fransen E, Dieltjens N, Huyghe JR, Ealy M, Claustres M, Cremers CRWJ, Dhooge I, Declau F, Van de Heyning P, Vincent R, Somers T, Offeciers E, Smith RJH, Van Camp G (2008) Association of bone morphogenetic proteins with otosclerosis. J Bone Min Res 23:507–516. https://doi.org/10.1359/jbmr.071112
Schrauwen I, Ealy M, Huentelman MJ, Thys M, Homer N, Vanderstraeten K, Fransen E, Corneveaux JJ, Craig DW, Claustres M, Cremers CWRJ, Dhooge I, Van de Heyning P, Vincent R, Offeciers E, Smith RJH, Van Camp G (2009) A genome-wide analysis identifies genetic variants in the RELN gene associated with otosclerosis. Am J Hum Genet 84:328–338. https://doi.org/10.1016/j.ajhg.2009.01.023
Schrauwen I, Weegerink NJ, Fransen E, Claes C, Pennings RJ, Cremers CW, Huygen PL, Kunst HP, Van Camp G (2011) A new locus for otosclerosis, OTSC10, maps to chromosome 1q41-44. Clin Genet 79:495–497. https://doi.org/10.1111/j.1399-0004.2010.01576.x
Schrauwen I, Khalfallah A, Ealy M, Fransen E, Claes C, Huber A, Murillo LR, Masmoudi S, Smith RJ, Van Camp G (2012) COL1A1 association and otosclerosis: a meta-analysis. Am J Med Genet A 158a:1066–1070. https://doi.org/10.1002/ajmg.a.35276
Schrauwen I, Valgaeren H, Tomas-Roca L, Sommen M, Altunoglu U, Wesdorp M, Beyens M, Fransen E, Nasir A, Vandeweyer G, Schepers A, Rahmoun M, van Beusekom E, Huentelman MJ, Offeciers E, Dhooghe I, Huber A, Van de Heyning P, Zanetti D, De Leenheer EMR, Gilissen C, Hoischen A, Cremers CW, Verbist B, de Brouwer APM, Padberg GW, Pennings R, Kayserili H, Kremer H, Van Camp G, van Bokhoven H (2019) Variants affecting diverse domains of MEPE are associated with two distinct bone disorders, a craniofacial bone defect and otosclerosis. Genet Med 21:1199–1208. https://doi.org/10.1038/s41436-018-0300-5
Sommen M, Van Camp G, Liktor B, Csomor P, Fransen E, Sziklai I, Schrauwen I, Karosi T (2014) Genetic association analysis in a clinically and histologically confirmed otosclerosis population confirms association with the TGFB1 gene but suggests an association of the RELN gene with a clinically indistinguishable otosclerosis-like phenotype. Otol Neurotol 35:1058–1064. https://doi.org/10.1097/mao.0000000000000334
Stattin E-L, Wiklund F, Lindblom K, Onnerfjord P, Jonsson B-A, Tegner Y, Sasaki T, Struglics A, Lohmander S, Dahl N, Heinegård D, Aspberg A (2010) A missense mutation in the aggrecan C-type lectin domain disrupts extracellular matrix interactions and causes dominant familial osteochondritis dissecans. Am J Hum Genet 86:126–137. https://doi.org/10.1016/j.ajhg.2009.12.018
Stavber L, Hovnik T, Kotnik P, Lovrecic L, Kovac J, Tesovnik T, Bertok S, Dovc K, Debeljak M, Battelino T, Avbelj Stefanija M (2020) High frequency of pathogenic ACAN variants including an intragenic deletion in selected individuals with short stature. Eur J Endocrinol 182:243–253. https://doi.org/10.1530/eje-19-0771
Sykes B, Ogilvie D, Wordsworth P, Anderson JN (1986) Osteogenesis imperfecta is linked to both type I collagen structural genes. Lancet 2:69–72
Sykes B, Ogilvie D, Wordsworth P, Wallis G, Mathew C, Beighton P, Nicholls A, Pope FM, Thompson E, Tsipouras P et al (1990) Consistent linkage of dominantly inherited osteogenesis imperfecta to the type I collagen loci: COL1A1 and COL1A2. Am J Hum Genet 46:293–307
The Genomes Project C (2015) A global reference for human genetic variation. Nature 526: 68. https://doi.org/10.1038/nature15393. https://www.nature.com/articles/nature15393#supplementary-information
Thorisson GA, Smith AV, Krishnan L, Stein LD (2005) The International HapMap Project Web site. Genome Res 15:1592–1593. https://doi.org/10.1101/gr.4413105
Thys M, Schrauwen I, Vanderstraeten K, Janssens K, Dieltjens N, Van Den Bogaert K, Fransen E, Chen W, Ealy M, Claustres M, Cremers CR, Dhooge I, Declau F, Claes J, Van de Heyning P, Vincent R, Somers T, Offeciers E, Smith RJ, Van Camp G (2007a) The coding polymorphism T263I in TGF-beta1 is associated with otosclerosis in two independent populations. Hum Mol Genet 16:2021–2030. https://doi.org/10.1093/hmg/ddm150
Thys M, Van Den Bogaert K, Iliadou V, Vanderstraeten K, Dieltjens N, Schrauwen I, Chen W, Eleftheriades N, Grigoriadou M, Pauw RJ, Cremers CRWJ, Smith RJH, Petersen MB, Van Camp G (2007b) A seventh locus for otosclerosis, OTSC7, maps to chromosome 6q13–16.1. Eur J Hum Genet 15:362–368. https://doi.org/10.1038/sj.ejhg.5201761
