Abstract
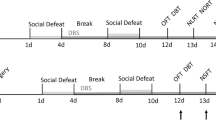
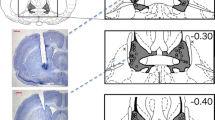
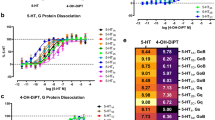
Electrical stimulation of the dorsolateral periaqueductal gray (dlPAG) in rats has been shown to elicit panic-like behaviour and can be a useful as an unconditioned stimulus for modelling anticipatory fear and agoraphobia in a contextual fear conditioning paradigm. In this study, we further analysed our previous data on the effects of escitalopram (a selective serotonin reuptake inhibitor, SSRI) and buspirone (a 5-HT1A receptor partial agonist) on dlPAG-induced anticipatory fear behaviour in a rat model using freezing as a measure. We then attempted to unravel some of the interactions with dopamine signalling using tyrosine hydroxylase (TH) immunohistochemistry to probe the effects on dopaminergic neurons. We showed that acute treatment of escitalopram, but not buspirone, was effective in reducing anticipatory freezing behaviour, while chronic administrations of both drugs were effective. We found that the dlPAG stimulation induced increase number of dopaminergic neurons in the ventral tegmental area (VTA) which was reversed in both chronic buspirone and escitalopram groups. We further found a strong positive correlation between the number of dopaminergic neurons and freezing in the VTA and showed positive correlations between dopaminergic neurons in the VTA and substantia nigra pars compacta (SNpc) in escitalopram and buspirone groups, respectively. Overall, we showed that chronic treatment with an SSRI and a 5-HT1A agonist reduced anticipatory freezing behaviour which seems to be associated, through correlative studies, with a reversal of dlPAG stimulation induced increase in number of dopaminergic neurons in the VTA and/or SNpc.



Similar content being viewed by others
References
Abraham AD, Neve KA, Lattal KM (2014) Dopamine and extinction: A convergence of theory with fear and reward circuitry. Neurobiol Learn Mem. https://doi.org/10.1016/j.nlm.2013.11.007.Dopamine
Azkona G, Sagarduy A, Aristieta A et al (2014) Buspirone anti-dyskinetic effect is correlated with temporal normalization of dysregulated striatal DRD1 signalling in l-DOPA-treated rats. Neuropharmacology 79:726–737. https://doi.org/10.1016/j.neuropharm.2013.11.024
Blanchard RJ, Blanchard DC (1969) Crouching as an index of fear. J Comp Physiol Psychol 67:370. https://doi.org/10.1037/h0026779
Di CP, Cormick PM, Stefan C, Wong E (2017) The effects of buspirone on occupancy of dopamine receptors and the rat gambling task. Psychopharmacology 234:3309–3320. https://doi.org/10.1007/s00213-017-4715-5
Daw ND, Kakade S, Dayan P (2002) Opponent interactions between serotonin and dopamine. Neural Networks 15:603–616
De OBM, Decola JP, Maren S, Fanselow MS (1998) Distinct regions of the periaqueductal gray are involved in the acquisition and expression of defensive responses. J Neurosci 18:3426–3432
Dremencov E, El MM, Blier P (2009) Effects of sustained serotonin reuptake inhibition on the firing of dopamine neurons in the rat ventral tegmental area. J Psychiatry Neurosci 34:223–229
Eilam D (2005) Die hard: A blend of freezing and fleeing as a dynamic defense - Implications for the control of defensive behavior. Neurosci Biobehav Rev 29:1181–1191. https://doi.org/10.1016/j.neubiorev.2005.03.027
Esposito E, Di MV, Di GG et al (2008) Serotonin – dopamine interaction : an overview. Prog Brain Res 172:3–6. https://doi.org/10.1016/S0079-6123(08)00901-1
Fadok JP, Dickerson TMK, Palmiter RD (2009) Dopamine is necessary for cue-dependent fear conditioning. J Neurosci 29:11089–11097. https://doi.org/10.1523/JNEUROSCI.1616-09.2009
Feria-velasco A, Gonza I (2008) Serotonin / dopamine interaction in memory formation. Prog Brain Res 172:603–623. https://doi.org/10.1016/S0079-6123(08)00928-X
Herry C, Ferraguti F, Singewald N et al (2010) Neuronal circuits of fear extinction. Eur J Neurosci 31:599–612. https://doi.org/10.1111/j.1460-9568.2010.07101.x
