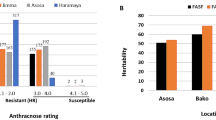
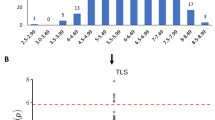
Abstract
Witchweeds (Striga spp.) greatly limit production of Africa’s most staple crops. These parasitic plants use strigolactones (SLs)—chemical germination stimulants, emitted from host’s roots to germinate, and locate their hosts for invasion. This information exchange provides opportunities for controlling the parasite by either stimulating parasite seed germination without a host (suicidal germination) or by inhibiting parasite seed germination (pre-attachment resistance). We sought to determine genetic factors that underpin Striga pre-attachment resistance in sorghum using the genome wide association study (GWAS) approach. Results revealed that Striga germination was associated with genes encoding hormone signaling functions, e.g., the Novel interactor of jaz (NINJA) and, Abscisic acid-insensitive 5 (ABI5). This pointed toward abscisic acid (ABA) and gibberellic acid (GA) as probable determinants of Striga germination. To test this hypothesis, we conditioned Striga using: ABA, ABA + its inhibitor fluridone (FLU), GA or water. Unexpectedly, Striga conditioned with FLU germinated after 4 days without SL. Upon germination stimulation using sorghum root exudate or the synthetic SL GR24, we found that ABA conditioned seeds had above 20-fold reduction in germination. Conversely, FLU conditioned seeds recorded above 20-fold increase in germination. Conditioning with GA reduced Striga seed germination 1.5-fold only in the GR24 treatment. Germination assays using seeds of a related parasitic plant (Alectra vogelii) showed similar degrees of stimulation and reduction of germination by the hormones further affirming the hormonal crosstalk. Our findings have far-reaching implications in the control of some of the most noxious pathogens of crops in Africa.






Similar content being viewed by others
Data availability
The datasets generated or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- ABI5:
-
Abscisic acid-insensitive 5
- GA:
-
Gibberellic acid (GA)
- GWAS:
-
Genome wide association study
- NINJA:
-
Novel interactor of jaz
- SL:
-
Strigolactones
References
Alexander DH, Novembre J, Lange K (2009) Fast model-based estimation of ancestry in unrelated individuals. Genome Res 19:1655–1664. https://doi.org/10.1101/gr.094052.109
Arroyo A, Bossi F, Finkelstein RR, León P (2003) Three genes that affect sugar sensing (abscisic acid insensitive 4, abscisic acid insensitive 5, and constitutive triple response 1) are differentially regulated by glucose in arabidopsis. Plant Physiol 133:231–242. https://doi.org/10.1104/pp.103.021089
Barrett JC, Fry B, Maller J, Daly MJ (2005) Haploview: analysis and visualization of LD and haplotype maps. Bioinformatics 21:263–265. https://doi.org/10.1093/bioinformatics/bth457
Bellis ES, Kelly EA, Lorts CM, et al (2020) Genomics of sorghum local adaptation to a parasitic plant. Proc Natl Acad Sci USA:201908707
Bunsick M, Toh S, Wong C et al (2020) SMAX1-dependent seed germination bypasses GA signalling in Arabidopsis and Striga. Nat Plants 6:646–652. https://doi.org/10.1038/s41477-020-0653-z
Chen M, Zhang B, Li C et al (2015) TRANSPARENT TESTA GLABRA1 regulates the accumulation of seed storage reserves in arabidopsis. Plant Physiol 169:391–402. https://doi.org/10.1104/pp.15.00943
Ejeta G (2007) The Striga scourge in Africa: a growing pandemic. In: Ejeta G, Gressel J (eds). World Scientific Publishing Company, pp 3-- 16
Gabriel SB, Schaffner SF, Nguyen H et al (2002) The structure of haplotype blocks in the human genome. Science 296:2225–2229. https://doi.org/10.1126/science.1069424
Glaubitz JC, Casstevens TM, Lu F et al (2014) TASSEL-GBS: a high capacity genoty** by sequencing analysis pipeline. PLoS One 9:e90346. https://doi.org/10.1371/journal.pone.0090346
Gobena D, Shimels M, Rich PJ et al (2017) mutation in sorghum low germination stimulant 1alters strigolactones and causes strigaresistance. Proc Natl Acad Sci 114:4471–4476. https://doi.org/10.1073/pnas.1618965114
Hess DE, Ejeta G (1992) Inheritance of resistance to striga in sorghum genotype SRN39. Plant Breed 109:233–241. https://doi.org/10.1111/j.1439-0523.1992.tb00178.x
