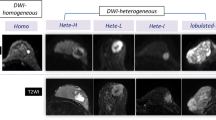
Abstract
Objectives
To evaluate the relationship of the signature index (S-index), an advanced diffusion MRI marker, and the immunohistochemical (IHC) status, proliferation rate, and molecular subtype of invasive breast cancers.
Methods
A retrospective study of patients with invasive carcinoma was conducted between 2017 and 2021. All patients underwent dynamic contrast-enhanced MRI and DWI using a 3-T system. For DWI, three b values (0, 200, and 1500 s/mm2) were used to derive the S-index. Three-dimensional ROIs were manually placed over the whole tumor on DWI. Mean and 85th percentile S-index values were compared to the IHC status, proliferation rate, and molecular subtypes of lesions.
Results
The study included 153 patients (mean age, 60 ± 13 years) with 160 invasive carcinomas. S-index values were significantly higher in estrogen receptor–positive (mean, p = .005; 85th percentile, p < .001) and progesterone receptor–positive (mean, p = .003; 85th percentile, p < .001) tumors, and significantly lower in human epidermal growth factor receptor 2 (HER2) – positive tumors (mean, p = .023; 85th percentile, p < .001). Mean and 85th percentile S-index values were significantly different among breast cancer subtypes (mean, p = .015; 85th percentile, p = .002), and the AUC of these values for the prediction of IHC status were 0.64 and 0.66 for HER2, and 0.70 and 0.74 for hormone receptors, respectively.
Conclusions
The DWI S-index showed significantly higher values in invasive carcinomas with immunohistochemical status associated with good prognosis, suggesting its usefulness as a noninvasive imaging biomarker to estimate IHC status and orient treatment.
Key Points
• The signature index, an advanced diffusion MRI marker, showed good discrimination of immunohistochemical status in invasive breast carcinomas.
• The signature index has the potential to differentiate noninvasively invasive breast carcinoma subtypes and appears as an imaging biomarker of prognostic factors and molecular phenotypes






Similar content being viewed by others
Abbreviations
- ADC:
-
Apparent diffusion coefficient
- AUC:
-
Area under the curve
- DWI:
-
Diffusion-weighted imaging
- DCE:
-
Dynamic contrast enhanced
- ER:
-
Estrogen receptor
- HER2:
-
Human epidermal growth factor receptor 2
- IHC:
-
Immunohistochemical
- IVIM:
-
Intravoxel incoherent motion
- PR:
-
Progesterone receptor
- ROC:
-
Receiver-operating characteristic
- ROI:
-
Region of interest
- S-index:
-
Signature index
- 3D:
-
Three dimensional
References
Baltzer P, Mann RM, Iima M et al (2020) Diffusion-weighted imaging of the breast-a consensus and mission statement from the EUSOBI International Breast Diffusion-Weighted Imaging working group. Eur Radiol 30:1436–1450
Iima M, Honda M, Sigmund EE, Ohno Kishimoto A, Kataoka M, Togashi K (2020) Diffusion MRI of the breast: current status and future directions. J Magn Reson Imaging 52:70–90
Goldhirsch A, Wood WC, Coates AS et al (2011) Strategies for subtypes--dealing with the diversity of breast cancer: highlights of the St. Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2011. Ann Oncol 22:1736–1747
Mann GB, Fahey VD, Feleppa F, Buchanan MR (2005) Reliance on hormone receptor assays of surgical specimens may compromise outcome in patients with breast cancer. J Clin Oncol 23:5148–5154
Burge CN, Chang HR, Apple SK (2006) Do the histologic features and results of breast cancer biomarker studies differ between core biopsy and surgical excision specimens? Breast 15:167–172
Orlando L, Viale G, Bria E et al (2016) Discordance in pathology report after central pathology review: implications for breast cancer adjuvant treatment. Breast 30:151–155
Martincich L, Deantoni V, Bertotto I et al (2012) Correlations between diffusion-weighted imaging and breast cancer biomarkers. Eur Radiol 22:1519–1528
Choi SY, Chang YW, Park HJ, Kim HJ, Hong SS, Seo DY (2012) Correlation of the apparent diffusion coefficiency values on diffusion-weighted imaging with prognostic factors for breast cancer. Br J Radiol 85:e474–e479
Horvat JV, Bernard-Davila B, Helbich TH et al (2019) Diffusion-weighted imaging (DWI) with apparent diffusion coefficient (ADC) map** as a quantitative imaging biomarker for prediction of immunohistochemical receptor status, proliferation rate, and molecular subtypes of breast cancer. J Magn Reson Imaging 50:836–846
Iima M, Kataoka M, Kanao S et al (2018) Intravoxel incoherent motion and quantitative non-Gaussian diffusion MR imaging: evaluation of the diagnostic and prognostic value of several markers of malignant and benign breast lesions. Radiology 287:432–441
Kim EJ, Kim SH, Park GE et al (2015) Histogram analysis of apparent diffusion coefficient at 3.0 t: correlation with prognostic factors and subtypes of invasive ductal carcinoma. J Magn Reson Imaging 42:1666–1678
Lee HS, Kim SH, Kang BJ, Baek JE, Song BJ (2016) Perfusion parameters in dynamic contrast-enhanced MRI and apparent diffusion coefficient value in diffusion-weighted MRI: association with prognostic factors in breast cancer. Acad Radiol 23:446–456
