Abstract
Purpose
Frailty is characterized by fragility and decline in physical, mental, and social activities; it is commonly observed in older adults. No studies have reported frailty status changes between the preoperative and postoperative periods, including mental and cognitive factors. Therefore, this study investigated frailty factors, including mental and cognitive functions, that change after non-cardiac surgery in older adults.
Methods
Patients aged ≥ 75 years who underwent non-cardiac surgery were surveyed using five tools (Eastern Cooperative Oncology Group-Performance Status (PS); handgrip strengths; Japan-Cardiovascular Health Study index (J-CHS index); Mini-Mental State Examination (MMSE); and Geriatric Depression Scale) for comprehensive evaluation of perioperative functions. The results before surgery, at discharge, and during follow-up at the outpatient clinic were compared.
Results
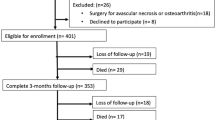
Fifty-three patients with a median age of 80 (IQR, 77–84) years were evaluated. MMSE scores did not change during the perioperative period. The PS and J-CHS index worsened significantly at discharge and did not improve at the outpatient clinic follow-up. The dominant handgrip strength decreased after surgery (p < 0.001) but improved during follow-up. Additionally, nondominant handgrip strength decreased after surgery (p < 0.001) but did not recover as much as the dominant handgrip strength during follow-up (p = 0.015).
Conclusion
Changes in physical frailty and mental and cognitive functions were not identical perioperatively in older adult patients undergoing non-cardiac surgery. Physical frailty did not improve 1 month after surgery, mental function recovered early, and cognitive function did not decline. This study may be important for frailty prevention in older adult patients.





Similar content being viewed by others
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Lin HS, Watts JN, Peel NM, Hubbard RE (2016) Frailty and post-operative outcomes in older surgical patients: a systematic review. BMC Geriatr 16:157–169. https://doi.org/10.1186/s12877-016-0329-8
Ligthart-Melis GC, Luiking YC, Kakourou A, Cederholm T, Maier AB, de van der Schueren MAE (2020) Frailty, sarcopenia, and malnutrition frequently (co-)occur in hospitalized older adults: a systematic review and meta-analysis. J Am Med Dir Assoc 21:1216–1228. https://doi.org/10.1016/j.jamda.2020.03.006
Fried LP, Tangen CM, Walston J et al (2001) Frailty in older adults: evidence for a phenotype. J Gerontol A Biol Sci Med Sci 56:M146–M156. https://doi.org/10.1093/gerona/56.3.m146
Ticinesi A, Tana C, Nouvenne A, Prati B, Lauretani F, Meschi T (2018) Gut microbiota, cognitive frailty and dementia in older individuals: a systematic review. Clin Interv Aging 13:1497–1511. https://doi.org/10.2147/cia.s139163
Gray SL, Anderson ML, Hubbard RA, LaCroix A, Crane PK, McCormick W, Bowen JD, McCurry SM, Larson EB (2013) Frailty and incident dementia. J Gerontol A Biol Sci Med Sci 68:1083–1090. https://doi.org/10.1093/gerona/glt013
de Vries NM, Staal JB, van Ravensberg CD, Hobbelen JSM, Olde Rikkert MGM, Nijhuis-van der Sanden MWG (2011) Outcome instruments to measure frailty: a systematic review. Ageing Res Rev 10:104–114. https://doi.org/10.1016/j.arr.2010.09.001
Roberts HC, Denison HJ, Martin HJ, Patel HP, Syddall H, Cooper C, Sayer AA (2011) A review of the measurement of grip strength in clinical and epidemiological studies: towards a standardised approach. Age Ageing 40:423–429. https://doi.org/10.1093/ageing/afr051
Welch C, Greig CA, Hassan-Smith ZK, Pinkney TD, Lord JM, Jackson TA (2019) A pilot observational study measuring acute sarcopenia in older colorectal surgery patients. BMC Res Notes 12:24. https://doi.org/10.1186/s13104-019-4049-y