Ito S, Yamagami D, Umehara M et al (2017) Regulation of strigolactone biosynthesis by gibberellin signaling. Plant Physiol 174:1250–1259. https://doi.org/10.1104/pp.17.00301
Jombart T (2008) adegenet: a R package for the multivariate analysis of genetic markers. Bioinform Oxf Engl 24:1403–1405. https://doi.org/10.1093/bioinformatics/btn129
Jombart T, Balloux F, Dray S (2010) adephylo: new tools for investigating the phylogenetic signal in biological traits. Bioinformatics 26:1907–1909. https://doi.org/10.1093/bioinformatics/btq292
Kavuluko J, Kibe M, Sugut I et al (2021) GWAS provides biological insights into mechanisms of the parasitic plant (Striga) resistance in sorghum. Bmc Plant Biol 21:392. https://doi.org/10.1186/s12870-021-03155-7
Khan ZR, Midega CAO, Pittchar JO et al (2014) Achieving food security for one million sub-Saharan African poor through push–pull innovation by 2020. Philos Trans Ro Soc B Biol Sci 369:20120284–20120284. https://doi.org/10.1098/rstb.2012.0284
Kountche BA, Jamil M, Yonli D et al (2019) Suicidal germination as a control strategy for Striga hermonthica (Benth.) in smallholder farms of sub-Saharan Africa. Plants People Planet 1:107–118. https://doi.org/10.1002/ppp3.32
Kusumoto D, Chae SH, Mukaida K et al (2006) Effects of fluridone and norflurazon on conditioning and germination of striga asiatica seeds. Plant Growth Regul 48:73–78. https://doi.org/10.1007/s10725-005-4746-5
Liu D, Wang L, Zhai H et al (2014) A novel α/β-hydrolase gene IbMas enhances salt tolerance in transgenic sweetpotato. PLoS One 9:e115128. https://doi.org/10.1371/journal.pone.0115128
Liu X, Huang M, Fan B et al (2016) Iterative usage of fixed and random effect models for powerful and efficient genome-wide association studies. Plos Genet 12:e1005767. https://doi.org/10.1371/journal.pgen.1005767
Mallu TS, Mutinda S, Githiri SM et al (2021) New pre-attachment Striga resistant sorghum adapted to African agro-ecologies. Pest Manag Sci. https://doi.org/10.1002/ps.6325
Marzec M (2017) Strigolactones and gibberellins: a new couple in the phytohormone world? Trends Plant Sci 22:813–815. https://doi.org/10.1016/j.tplants.2017.08.001
Matusova R, Rani K, Verstappen FWA et al (2005) The strigolactone germination stimulants of the plant-parasitic striga and Orobanche spp. are derived from the carotenoid pathway. Plant Physiol 139:920–934. https://doi.org/10.1104/pp.105.061382
Mohemed N, Charnikhova T, Fradin EF et al (2018) Genetic variation in Sorghum bicolor strigolactones and their role in resistance against Striga hermonthica. J Exp Bot 69:2415–2430. https://doi.org/10.1093/jxb/ery041
Morris GP, Ramu P, Deshpande SP et al (2013) Population genomic and genome-wide association studies of agroclimatic traits in sorghum. Proc National Acad Sci 110:453–458. https://doi.org/10.1073/pnas.1215985110
Mwakha FA, Budambula NLM, Neondo JO et al (2020) Witchweed’s suicidal germination: can slenderleaf help? Agronomy 10:873. https://doi.org/10.3390/agronomy10060873
Pauwels L, Barbero GF, Geerinck J et al (2010) NINJA connects the co-repressor TOPLESS to jasmonate signalling. Nature 464:788–791. https://doi.org/10.1038/nature08854
Pruim RJ, Welch RP, Sanna S et al (2010) LocusZoom: regional visualization of genome-wide association scan results. Bioinformatics 26:2336–2337. https://doi.org/10.1093/bioinformatics/btq419
Revell LJ (2012) phytools: an R package for phylogenetic comparative biology (and other things). Methods Ecol Evol 3:217–223. https://doi.org/10.1111/j.2041-210x.2011.00169.x
Runo S, Kuria EK (2018) Habits of a highly successful cereal killer. Striga Plos Pathog 14:e1006731. https://doi.org/10.1371/journal.ppat.1006731
Sansaloni C, Petroli C, Jaccoud D et al (2011) Diversity arrays technology (DArT) and next-generation sequencing combined: genome-wide, high throughput, highly informative genoty** for molecular breeding of Eucalyptus. BMC Proc 5:P54. https://doi.org/10.1186/1753-6561-5-s7-p54
Scheet P, Stephens M (2006) A fast and flexible statistical model for large-scale population genotype data: applications to inferring missing genotypes and haplotypic phase. Am J Hum Genet 78:629–644. https://doi.org/10.1086/502802
Segura V, Vilhjálmsson BJ, Platt A et al (2012) An efficient multi-locus mixed-model approach for genome-wide association studies in structured populations. Nat Genet 44:825–830. https://doi.org/10.1038/ng.2314
