Abstract
Introduction
Two phosphodiesterase-type 5 (PDE-5) inhibitors, sildenafil and tadalafil, are approved for treatment of pulmonary arterial hypertension (PAH). It has not yet been observed if transition from sildenafil to tadalafil is beneficial in patients suffering from adverse reactions. Aim of this study was to analyze safety and long-term effects in PAH patients whose treatment was transitioned from sildenafil to tadalafil due to intolerable side-effects.
Methods
A retrospective analysis of PAH-patients who were stable on sildenafil for >3 months and transitioned to tadalafil due to adverse events was performed. Data collected included demographics, PAH-etiology, WHO-functional class, 6 min walking distance (6MWD), echocardiography, lung function tests, and NTproBNP pre-transition and 3, 6, and 12 months post-transition.
Results
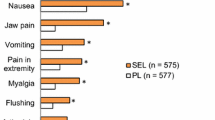
Included were 13 PAH patients (8 females mean age 64 ± 10 years) who had been on sildenafil for a mean of 12 ± 8.4 months. In six patients (46.1 %) a switch to tadalafil was feasible and resulted in tolerable side effects and a stable clinical course with improvement of symptoms, 6MWD, stable echocardiographic findings, and NTproBNP-levels during a follow-up of 11 ± 3 months. In 5 out of 13 patients (38.5 %) adverse events occurred under tadalafil as well and therapy with PDE-5 inhibitors was discontinued. In two patients (15.4 %) sildenafil-treatment could be successfully restarted after an intermittent switch to tadalafil.
Conclusions
The observations of this study indicate that a transition of sildenafil to tadalafil in case of intolerable side effects is a reasonable therapy option in about 50 % of the patients. These results should be verified by a larger prospective study.

Similar content being viewed by others
Abbreviations
- 6MWD:
-
6 min walking distance
- NO:
-
Nitric oxide
- NTproBNP:
-
N-terminal prohormone of brain natriuretic peptide
- PAH:
-
Pulmonary arterial hypertension
- PASP:
-
Systolic pulmonary arterial pressures
- PDE-5:
-
Phosphodiesterase-type 5
- PH:
-
Pulmonary hypertension
- q.d.:
-
Quaque die
- TAPSE:
-
Tricuspid annular plane systolic excursion
- t.i.d.:
-
Ter in die
- WHO:
-
World Health Organization
References
Galiè N, Corris PA, Frost A, Girgis RE, Granton J, **g ZC, Klepetko W, McGoon MD, McLaughlin VV, Preston IR, Rubin LJ, Sandoval J, Seeger W, Keogh A (2013) Updated treatment algorithm of pulmonary arterial hypertension. J Am Coll Cardiol 62(25 Suppl):D60–D72
Galiè N, Hoeper MM, Humbert M, Torbicki A, Vachiery JL, Barbera JA, Beghetti M, Corris P, Gaine S, Gibbs JS, Gomez-Sanchez MA, Jondeau G, Klepetko W, Opitz C, Peacock A, Rubin L, Zellweger M, Simonneau G (2009) Guidelines for the diagnosis and treatment of pulmonary hypertension the task force for the diagnosis and treatment of pulmonary hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS), endorsed by the International Society of Heart and Lung Transplantation (ISHLT). Eur Heart J 30:2493–2537
Badesch DB, Abman SH, Simonneau G, Rubin LJ, McLaughlin VV (2007) Medical therapy for pulmonary arterial hypertension: updated ACCP evidence-based clinical practice guidelines. Chest 131:1917–1928
Galiè N, Ghofrani HA, Torbicki A, Barst RJ, Rubin LJ, Badesch D, Fleming T, Parpia T, Burgess G, Branzi A, Grimminger F, Kurzyna M, Simonneau G (2005) Sildenafil use in pulmonary arterial hypertension (SUPER) Study Group: sildenafil citrate therapy for pulmonary arterial hypertension. N Engl J Med 353(20):2148–2157
Galiè N, Brundage BH, Ghofrani HA, Oudiz RJ, Simonneau G, Safdar Z, Shapiro S, White RJ, Chan M, Beardsworth A, Frumkin L, Barst RJ (2011) Pulmonary Arterial Hypertension and Response to Tadalafil (PHIRST) Study Group: tadalafil therapy for pulmonary arterial hypertension. Circulation 119(22):2894–2903 Erratum in: Circulation. 2011, 124(10):e279
Oudiz RJ, Brundage BH, Galiè N, Ghofrani HA, Simonneau G, Botros FT, Chan M, Beardsworth A, Barst RJ (2012) Tadalafil for the treatment of pulmonary arterial hypertension: a double-blind 52-week uncontrolled extension study. J Am Coll Cardiol 60(8):768–774
Rubin LJ, Badesch DB, Fleming TR, Galiè N, Simonneau G, Ghofrani HA, Oakes M, Layton G, Serdarevic-Pehar M, McLaughlin VV, Barst RJ; SUPER-2 Study Group (2011) Long-term treatment with sildenafil citrate in pulmonary arterial hypertension: the SUPER-2 study. Chest 140(5):1274–1283
Chin KM, Rubin LJ (2008) Pulmonary arterial hypertension. J Am Coll Cardiol 51(16):1527–1538
Archer SL, Michelakis ED (2009) Phosphodiesterase type 5 inhibitors for pulmonary arterial hypertension. N Engl J Med 361(19):1864–1871
Tay EL, Geok-Mui MK, Poh-Hoon MC, Yip J (2008) Sustained benefit of tadalafil in patients with pulmonary arterial hypertension with prior response to sildenafil: a case series of 12 patients. Int J Cardiol 125(3):416–417
Shlobin OA, Brown AW, Weir N, Ahmad S, Lemma M, Nathan SD (2012) Transition of PH patients from sildenafil to tadalafil: feasibility and practical considerations. Lung 190(5):573–578
Shapiro S, Traiger G, Hill W, Zhang L, Doran AK (2013) Safety, tolerability, and efficacy of overnight switching from sildenafil to tadalafil in patients with pulmonary arterial hypertension. Cardiovasc Ther 31(5):274–279
Gupta M, Kovar A, Meibohm B (2005) The clinical pharmacokinetics of phosphodiesterase-5 inhibitors for erectile dysfunction. J Clin Pharmacol 45:987–1003
Wilkins MR, Wharton J, Grimminger F, Ghofrani HA (2008) Phosphodiesterase inhibitors for the treatment of pulmonary hypertension. Eur Respir J 32(1):198–209
Acknowledgments
This study was funded by Eli Lilly and Company.
Conflict of interest
ML: receives consultancy fees from Actelion. DH: nothing to disclose. NE: receives speaker fees from Bayer HealthCare, Pfizer, Actelion, Lilly. AM: nothing to disclose. FP: nothing to disclose. EG: receives consultancy/speaker fees from Bayer HealthCare, Pfizer, Actelion, Lilly, Encysive, Alexion, United Therapeutics, GlaxoSmithKline, Miltenyi, Sinoxa Pharma and Novartis. BE: nothing to disclose.
Author information
Authors and Affiliations
Corresponding author
Additional information
Mona Lichtblau and Dominik Harzheim made an equal contribution to the study.
Rights and permissions
About this article
Cite this article
Lichtblau, M., Harzheim, D., Ehlken, N. et al. Safety and Long-Term Efficacy of Transition from Sildenafil to Tadalafil due to Side Effects in Patients with Pulmonary Arterial Hypertension. Lung 193, 105–112 (2015). https://doi.org/10.1007/s00408-014-9657-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00408-014-9657-7