Hogg S, Michan L, Jessa M (2006) Prediction of anti-panic properties of escitalopram in the dorsal periaqueductal grey model of panic anxiety. Neuropharmacology 51:141–145. https://doi.org/10.1016/j.neuropharm.2006.03.009
Ivanov AV, Konradsson-geuken A (2011) Effects of S-Citalopram, citalopram, and R-citalopram on the firing patterns of dopamine neurons in the ventral tegmental area, N -methyl- D -aspartate receptor-mediated transmission in the medial prefrontal cortex and cognitive function in the rat. Synapse 65:357–367. https://doi.org/10.1002/syn.20853
Jahanshahi A, Wei L, Steinbusch HWM et al (2010) Neuroscience Letters Buspirone-induced changes in the serotonergic and non-serotonergic cells in the dorsal raphe nucleus of rats. Neurosci Lett 473:136–140. https://doi.org/10.1016/j.neulet.2010.02.038
Jenck F, Broekkamp CLE, Van DAML (1990) The effect of antidepressants on aversive periaqueductal gray stimulation in rats. Eur J Pharmacol 177:201–204
Jenck F, Moreau J, Berendsen HHG et al (1998) Antiaversive effects of 5HT 2C receptor agonists and fluoxetine in a model of panic-like anxiety in rats. Eur Neuropsychopharmacol 8:161–168
Jenck F, Moreau J, Martin JR (1995) Dorsal periaqueductal gray-induced aversion as a simulation of panic anxiety : Elements of face and predictive validity. Psychiatry Res 57:181–191
Kakui N, Yokoyama F, Yamauchi M et al (2009) Anxiolytic-like profile of mirtazapine in rat conditioned fear stress model: Functional significance of 5-hydroxytryptamine 1A receptor and α1-adrenergic receptor. Pharmacol Biochem Behav 92:393–398. https://doi.org/10.1016/j.pbb.2008.12.022
Lechin F, Van Der DB, Jara H et al (1998) Effects of buspirone on plasma neurotransmitters in healthy subjects. J Neural Transm 105:561–573
Lim LW, Blokland A, Van DM et al (2011) Increased plasma corticosterone levels after periaqueductal gray stimulation-induced escape reaction or panic attacks in rats. Behav Brain Res 218:301–307. https://doi.org/10.1016/j.bbr.2010.12.026
Lim LW, Blokland A, Tan S et al (2010) Attenuation of fear-like response by escitalopram treatment after electrical stimulation of the midbrain dorsolateral periaqueductal gray. Exp Neurol 226:293–300. https://doi.org/10.1016/j.expneurol.2010.08.035
Lim LW, Blokland A, Visser-Vandewalle V et al (2008) High-frequency stimulation of the dorsolateral periaqueductal gray and ventromedial hypothalamus fails to inhibit panic-like behaviour. Behav Brain Res 193:197–203. https://doi.org/10.1016/j.bbr.2008.05.020
Lim LW, Temel Y, Visser-Vandewalle V et al (2009) Fos immunoreactivity in the rat forebrain induced by electrical stimulation of the dorsolateral periaqueductal gray matter. J Chem Neuroanat 38:83–96. https://doi.org/10.1016/j.jchemneu.2009.06.011
Liu A, Jain N, Vyas A, Lim LW (2015) Ventromedial prefrontal cortex stimulation enhances memory and hippocampal neurogenesis in the middle-aged rats. Elife. https://doi.org/10.7554/eLife.04803
Lugenbiel P, Sartorius A, Vollmayr B, Schloss P (2010) Creatine transporter expression after antidepressant therapy in rats bred for learned helplessness. World J Biol Psychiatry 11:329–333. https://doi.org/10.3109/15622970903131597
Luo R, Uematsu A, Weitemier A et al (2018) A dopaminergic switch for fear to safety transitions. Nat Commun 9:1–11. https://doi.org/10.1038/s41467-018-04784-7
MacGillivray L, Reynolds KB, Sickand M et al (2011) Inhibition of the serotonin transporter induces microglial activation and downregulation of dopaminergic neurons in the substantia nigra. Synapse 65:1166–1172. https://doi.org/10.1002/syn.20954
McIntyre CC, Anderson RW (2016) Deep brain stimulation mechanisms: the control of network activity via neurochemistry modulation. J Neurochem 139:338–345. https://doi.org/10.1111/jnc.13649
Motulsky HJ, Brown RE (2006) Detecting outliers when fitting data with nonlinear regression - A new method based on robust nonlinear regression and the false discovery rate. BMC Bioinformatics 7:1–20. https://doi.org/10.1186/1471-2105-7-123
Paxinos G, Watson C (2006) The rat brain in stereotaxic coordinates sixth edition by. Acad Press 170:547612. 10.1016/0143–4179(83)90049–5