Seo M, Nambara E, Choi G, Yamaguchi S (2009) Interaction of light and hormone signals in germinating seeds. Plant Mol Biol 69:463–472. https://doi.org/10.1007/s11103-008-9429-y
Shang Y, Yang D, Ha Y et al (2019) RPK1 and BAK1 sequentially form complexes with OST1 to regulate ABA-induced stomatal closure. J Exp Bot. https://doi.org/10.1093/jxb/erz489
Skubacz A, Daszkowska-Golec A, Szarejko I (2016) The role and regulation of ABI5 (ABA-Insensitive 5) in plant development, abiotic stress responses and phytohormone crosstalk. Front Plant Sci 7:1884. https://doi.org/10.3389/fpls.2016.01884
Tang Y, Liu X, Wang J et al (2016) GAPIT version 2: an enhanced integrated tool for genomic association and prediction. Plant Genom. https://doi.org/10.3835/plantgenome2015.11.0120
Toh S, Kamiya Y, Kawakami N et al (2012) Thermoinhibition uncovers a role for strigolactones in arabidopsis seed germination. Plant Cell Physiol 53:107–117. https://doi.org/10.1093/pcp/pcr176
Uraguchi D, Kuwata K, Hijikata Y et al (2018) A femtomolar-range suicide germination stimulant for the parasitic plant Striga hermonthica. Science 362:1301–1305. https://doi.org/10.1126/science.aau5445
Wang W, Messing J (2012) Analysis of ADP-glucose pyrophosphorylase expression during turion formation induced by abscisic acid in Spirodela polyrhiza (greater duckweed). BMC Plant Biol 12:5. https://doi.org/10.1186/1471-2229-12-5
Wang F, Niu H, **n D, et al (2021) A novel maspardin protein gene OsMas is involved in salt and drought tolerance in rice. https://doi.org/10.21203/rs.3.rs-778487/v1
Wickham H (2016) ggplot2: elegant graphics for data analysis. Springer-Verlag, New York
**e Z, Nolan TM, Jiang H, Yin Y (2019) AP2/ERF transcription factor regulatory networks in hormone and abiotic stress responses in arabidopsis. Front Plant Sci 10:228. https://doi.org/10.3389/fpls.2019.00228
Yamaguchi S, Kamiya Y, Nambara E (2007) Regulation of ABA and GA levels during seed development and germination in Arabidopsis. In: Bradford KJ, Nonogaki H (eds) Seed development, dormancy and germination. Blackwell Publishing Ltd., Oxford, pp 224–247
Acknowledgements
The research leading to these results received funding from the National Academies of Science (NAS) under the Partnerships for Enhanced Engagement in Research (PEER) program, Grant/Award Number: PGA- 2000008288; NAS and the United States Agency for International Development, Grant/Award Number: AID-OAA-A-11-00012. ICRISAT, Eastern and Southern Africa provided funding to conduct DArT seq. TM’s PhD fellowship is sponsored by the Pan African University (PAUSTI) through an African Union Initiative.
Funding
National Academies of Science (NAS) under the Partnerships for Enhanced Engagement in Research (PEER) program, Grant/Award Number: PGA- 2000008288; NAS and the United States Agency for International Development, Grant/Award Number: AID-OAA-A-11–00012.
Author information
Authors and Affiliations
Contributions
SR and DAO conceived and designed the study. TSM, GI and SM performed Striga and Alectra germination and GWAS analysis guided by SR, DAO and SMG. EO carried out analysis of sequence data, quality control and participated in the GWAS analysis. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflicts of interest
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
438_2022_1882_MOESM1_ESM.tif
Suppl. Fig. S1 Geographic origins of the sorghum used in the GWAS study. Categories are based on the major sorghum races (TIF 9498 kb)
438_2022_1882_MOESM2_ESM.tif
Suppl Fig. 2 Bayesian Information Criterion (BIC) identification of optimal K clustering. The lowest K (K=8) is indicated by an elbow curve of BIC values (TIF 2850 kb)
438_2022_1882_MOESM3_ESM.tif
Suppl Fig. S3 Hierarchical Bayesian clustering of sorghum accessions used in the GWAS study using various K values K=7; (CV=0.35293 ) K = 8 (CV=0.35673) and K = 9 (CV=0.36229) (TIF 6641 kb)
Rights and permissions
About this article
Cite this article
Mallu, T.S., Irafasha, G., Mutinda, S. et al. Mechanisms of pre-attachment Striga resistance in sorghum through genome-wide association studies. Mol Genet Genomics 297, 751–762 (2022). https://doi.org/10.1007/s00438-022-01882-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-022-01882-6