Abstract
Objectives
To use pericardial adipose tissue (PAT) radiomics phenoty** to differentiate existing and predict future heart failure (HF) cases in the UK Biobank.
Methods
PAT segmentations were derived from cardiovascular magnetic resonance (CMR) studies using an automated quality-controlled model to define the region-of-interest for radiomics analysis. Prevalent (present at time of imaging) and incident (first occurrence after imaging) HF were ascertained using health record linkage. We created balanced cohorts of non-HF individuals for comparison. PyRadiomics was utilised to extract 104 radiomics features, of which 28 were chosen after excluding highly correlated ones (0.8). These features, plus sex and age, served as predictors in binary classification models trained separately to detect (1) prevalent and (2) incident HF. We tested seven modeling methods using tenfold nested cross-validation and examined feature importance with explainability methods.
Results
We studied 1204 participants in total, 297 participants with prevalent (60 ± 7 years, 21% female) and 305 with incident (61 ± 6 years, 32% female) HF, and an equal number of non-HF comparators. We achieved good discriminative performance for both prevalent (voting classifier; AUC: 0.76; F1 score: 0.70) and incident (light gradient boosting machine: AUC: 0.74; F1 score: 0.68) HF. Our radiomics models showed marginally better performance compared to PAT area alone.
Increased PAT size (maximum 2D diameter in a given column or slice) and texture heterogeneity (sum entropy) were important features for prevalent and incident HF classification models.
Conclusions
The amount and character of PAT discriminate individuals with prevalent HF and predict incidence of future HF.
Clinical relevance statement
This study presents an innovative application of pericardial adipose tissue (PAT) radiomics phenoty** as a predictive tool for heart failure (HF), a major public health concern. By leveraging advanced machine learning methods, the research uncovers that the quantity and characteristics of PAT can be used to identify existing cases of HF and predict future occurrences. The enhanced performance of these radiomics models over PAT area alone supports the potential for better personalised care through earlier detection and prevention of HF.
Key Points
•PAT radiomics applied to CMR was used for the first time to derive binary machine learning classifiers to develop models for discrimination of prevalence and prediction of incident heart failure.
•Models using PAT area provided acceptable discrimination between cases of prevalent or incident heart failure and comparator groups.
•An increased PAT volume (increased diameter using shape features) and greater texture heterogeneity captured by radiomics texture features (increased sum entropy) can be used as an additional classifier marker for heart failure.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Pericardial adipose tissue (PAT) is the visceral adipose tissue compartment surrounding the heart and coronary vasculature. An increasing body of evidence highlights associations between greater amounts of PAT and poorer cardiovascular outcomes [1,2,3,4,5]. Furthermore, higher PAT has been linked to adverse cardiovascular phenotypes, independent of multiple other measures of adiposity [6]. These associations are highly suggestive of a distinct mechanistic role of PAT in driving adverse cardiac remodelling, which are precursors of heart failure [6].