Pezze MA, Feldon J (2004) Mesolimbic dopaminergic pathways in fear conditioning. Prog Neurobiol 74:301–320. https://doi.org/10.1016/j.pneurobio.2004.09.004
Pignatelli M, Bonci A (2015) Review role of dopamine neurons in reward and aversion : a synaptic plasticity perspective. Neuron 86:1145–1157. https://doi.org/10.1016/j.neuron.2015.04.015
Sagarduy A, Llorente J, Miguelez C et al (2016) Buspirone requires the intact nigrostriatal pathway to reduce the activity of the subthalamic nucleus via 5-HT1A receptors. Exp Neurol 277:35–45. https://doi.org/10.1016/j.expneurol.2015.12.005
Sánchez C, Bergqvist PBF, Brennum LT et al (2003) Escitalopram, the S-(+)-enantiomer of citalopram, is a selective serotonin reuptake inhibitor with potent effects in animal models predictive of antidepressant and anxiolytic activities. Psychopharmacology 167:353–362. https://doi.org/10.1007/s00213-002-1364-z
Schenberg LC, Capucho LB, Vatanabe RO, Vargas LC (2002) Acute effects of clomipramine and fluoxetine on dorsal periaqueductal grey-evoked unconditioned defensive behaviours of the rat. Psychopharmacology 159:138–144. https://doi.org/10.1007/s002130100883
Sharifi H, Nayebi AM, Farajnia S (2012) The effect of chronic administration of buspirone on 6-hydroxydopamine-induced catalepsy in rats. Adv Pharm Bull 2:127–131. https://doi.org/10.5681/apb.2012.019
Tan SZK, Poon CH, Chan Y-S, Lim LW (2019a) Deep brain stimulation of the ventromedial prefrontal cortex disrupts consolidation of fear memories. bioRxiv 537514. 10.1101/537514
Tan SZK, Sheng V, Chan Y-S, Lim LW (2019) Eternal sunshine of the neuromodulated mind: altering fear memories through neuromodulation. Exp Neurol 314:9–19
Temel Y, Boothman LJ, Blokland A et al (2007) Inhibition of 5-HT neuron activity and induction of depressive-like behavior by high-frequency stimulation of the subthalamic nucleus. Proc Natl Acad Sci U S A 104:17087–17092. https://doi.org/10.1073/pnas.0704144104
Tunnicliff G, Brokaw JJ, Hausz JA et al (1992) Influence of repeated treatment with buspirone on central 5-hydroxytryptamine and dopamine synthesis. Neuropharmacology 31:991–995. https://doi.org/10.1016/0028-3908(92)90099-B
Walker DL, Davis M (1997) Involvement of the dorsal periaqueductal gray in the loss of fear-potentiated startle accompanying high footshock training. Behav Neurosci 111:692–702
Wickham H (2016) ggplot2: Elegant Graphics for Data Analysis. Springer-Verlag, New York
Zanoveli JM, Nogueira RL, Zangrossi H (2005) Chronic imipramine treatment sensitizes 5-HT1A and 5-HT 2A receptors in the dorsal periaqueductal gray matter: Evidence from the elevated T-maze test of anxiety. Behav Pharmacol 16:543–552. https://doi.org/10.1097/01.fbp.0000179280.05654.5a
Funding
The scientific work was funded by grants from the Hong Kong Research Grants Council and the University of Hong Kong Seed Fund for Basic Research awarded to L.W.L. W.L.L. was the recipient of the International Brain Research Organization-Asia Pacific Regional Committee (IBRO-APRC) Exchange Fellowship to work on this project.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
All authors declared no biomedical financial interests or potential conflicts of interests.
Ethical statement
All animal works was approved by the Animal Experiments and Ethics Committee of Maastricht University, as well as the Committee on the Use of Live Animals in Teaching and Research (CULATR) of The University of Hong Kong.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
429_2020_2102_MOESM1_ESM.pdf
Supplementary Fig. 1 Heat plot of p-values from Tukey’s test. numbers in the table are p-values from Tukey’s post-hoc tests from the ANOVAs. The heatmaps shows the levels of significance (red represents more significance and green represents less significance) (PDF 131 kb)
Rights and permissions
About this article
Cite this article
Tan, S.Z.K., Temel, Y., Chan, A.Y. et al. Serotonergic treatment normalizes midbrain dopaminergic neuron increase after periaqueductal gray stimulation. Brain Struct Funct 225, 1957–1966 (2020). https://doi.org/10.1007/s00429-020-02102-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-020-02102-w