Tomek MS, Brown MR, Mani SR, Ramesh A, Srisailapathy CR, Coucke P, Zbar RI, Bell AM, McGuirt WT, Fukushima K, Willems PJ, Van Camp G, Smith RJ (1998) Localization of a gene for otosclerosis to chromosome 15q25-q26. Hum Mol Genet 7:285–290. https://doi.org/10.1093/hmg/7.2.285
Untergasser A, Cutcutache I, Koressaar T, Ye J, Faircloth BC, Remm M, Rozen SG (2012) Primer3–new capabilities and interfaces. Nucleic Acids Res 40:e115–e115. https://doi.org/10.1093/nar/gks596
Valgaeren H, Sommen M, Beyens M, Vandeweyer G, Schrauwen I, Schepers A, Schatteman I, Topsakal V, Dhooge I, Kunst H, Zanetti D, Huber AM, Hoischen A, Fransen E, Van Camp G (2019) Insufficient evidence for a role of SERPINF1 in otosclerosis. Mol Genet Genomics 294:1001–1006. https://doi.org/10.1007/s00438-019-01558-8
Valhmu WB, Palmer GD, Dobson J, Fischer SG, Ratcliffe A (1998) Regulatory activities of the 5′- and 3′-untranslated regions and promoter of the human aggrecan gene. J Biol Chem 273:6196–6202. https://doi.org/10.1074/jbc.273.11.6196
Van Den Bogaert K, Govaerts PJ, Schatteman I, Brown MR, Caethoven G, Offeciers FE, Somers T, Declau F, Coucke P, Van de Heyning P, Smith RJ, Van Camp G (2001) A second gene for otosclerosis, OTSC2, maps to chromosome 7q34-36. Am J Hum Genet 68:495–500. https://doi.org/10.1086/318185
Van Den Bogaert K, De Leenheer EMR, Chen W, Lee Y, Nürnberg P, Pennings RJE, Vanderstraeten K, Thys M, Cremers CWRJ, Smith RJH, Van Camp G (2004) A fifth locus for otosclerosis, OTSC5, maps to chromosome 3q22-24. J Med Genet 41:450–453. https://doi.org/10.1136/jmg.2004.018671
Watanabe H, Kimata K, Line S, Strong D, Gao LY, Kozak CA, Yamada Y (1994) Mouse cartilage matrix deficiency (cmd) caused by a 7 bp deletion in the aggrecan gene. Nat Genet 7:154–157. https://doi.org/10.1038/ng0694-154
Yoo TJ, Cho H, Yamada Y (1991) Hearing impairment in mice with the cmd/cmd (cartilage matrix deficiency) mutant gene. Ann NY Acad Sci 630:265–267. https://doi.org/10.1111/j.1749-6632.1991.tb19600.x
Ziff JL, Crompton M, Powell HR, Lavy JA, Aldren CP, Steel KP, Saeed SR, Dawson SJ (2016) Mutations and altered expression of SERPINF1 in patients with familial otosclerosis. Hum Mol Genet 25:2393–2403. https://doi.org/10.1093/hmg/ddw106
Acknowledgements
We would like to acknowledge Robert Vincent (Causse Ear Clinic, France) and Marcel Cosgarea (Iuliu Hatieganu University of Medicine and Pharmacy, Romania) for providing patients for this study. This study was supported by funding from the Belgian Science Policy Office Interuniversity Attraction Poles (BELSPO-IAP) program (project IAP P7/43-BeMGI to G.V.C.) and the Obelske Familiefond (to A.T.H. and M.B.P.).
Funding
This study was supported by funding from the Belgian Science Policy Office Interuniversity Attraction Poles (BELSPO-IAP) program (project IAP P7/43-BeMGI to G.V.C.) and the Obelske Familiefond (to A.T.H. and M.B.P.).
Author information
Authors and Affiliations
Contributions
GVC and EF conceived and designed the study and were in charge of overall direction and planning. VT, IS, ID, AH, DZ and HPMK recruited patients and controls in the participating centers and collected samples. ATH, MS, and AH designed the smMIPs experiment. ATH, LJMT, and MS performed the laboratory experiments and collected data in the lab. EF performed the statistical analyses. ATH, LJMT, IS, GVC and EF all contributed to the interpretation of the results. ATH and LJMT have drafted the manuscript and IS, MBP, GVC, and EF substantially revised it. All authors critically read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Ethics approval and consent to participate
Informed consent was obtained from all participants and local ethics committees approved all procedures in accordance with the World Medical Association’s Declaration of Helsinki.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Højland, A.T., Tavernier, L.J.M., Schrauwen, I. et al. A wide range of protective and predisposing variants in aggrecan influence the susceptibility for otosclerosis. Hum Genet 141, 951–963 (2022). https://doi.org/10.1007/s00439-021-02334-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-021-02334-8