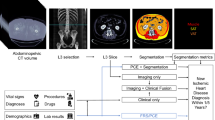
The mechanisms through which PAT influences myocardial structure and function are likely multifactorial, involving paracrine, vasocrine, and inflammatory pathways. PAT is known to secrete inflammatory factors and lipid metabolites [7], and this metabolic and secretory activity has been highlighted as an important factor driving adverse cardiovascular outcomes. At a cellular level, the secretome of PAT has been shown to adversely impact cardiomyocyte contractility [8], metabolism [9], and disrupt adhesion molecule expression in cardiac endothelial cells [10]. In the setting of ischemic heart disease, patterns of coronary atherosclerosis have been shown to closely follow superficial PAT distribution [ We provide a schematic illustration of the study pipeline in Fig. 1. Schematic illustration of the study pipeline. A The segmentation of pericardial adipose tissue (PAT); color overlay represents the segmentation results as derived by Bard et al. B The radiomics feature extraction: PAT segmentation output was used to extract radiomics features from cardiovascular magnetic resonance imaging data. C The final cohort, which was assembled from prevalent and incident heart failure cases and randomly selected control individuals from the UK Biobank dataset The UK Biobank is a cohort study incorporating more than 500,000 individuals from across the UK, aged 40–69 years old at recruitment between 2006 and 2010. Baseline assessment included socio-demographics, lifestyle, environmental factors, medical history, and a range of physical measures. Extensive electronic health record (EHR) linkages permit prospective tracking of health outcomes for all participants. The UK Biobank Imaging Study, which includes CMR, aims to scan a randomly selected 20% subset of the original participants. HF status was ascertained using diseases codes from UK Biobank assessments and linked EHRs (Supplementary Table 1). Prevalent HF was defined as HF present at the time of imaging. Incident HF was considered as first occurrence of HF after imaging. The censor date was 30 September 2021 for incident HF outcomes giving an average follow-up of 3.7 ± 1.5 years from imaging. Participants with CMR available and no record of HF (prevalent or incident) were eligible for inclusion in the control group (n = 42,327). There was substantial imbalance between cases and eligible controls. Such imbalance results in poor model performance as the model prediction will be dominated by the majority class [16]. Given the extreme imbalance in our dataset, with a large number of controls compared to cases, and considering computational constraints we applied random undersampling to reduce the frequency of the controls relative to the cases. This approach randomly removes subjects from the majority classes to reach a final set of subjects in the majority class that are similar to the minority class. The final sample contained an equal number of randomly selected non-HF controls for both the prevalent and incident HF groups. We accessed self-reported fields for participants’ educational level and smoking status. Material deprivation is reported as the Townsend index. Physical activity was measured via self-reported responses to the International Physical Activity Questionnaire. Diabetes, hypertension, and high cholesterol status were ascertained using information from self-report questions, physical measurements, and EHR data (Supplementary Table 2). CMR scans were performed according to a pre-defined acquisition protocol using 1.5-Tesla scanners (MAGNETOM Aera, Syngo Platform VD13A, Siemens Healthcare) [17]. Cardiac function was assessed using standard long- and short-axis balanced steady-state free precession cine sequences. PAT segmentation was performed using an automated quality-controlled pipeline developed and validated in the UK Biobank and in an external cohort, as described by Bard et al [15]. In brief, PAT was measured from standard four-chamber cine images (single 2D slice) at phase 1 of the image cycle, which approximates end-diastole (ED). The contour was drawn to select areas of high signal intensity bordering