Satake S, Shimada H, Yamada M et al (2017) Prevalence of frailty among community-dwellers and outpatients in Japan as defined by the Japanese version of the Cardiovascular Health Study criteria. Geriatr Gerontol Int 17:2629–2634. https://doi.org/10.1111/ggi.13129
Folstein MF, Folstein SE, McHugh PR (1975) “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 12:189–198. https://doi.org/10.1016/0022-3956(75)90026-6
Sheikh JI, Yesavage JA (1986) Geriatric Depression Scale (GDS): recent evidence and development of a shorter version. Clin Gerontol 5:165–173. https://doi.org/10.1300/J018v05n01_09
Mitchell AJ, Bird V, Rizzo M, Meader N (2010) Diagnostic validity and added value of the geriatric depression scale for depression in primary care: a meta-analysis of GDS30 and GDS15. J Affect Disord 125:10–17. https://doi.org/10.1016/j.jad.2009.08.019
Kim S, Park JL, Hwang HS, Kim YP (2014) Correlation between frailty and cognitive function in non-demented community dwelling older Koreans. Korean J Fam Med 35:309–320. https://doi.org/10.4082/kjfm.2014.35.6.309
Montorio I, Izal M (1996) The Geriatric Depression Scale: a review of its development and utility. Int Psychogeriatr 8:103–112. https://doi.org/10.1017/s1041610296002505
Soysal P, Veronese N, Thompson T et al (2017) Relationship between depression and frailty in older adults: a systematic review and meta-analysis. Ageing Res Rev 36:78–87. https://doi.org/10.1016/j.arr.2017.03.005
Panagiotakis SH, Simos P, Basta M et al (2021) Interactions of Mediterranean diet, obesity, polypharmacy, depression and systemic inflammation with frailty status. Maedica 17:20–27. https://doi.org/10.26574/maedica.2022.17.1.20
Sugishita K, Sugishita M, Hemmi I, Asada T, Tanigawa T (2017) A validity and reliability study of the Japanese version of the Geriatric Depression Scale 15 (GDS-15-J). Clin Gerontol 40:233–240. https://doi.org/10.1080/07317115.2016.1199452
Oliveira JS, Pinheiro MB, Fairhall N, Walsh S, Franks TC, Kwok W, Bauman A, Sherrington C (2020) Evidence on physical activity and the prevention of frailty and sarcopenia among older people: a systematic review to inform the World Health Organization physical activity guidelines. J Phys Act Health 17:1247–1258. https://doi.org/10.1123/jpah.2020-0323
Richards SJG, Senadeera SC, Frizelle FA (2020) Sarcopenia, as assessed by psoas cross-sectional area, is predictive of adverse postoperative outcomes in patients undergoing colorectal cancer surgery. Dis Colon Rectum 63:807–815. https://doi.org/10.1097/dcr.0000000000001633
Houghton JSM, Nickinson ATO, Morton AJ, Nduwayo S, Pepper CJ, Rayt HS, Gray LJ, Conroy SP, Haunton VJ, Sayers RD (2020) Frailty factors and outcomes in vascular surgery patients: a systematic review and meta-analysis. Ann Surg 272:266–276. https://doi.org/10.1097/sla.0000000000003642
Takahashi S, Shimizu S, Nagai S, Watanabe H, Nishitani Y, Kurisu Y (2019) Characteristics of sarcopenia after distal gastrectomy in elderly patients. PLoS One 14:e0222412. https://doi.org/10.1371/journal.pone.0222412
Gustafsson UO, Scott MJ, Hubner M et al (2019) Guidelines for Perioperative Care in Elective Colorectal Surgery: Enhanced Recovery After Surgery (ERAS) Society Recommendations: 2018. World J Surg 43:659–695. https://doi.org/10.1007/s00268-018-4844
Ripollés-M J, Ramírez-R JM, Casans-F R et al (2019) Association between use of enhanced recovery after surgery protocol and postoperative complications in colorectal surgery: the Postoperative Outcomes Within Enhanced Recovery After Surgery Protocol (POWER) Study. JAMA Surg 154(8):725–736. https://doi.org/10.1001/jamasurg.2019.0995
Poole L, Leigh E, Kidd T, Ronaldson A, Jahangiri M, Steptoe A (2014) The combined association of depression and socioeconomic status with length of post-operative hospital stay following coronary artery bypass graft surgery: data from a prospective cohort study. J Psychosom Res 76:34–40. https://doi.org/10.1016/j.jpsychores.2013.10.019