Makkat S, Luypaert R, Stadnik T et al (2008) Deconvolution-based dynamic contrast-enhanced MR imaging of breast tumors: correlation of tumor blood flow with human epidermal growth factor receptor 2 status and clinicopathologic findings--preliminary results. Radiology 249:471–482
Park SH, Choi HY, Hahn SY (2015) Correlations between apparent diffusion coefficient values of invasive ductal carcinoma and pathologic factors on diffusion-weighted MRI at 3.0 Tesla. J Magn Reson Imaging 41:175–182
Shen L, Zhou G, Tong T et al (2018) ADC at 3.0 T as a noninvasive biomarker for preoperative prediction of Ki67 expression in invasive ductal carcinoma of breast. Clin Imaging 52:16–22
Surov A, Clauser P, Chang YW et al (2018) Can diffusion-weighted imaging predict tumor grade and expression of Ki-67 in breast cancer? A multicenter analysis. Breast Cancer Res 20:58
Bokacheva L, Kaplan JB, Giri DD et al (2014) Intravoxel incoherent motion diffusion-weighted MRI at 3.0 T differentiates malignant breast lesions from benign lesions and breast parenchyma. J Magn Reson Imaging 40:813–823
Cho GY, Moy L, Kim SG et al (2016) Evaluation of breast cancer using intravoxel incoherent motion (IVIM) histogram analysis: comparison with malignant status, histological subtype, and molecular prognostic factors. Eur Radiol 26:2547–2558
Iima M, Le Bihan D (2016) Clinical intravoxel incoherent motion and diffusion MR imaging: past, present, and future. Radiology 278:13–32
Iima M, Yano K, Kataoka M et al (2015) Quantitative non-Gaussian diffusion and intravoxel incoherent motion magnetic resonance imaging: differentiation of malignant and benign breast lesions. Invest Radiol 50:205–211
Ma D, Lu F, Zou X et al (2017) Intravoxel incoherent motion diffusion-weighted imaging as an adjunct to dynamic contrast-enhanced MRI to improve accuracy of the differential diagnosis of benign and malignant breast lesions. Magn Reson Imaging 36:175–179
Sun K, Chen X, Chai W et al (2015) Breast cancer: diffusion kurtosis MR imaging-diagnostic accuracy and correlation with clinical-pathologic factors. Radiology 277:46–55
Goto M, Le Bihan D, Yoshida M, Sakai K, Yamada K (2019) Adding a model-free diffusion MRI marker to BI-RADS assessment improves specificity for diagnosing breast lesions. Radiology 292:84–93
Allison KH, Hammond MEH, Dowsett M et al (2020) Estrogen and progesterone receptor testing in breast cancer: ASCO/CAP guideline update. J Clin Oncol 38:1346–1366
Wolff AC, Hammond MEH, Allison KH et al (2018) Human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists clinical practice guideline focused update. J Clin Oncol 36:2105–2122
Dowsett M, Nielsen TO, A'Hern R et al (2011) Assessment of Ki67 in breast cancer: recommendations from the International Ki67 in Breast Cancer working group. J Natl Cancer Inst 103:1656–1664
Schnitt SJ (2010) Classification and prognosis of invasive breast cancer: from morphology to molecular taxonomy. Mod Pathol 23(Suppl 2):S60–S64
Chen X, Yuan Y, Gu Z, Shen K (2012) Accuracy of estrogen receptor, progesterone receptor, and HER2 status between core needle and open excision biopsy in breast cancer: a meta-analysis. Breast Cancer Res Treat 134:957–967
Pinker K, Shitano F, Sala E et al (2018) Background, current role, and potential applications of radiogenomics. J Magn Reson Imaging 47:604–620
Acknowledgements
We thank Cecilia Garrec for the English language corrections.
Funding
The authors state that this work has not received any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Mariko Goto, MD, PhD, lecturer of the Department of Radiology, Kyoto Prefectural University of Medicine.
Conflict of interest
The authors of this manuscript declare no relationships with any companies, whose products or services may be related to the subject matter of the article.
Statistics and Biometry
No complex statistical methods were necessary for this paper.
Informed consent
Written informed consent was waived by the Institutional Review Board.
Ethical approval
Institutional Review Board approval was obtained.
Study subjects or cohorts overlap
Some subjects have been included in a previous study which was published in Radiology (reference #23) to introduce and validate the method (S-index) for diagnosis purposes. The present study was performed with a cohort of 153 patients (with 160 lesions), including the 56 invasive breast carcinoma patients (with 60 lesions) included in the previous study, with a different goal (correlation with molecular biomarkers). We have included this important information in the manuscript at the part of “Materials and Methods.”
Methodology
• retrospective
• diagnostic or prognostic study
• performed at one institution
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Goto, M., Le Bihan, D., Sakai, K. et al. The diffusion MRI signature index is highly correlated with immunohistochemical status and molecular subtype of invasive breast carcinoma. Eur Radiol 32, 4879–4888 (2022). https://doi.org/10.1007/s00330-022-08562-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-022-08562-4