the epicardial surface of the left and right ventricular myocardia. The ground truth manual segmentation was based on a sample of 500 randomly selected UK Biobank imaging sub-study participants using CVI42 post-processing software (Version 5.11, Circle Cardiovascular Imaging Inc.). Using the manual segmentation, a MultiResUNet neural network with Bayesian modification was trained for automated PAT segmentation with inbuilt quality control. Overall, the performance of the algorithm in test set relative to manual segmentations was good and very similar to the agreement between human observers (mean Dice score = 0.8) [15]. Automated PAT analysis was performed for all participants with adequate CMR imaging available (n = 42,929). Radiomics is an analysis technique permitting the computation of multiple descriptors of shape and texture [18]. The relevant information present in the image is extracted using three classes of features, namely (i) shape, (ii) first-order, and (iii) texture-based features. First-order features are histogram-based and relate to the distribution of the grey-level values in the tissue. Shape features capture the geometrical properties of the region of interest (ROI), including volume, diameter, minor/major axis, or sphericity. Texture features are derived from images using five matrices that encode the global texture information. They aim to describe patterns using mathematical formulae based on the spatial arrangement of pixels. Lately, CMR radiomics features have been utilised to appreciate the heart’s complexity derived from the left and right ventricles, revealing patterns invisible to the naked eye [14]. There are as yet no existing reports of clinical models based on PAT radiomics features, likely due to the absence of appropriate datasets. We used the PAT segmentation defined on the long-axis four-chamber images in the ED phase using the automated pipeline described above to derive our regions of interest (ROI) for radiomics analysis. We converted the contour points into binary masks, using a tool developed in-house, which we have made publicly available [19]. This software transformed delineated contour points for each ROI into a filled polygon in the coordinate space to form the binary mask. The harmonisation of the images was conducted using a histogram-matching technique applied to a reference image. The grey value discretisation was performed using a bin width of 25 to pull the intensity-based and texture radiomics features. The reference image for histogram matching was randomly selected, with careful consideration to ensure the chosen image did not contain artifacts. Histogram matching has been utilised effectively in prior radiomics-based models to standardise the intensity scale, thereby enhancing the model’s generalisation and classification capabilities [20,21,22]. In this study, imaging data was acquired using the same protocol (i.e., identical scanners and parameters) [17]. Therefore, we selected a single participant at random as the template for histogram harmonisation. This approach has been previously demonstrated to yield successful results in similar studies [20, 22]. The PyRadiomics platform (version 2.2.0) was adopted to extract 104 shape (n = 12), first-order (n = 17), and texture (n = 75) features from all PAT ROIs. To mitigate the risk of multicollinearity and increase the interpretability of our models, after feature extraction we performed a correlation analysis among radiomics features. Pairs of features exhibiting a correlation coefficient with an absolute value of 0.8 or above were identified. From each pair, we removed one feature to maintain the distinctiveness of the predictors in our model. Following this correlation-based feature selection process, we retained 28 features from the original 104. We also included age and sex in our model as they are known to significantly influence cardiac health. For comparative purposes, we also developed another model which incorporated overall PAT area, age, and sex as predictors. All the methods were implemented using Python version 3.9 and Scikit-learn [23] version 1.0.2. PAT radiomics features were