Hassani D, Koelper N, Borodyanskaya Y, Arya NG, Rao H, Andy U (2022) Cognitive function following surgery for pelvic organ prolapse. Neurourol Urodyn 41:1853–1861. https://doi.org/10.1002/nau.25035
Yoshimoto A, Inoue T, Morizumi S, Nishi S, Shimizu T, Kurahashi K, Suematsu Y (2019) Analysis of the perioperative change in cognitive function of patients with risk factors for cognitive impairment in cardiovascular surgery. Gen Thorac Cardiovasc Surg 67:214–218. https://doi.org/10.1007/s11748-018-0999-6
van Wijk L, van Duinhoven S, Liem MSL, Bouman DE, Viddeleer AR, Klaase JM (2021) Risk factors for surgery-related muscle quantity and muscle quality loss and their impact on outcome. Eur J Med Res 26:36–45. https://doi.org/10.1186/s40001-021-00507-9
Kubota T, Shoda K, Konishi H, Okamoto K, Otsuji E (2020) Nutrition update in gastric cancer surgery. Ann Gastroenterol Surg 4:360–368. https://doi.org/10.1002/ags3.12351
Xu J, Wan CS, Ktoris K, Reijnierse EM, Maier AB (2022) Sarcopenia is associated with mortality in adults: a systematic review and meta-analysis. Gerontology 68:361–376. https://doi.org/10.1159/000517099
Espinoza SE, Jung I, Hazuda H (2012) Frailty transitions in the San Antonio Longitudinal Study of Aging. J Am Geriatr Soc 60:652–660. https://doi.org/10.1111/j.1532-5415.2011.03882
Koh FH, Chua JM, Tan JL et al (2021) Paradigm shift in gastrointestinal surgery - combating sarcopenia with prehabilitation: multimodal review of clinical and scientific data. World J Gastrointest Surg 13:734–755. https://doi.org/10.4240/wjgs.v13.i8.734
Zhao H, Cheng R, Song G, Teng J, Shen S, Fu X, Yan Y, Liu C (2022) The effect of resistance training on the rehabilitation of elderly patients with sarcopenia: a meta-analysis. Int J Environ Res Public Health 19:15491. https://doi.org/10.3390/ijerph192315491
Cruz-Jentoft AJ, Landi F, Schneider SM et al (2014) Prevalence of and interventions for sarcopenia in ageing adults: a systematic review. Report of the International Sarcopenia Initiative (EWGSOP and IWGS). Age Ageing 43:748–759. https://doi.org/10.1093/ageing/afu115
Sadjapong U, Yodkeeree S, Sungkarat S, Siviroj P (2020) Multicomponent exercise program reduces frailty and inflammatory biomarkers and improves physical performance in community-dwelling older adults: A randomized controlled trial. Int J Environ Res Public Health 17:3760–3774. https://doi.org/10.3390/ijerph17113760
Ni Lochlainn M, Cox NJ, Wilson T (2021) Nutrition and frailty: opportunities for prevention and treatment. Nutrients 13:2349. https://doi.org/10.3390/nu13072349
Niedzwiedz CL, Knifton L, Robb KA, Katikireddi SV, Smith DJ (2019) Depression and anxiety among people living with and beyond cancer: a growing clinical and research priority. BMC Cancer 19:943–951. https://doi.org/10.1186/s12885-019-6181-4
Lafont C, Chah Wakilian A, Lemogne C et al (2021) Diagnostic performance of the 4-item Geriatric Depression Scale for depression screening in older patients with cancer:the ELCAPA cohort study. Oncologist 26:e983–e991. https://doi.org/10.1002/onco.13746
Watts S, Prescott P, Mason J, McLeod N, Lewith G (2015) Depression and anxiety in ovarian cancer: a systematic review and meta-analysis of prevalence rates. BMJ Open 5:e007618. https://doi.org/10.1136/bmjopen-2015-007618
Ehlenbach WJ, Hough CL, Crane PK, Haneuse SJPA, Carson SS, Curtis JR, Larson EB (2010) Association between acute care and critical illness hospitalization and cognitive function in older adults. JAMA 303:763–770. https://doi.org/10.1001/jama.2010.167
Avila-Funes JA, Amieva H, Barberger-Gateau P et al (2009) Cognitive impairment improves the predictive validity of the phenotype of frailty for adverse health outcomes: the three-city study. J Am Geriatr Soc 57:453–461. https://doi.org/10.1111/j.1532-5415.2008.02136.x
Acknowledgements
The authors express gratitude to Hiroko Shiraki, Hisako Akama, and Minori Oogiyama for their support in the questionnaire survey and data collection.
Author information
Authors and Affiliations
Contributions
Study conception and design: Tomokazu Kishiki, Hiroshi Hasegawa, Hideaki Yoshino, Seiichi Taniai, Kumi Moriyama, Tomoko Yorozu, Haruhiko Kondo, Yoshihiro Sakamoto, Nobutsugu Abe, and Eiji Sunami; Acquisition of data: Tomokazu Kishiki, Hiroshi Hasegawa, Hideaki Yoshino, Nobuyoshi Aso, Aiko Iioka, Takashi Wakamatsu, Kazuna Honda, Isao Kataoka, Sangchul Kim, Shun Ishii, Satoshi Isobe, Toshiya Shirota, Mayumi Ide, Seiichi Taniai, Kumi Moriyama, Tomoko Yorozu, Haruhiko Kondo, Yoshihiro Sakamoto, Nobutsugu Abe, and Eiji Sunami; Analysis and interpretation of data: Tomokazu Kishiki, Hiroshi Hasegawa, Hideaki Yoshino, Seiichi Taniai, Kumi Moriyama, Tomoko Yorozu, Haruhiko Kondo, Yoshihiro Sakamoto, Nobutsugu Abe, and Eiji Sunami; Drafting of manuscript: Tomokazu Kishiki, Hiroshi Hasegawa, Hideaki Yoshino, Nobuyoshi Aso, Aiko Iioka, Takashi Wakamatsu, Kazuna Honda, Isao Kataoka, Sangchul Kim, Shun Ishii, Satoshi Isobe, Toshiya Shirota, Mayumi Ide, Seiichi Taniai, Kumi Moriyama, Tomoko Yorozu, Haruhiko Kondo, Yoshihiro Sakamoto, Nobutsugu Abe, and Eiji Sunami; Critical revision of manuscript: Tomokazu Kishiki, Hiroshi Hasegawa, Hideaki Yoshino, Nobuyoshi Aso, Aiko Iioka, Takashi Wakamatsu, Kazuna Honda, Isao Kataoka, Sangchul Kim, Shun Ishii, Satoshi Isobe, Toshiya Shirota, Mayumi Ide, Seiichi Taniai, Kumi Moriyama, Tomoko Yorozu, Haruhiko Kondo, Yoshihiro Sakamoto, Nobutsugu Abe, and Eiji Sunami.
Corresponding author
Ethics declarations
Ethics approval
The study was conducted in accordance with the ethical guidelines of the 1975 Declaration of Helsinki. This study protocol was approved by the Research Ethics Committee of Kyorin University Hospital (approval date and number: November 14, 2017, and 1013).
Consent to participate
Written informed consent was obtained from all the participants.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix
Appendix
ECOG Performance Status | |
|---|---|
Grade | ECOG |
0 | Fully active, able to carry on all pre-disease performance without restriction |
1 | Restricted in physically strenuous activity but ambulatory and able to carry out work of a light or sedentary nature, e.g., light housework, office work |
2 | Ambulatory and capable of all self-care actions but unable to carry out any work-related activities. Up and about more than 50% of waking hours |
3 | Capable of only limited self-care, confined to bed or chair more than 50% of waking hours |
4 | Completely disabled. Cannot carry on any self-care actions. Totally confined to bed or chair |
5 | Dead |
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kishiki, T., Hasegawa, H., Yoshino, H. et al. Physical frailty recovery is slower than mental frailty recovery after non-cardiac surgery in older adult patients. Langenbecks Arch Surg 408, 395 (2023). https://doi.org/10.1007/s00423-023-03123-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00423-023-03123-9