used as predictors to classify prevalent HF and predict incident HF from non-HF controls. The features were normalised to zero mean and unit variance. We used seven binary classifiers followed by a voting classifier. We included the following classifiers to consider a wide variety of potential approaches: logistic regression (LR) [24], support vector classifier (SVC) [25], random forest (RF) [26], K-nearest neighbours (KNN) [27], decision tree (DT) [28], light gradient boosting machine (LGBM) [29], and multi-layer perceptron (MLP) [30]. To obtain each classifier’s optimal parameters, we used hyperparameter tuning and tenfold nested cross-validation, which consists of two loops [39,40,41] and different cardiomyopathies [42,43,44]. Pericardial fat radiomics represents an additional layer of information we can derive from standard of care CMR scans. Critically, our results demonstrate that radiomics can be used to discriminate HF cases from controls, signifying a potential novel avenue for better diagnostic and prognostic assessment. Our study provides initial insights into PAT radiomics for predicting HF though it has some important limitations. The models are preliminary and need further independent external validation. We used random undersampling due to extreme dataset imbalance, potentially leading to bias and overoptimistic performance estimates. While our models used a range of radiomics features and demographic variables, other relevant factors were not included, warranting comprehensive patient information in future research to enhance model performance. Machine learning classifiers built upon radiomics features depicting the amount (larger PAT diameters) and texture character (greater tissue heterogeneity) of pericardial fat can be used to discriminate individuals with prevalent heart failure and predict incidence of future heart failure.Material and methods
Setting and study population
Ascertainment of heart failure status
Definition of the comparator group
Characterising the study sample
Image acquisition
Extraction of pericardial fat segmentations
Background to radiomics
PAT radiomics feature extraction
Feature selection
Predictive models
Limitations
Conclusions
Abbreviations
- CAD:
-
Coronary artery disease
- CMR:
-
Cardiovascular magnetic resonance
- DT:
-
Decision tree
- HF:
-
Heart failure
- KNN:
-
K-nearest neighbours
- LGBM:
-
Light gradient boosting machine
- LIME:
-
Local interpretable model-agnostic explanations
- LR:
-
Logistic regression
- MLP:
-
Multi-layer perceptron
- PAT:
-
Pericardial adipose tissue
- RF:
-
Random forest
- ROC:
-
Receiver operating characteristic curves
- SHAP:
-
SHaply Additive exPlanations
- SI:
-
Signal intensity
- SVC:
-
Support vector classifier
References
Wong CX, Ganesan AN, Selvanayagam JB (2017) Epicardial fat and atrial fibrillation: current evidence, potential mechanisms, clinical implications, and future directions. Eur Heart J 38:1294–1302. https://doi.org/10.1093/eurheartj/ehw045
Shah RV, Anderson A, Ding J et al (2017) Pericardial, but not hepatic, fat by CT is associated with CV outcomes and structure: the Multi-Ethnic Study of Atherosclerosis. JACC Cardiovasc Imaging 10:1016–1027. https://doi.org/10.1016/j.jcmg.2016.10.024
Greif M, Becker A, Von Ziegler F et al (2009) Pericardial adipose tissue determined by dual source CT is a risk factor for coronary atherosclerosis. Arterioscler Thromb Vasc Biol 29:781–786. https://doi.org/10.1161/ATVBAHA.108.180653
Batal O, Schoenhagen P, Shao M et al (2010) Left atrial epicardial adiposity and atrial fibrillation. Circ Arrhythmia Electrophysiol 3:230–236. https://doi.org/10.1161/CIRCEP.110.957241
Kenchaiah S, Ding J, Carr JJ et al (2021) Pericardial fat and the risk of heart failure. J Am Coll Cardiol 77:2638–2652. https://doi.org/10.1016/j.jacc.2021.04.003
Ardissino M, McCracken C, Bard A et al (2022) Pericardial adiposity is independently linked to adverse cardiovascular phenotypes: a CMR study of 42 598 UK Biobank participants. Eur Hear J Cardiovasc Imaging 23(11):1471–1481. https://doi.org/10.1093/ehjci/jeac101
Iacobellis G (2015) Local and systemic effects of the multifaceted epicardial adipose tissue depot. Nat Rev Endocrinol 11:363–371. https://doi.org/10.1038/nrendo.2015.58
Greulich S, Maxhera B, Vandenplas G et al (2012) Secretory products from epicardial adipose tissue of patients with type 2 diabetes mellitus induce cardiomyocyte dysfunction. Circulation 126:2324–2334. https://doi.org/10.1161/CIRCULATIONAHA.111.039586
Blumensatt M, Fahlbusch P, Hilgers R et al (2017) Secretory products from epicardial adipose tissue from patients with type 2 diabetes impair mitochondrial β-oxidation in cardiomyocytes via activation of the cardiac renin–angiotensin system and induction of miR-208a. Basic Res Cardiol 112:1–13. https://doi.org/10.1007/s00395-016-0591-0
Chechi K, Voisine P, Mathieu P et al (2017) Functional characterization of the Ucp1-associated oxidative phenotype of human epicardial adipose tissue. Sci Rep 7:1–15. https://doi.org/10.1038/s41598-017-15501-7
**e Z, Zhu J, Li W et al (2021) Relationship of epicardial fat volume with coronary plaque characteristics, coronary artery calcification score, coronary stenosis, and CT-FFR for lesion-specific ischemia in patients with known or suspected coronary artery disease. Int J Cardiol 332:8–14. https://doi.org/10.1016/j.ijcard.2021.03.052
Mahmoud I, Dykun I, Kärner L et al (2021) Epicardial adipose tissue differentiates in patients with and without coronary microvascular dysfunction. Int J Obes 45:2058–2063. https://doi.org/10.1038/s41366-021-00875-6
Oikonomou EK, Williams MC, Kotanidis CP et al (2019) A novel machine learning-derived radiotranscriptomic signature of perivascular fat improves cardiac risk prediction using coronary CTangiography. Eur Heart J 40:3529–3543. https://doi.org/10.1093/eurheartj/ehz592
Raisi-Estabragh Z, Izquierdo C, Campello VM et al (2020) Cardiac magnetic resonance radiomics: basic principles and clinical perspectives. Eur Heart J Cardiovasc Imaging 21:349–356. https://doi.org/10.1093/ehjci/jeaa028
Bard A, Raisi-Estabragh Z, Ardissino M et al (2021) Automated quality-controlled cardiovascular magnetic resonance pericardial fat quantification using a convolutional neural network in the UK Biobank. Front Cardiovasc Med 8:1–11. https://doi.org/10.3389/fcvm.2021.677574
Lee W, Seo K (2022) Downsampling for binary classification with a highly imbalanced dataset using active learning. Big Data Res 28:100314. https://doi.org/10.1016/j.bdr.2022.100314
Petersen SE, Matthews PM, Francis JM et al (2016) UK Biobank's cardiovascular magnetic resonance protocol. J Cardiovasc Magn Reson 18:8. https://doi.org/10.1186/s12968-016-0227-4
Lambin P, Leijenaar RTH, Deist TM et al (2017) Radiomics: the bridge between medical imaging and personalized medicine. Nat Rev Clin Oncol 14:749–762. https://doi.org/10.1038/nrclinonc.2017.141
EuCanSHare/DICOM2NITFI: Tool for medical image conversion from DICOM to NIFTI format., GitHub. Available at: https://github.com/euCanSHare/dicom2nitfi. Accessed 14 Jul 2022
Pujadas ER, Raisi-Estabragh Z, Szabo L et al (2023) Prediction of incident cardiovascular events using machine learning and CMR radiomics. Eur Radiol 33(5):3488–3500. https://doi.org/10.1007/s00330-022-09323-z
Campello VM, Martín-Isla C, Izquierdo C et al (2022) Minimising multi-centre radiomics variability through image normalisation: a pilot study. Sci Rep 12:1–10. https://doi.org/10.1038/s41598-022-16375-0
Pujadas ER, Raisi-Estabragh Z, Szabo L et al (2022) Atrial fibrillation prediction by combining ECG markers and CMR radiomics. Sci Rep 12:1–15. https://doi.org/10.1038/s41598-022-21663-w
Pedregosa F, Varoquaux G, Gramfort A et al (2011) Scikit-learn: machine learning in Python. J Mach Learn Res 12:2825–2830
Cox DR (1958) The regression analysis of binary sequences. J R Stat Soc Ser B 20:215–232. https://doi.org/10.1111/j.2517-6161.1958.tb00292.x
Avendaño-Valencia LD, Fassois SD (2015) Natural vibration response based damage detection for an operating wind turbine via random coefficient linear parameter varying AR modelling. J Phys Conf Ser 628:273–297. https://doi.org/10.1088/1742-6596/628/1/012073
Ho TK (1995) Random decision forests. In: Proceedings of 3rd International Conference on Document Analysis and Recognition, vol 1. Montreal, QC, Canada, pp. 278–282. https://doi.org/10.1109/ICDAR.1995.598994
Altman NS (1992) An introduction to kernel and nearest-neighbor nonparametric regression. Am Stat 46(3):175-185. https://doi.org/10.1080/00031305.1992.10475879
Loh WY (2011) Classification and regression trees. Wiley Interdiscip Rev Data Min Knowl Discov 1:14–23. https://doi.org/10.1002/widm.8
Ke G, Meng Q, Finley T et al (2017) Lightgbm: a highly efficient gradient boosting decision tree. Adv Neural Inf Process Syst 30:3146–3154
Hinton GE (1989) Connectionist learning procedures, vol 40. Artificial Intelligence, Machine Learning, pp. 555–610. https://doi.org/10.1016/B978-0-08-051055-2.50029-8
Raschka S (2018) Model evaluation, model selection, and algorithm selection in machine learning. https://doi.org/10.48550/ar**v.1811.12808
Varma S, Simon R (2006) Bias in error estimation when using cross-validation for model selection. BMC Bioinformatics 7:1–8. https://doi.org/10.1186/1471-2105-7-91
Lundberg SM, Lee SI (2017) A unified approach to interpreting model predictions. Advances in neural information processing systems 30. https://doi.org/10.48550/ar**v.1705.07874
Ribeiro MT, Singh S, Guestrin C (2016) “Why should i trust you?” explaining the predictions of any classifier. In: Proceedings of the 22nd ACM SIGKDD international conference on knowledge discovery and data mining, pp. 1135–1144. https://doi.org/10.48550/ar**v.1602.04938
Ong KL, Ding J, McClelland RL et al (2015) Relationship of pericardial fat with biomarkers of inflammation and hemostasis, and cardiovascular disease: the Multi-Ethnic Study of Atherosclerosis. Atherosclerosis 239:386–392. https://doi.org/10.1016/j.atherosclerosis.2015.01.033
Mazurek T, Zhang L, Zalewski A et al (2003) Human epicardial adipose tissue is a source of inflammatory mediators. Circulation 108:2460–2466. https://doi.org/10.1161/01.CIR.0000099542.57313.C5
Zimmermann GS, Ruether T, von Ziegler F et al (2021) Increased pericardial adipose tissue in smokers. J Clin Med 10:3382. https://doi.org/10.3390/JCM10153382
Chang KF, Lin G, Huang PC et al (2020) Left ventricular function and myocardial triglyceride content on 3T cardiac MR predict major cardiovascular adverse events and readmission in patients hospitalized with acute heart failure. J Clin Med 9:1–15. https://doi.org/10.3390/jcm9010169
Ma Q, Ma Y, Yu T, Sun Z, Hou Y (2021) Radiomics of non-contrast-enhanced T1 map**: diagnostic and predictive performance for myocardial injury in acute ST-segment-elevation myocardial infarction. Korean J Radiol 22:535–546. https://doi.org/10.3348/kjr.2019.0969
Avard E, Shiri I, Hajianfar G et al (2022) Non-contrast cine cardiac magnetic resonance image radiomics features and machine learning algorithms for myocardial infarction detection. Comput Biol Med 141:105145. https://doi.org/10.1016/j.compbiomed.2021.105145
Chen BH, An DA, He J et al (2021) Myocardial extracellular volume fraction radiomics analysis for differentiation of reversible versus irreversible myocardial damage and prediction of left ventricular adverse remodeling after ST-elevation myocardial infarction. Eur Radiol 31:504–514. https://doi.org/10.1007/s00330-020-07117-9
Antonopoulos AS, Boutsikou M, Simantiris S et al (2021) Machine learning of native T1 map** radiomics for classification of hypertrophic cardiomyopathy phenotypes. Sci Rep 11:1–11. https://doi.org/10.1038/s41598-021-02971-z
Cheng S, Fang M, Cui C et al (2018) LGE-CMR-derived texture features reflect poor prognosis in hypertrophic cardiomyopathy patients with systolic dysfunction: preliminary results. Eur Radiol 28:4615–4624. https://doi.org/10.1007/s00330-018-5391-5
Neisius U, El-Rewaidy H, Kucukseymen S et al (2020) Texture signatures of native myocardial T1 as novel imaging markers for identification of hypertrophic cardiomyopathy patients without scar. J Magn Reson Imaging 52:906–919. https://doi.org/10.1002/jmri.27048
Acknowledgements
This study was conducted using the UK Biobank resource under access application 2964. We would like to thank all the UK Biobank participants, staff involved with planning, collection, and analysis, including core lab analysis of the CMR imaging data.
Funding
LS was supported by the ÚNKP-22-4-I New National Excellence Program of the Ministry for Culture and Innovation from the National Research, Development and Innovation Fund and the European Association of Cardiovascular Imaging (EACVI Research Grant App000076437). AS was supported by a British Heart Foundation (BHF) project grant (PG/21/10619). CM and SN are supported by the Oxford National Institute for Health and Care Research (NIHR) Biomedical Research Centre (IS-BRC-1215–20008) and the Oxford BHF Centre of Research Excellence. CA acknowledges support from the Oxford BHF (CH/F/21/90009, TG/19/2/34831 and RG/F/21/110040), the Oxford BHF Centre of Research Excellence (RE/18/3/34214), and Innovate UK. NCH acknowledges support from Medical Research Council (MRC, MC_UU_12011/1) and NIHR Southampton Biomedical Research Centre. This work was supported by Health Data Research UK, an initiative funded by UK Research and Innovation, Department of Health and Social Care (England), and the devolved administrations, and leading medical research charities. SEP acknowledges support from the NIHR Biomedical Research Centre at Barts. SEP, ERP, and LS have received funding from the European Union’s Horizon 2020 research and innovation programme under grant agreement No 825903 (euCanSHare project). ERP was partly funded by the European Union’s Horizon 2020 research and innovation programme under grant agreement no. 965345 (HealthyCloud project). SEP acknowledges support from the “SmartHeart” EPSRC programme grant (www.nihr.ac.uk; EP/P001009/1). ZRE recognises the NIHR Integrated Academic Training programme which supports her Academic Clinical Lectureship post and was also supported by BHF Clinical Research Training Fellowship No. FS/17/81/33318. This project was enabled through access to the MRC eMedLab Medical Bioinformatics infrastructure (www.mrc.ac.uk; MR/L016311/1). HV and BM acknowledge funding from project no. TKP2021-NKTA-46 with the support provided by the Ministry of Innovation and Technology of Hungary from the National Research, Development and Innovation Fund, financed under the TKP2021-NKTA funding scheme and Project no. RRF-2.3.1–21-2022–00004 (MILAB) has been implemented with the support provided by the European Union.
The funders provided support in the form of salaries for authors as detailed above but did not have any additional role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Prof. Steffen Petersen.
Conflict of interest
SEP provides consultancy to Circle Cardiovascular Imaging Inc, Calgary, Alberta, Canada. CA and SN are Founders, Shareholders, and Directors of Caristo Diagnostics, a CT Image analysis spinout company from the University of Oxford. CA is also the inventor of relevant patents US10,695,023B2, PCT/GB2017/053262, GB2018/1818049.7, GR20180100490, and GR20180100510, licensed through exclusive license to Caristo Diagnostics. The remaining authors have no disclosures.
Statistics and biometry
AS and CM have significant statistical expertise and extensive experience working with UK Biobank data. AS led analysis and model development steps. CM led the creation of diagnostic labels, and performed dataset creation for all non-radiomic features.
Informed consent
Written informed consent was obtained from all subjects (patients) in this study.
Ethical approval
This study complies with the Declaration of Helsinki; the work was covered by the ethical approval for UK Biobank studies from the National Health Service (NHS) National Research Ethics Service on 17 June 2011 (Ref 11/NW/0382) and extended on 18 June 2021 (Ref 21/NW/0157) with written informed consent obtained from all participants.
Study subjects or cohorts overlap
This research was conducted using the UK Biobank resource under access application 2964. UK Biobank will make the data available to all bona fide researchers for all types of health-related research that is in the public interest, without preferential or exclusive access for any persons. All researchers will be subject to the same application process and approval criteria as specified by UK Biobank. For more details on the access procedure, see the UK Biobank website: http://www.ukbiobank.ac.uk/register-apply. Automated, quality-controlled pericardial adipose tissue segmentation was conducted in a previous publication by our research group (Bard et al, https://www.frontiersin.org/articles/10.3389/fcvm.2021.677574/full). This article is referenced in the manuscript. Of note, the current study is not an extension but rather a completely new layer of the research conducted using pericardial adipose tissue analysis. The analysis and model development of this study were conducted independently.
Methodology
•observational
•multicentre study
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
SEP and ZRE are joint senior authors.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Szabo, L., Salih, A., Pujadas, E.R. et al. Radiomics of pericardial fat: a new frontier in heart failure discrimination and prediction. Eur Radiol 34, 4113–4126 (2024). https://doi.org/10.1007/s00330-023-10311-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-023-10311-0