Abstract
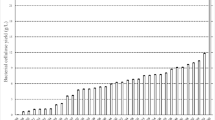
In the industrial application of bacterial cellulose (BC), the yield becomes a crucial factor because it drives the selling price of BC. A Box–Behnken experimental design with three components and three levels was employed to create seventeen samples. A response surface method was then used to optimize the BC yield from the most relevant parameters, including incubation temperature, medium shaking intensity, and nitrogen supply pH. Fourier transform infrared (FTIR) analysis proved that BC was pure. By analyzing BC morphology using scanning electron microscopy (SEM), rod-like microfibrils with an average diameter of 6.5 were revealed. X-ray diffraction (XRD) tested the crystalline size and crystallinity and found 4.7 nm and 69%, respectively. Thermal transition and stability were assessed using a differential scanning calorimeter (DSC) and a thermogravimetric analyzer (TGA). For the manufacture of BC composite hydrogel, a chemical test method was used to determine the solubility of BC. This study aimed to identify the ideal circumstances for using leftover sweet lime pulp for biomedical applications to produce reasonably priced and effective BC. At 28.87 °C, 125.91 rpm shaking frequency, and 5.65 pH, a sweet lime pulp waste medium produced the highest yield of BC, three times greater than the yield in a Hestrin–Schramm medium under static circumstances.
Graphical abstract








Similar content being viewed by others
Availability of data and materials
The raw data and all materials related to this research work are available with authors.
References
Abdel-Aziz MM, Elella MHA, Mohamed RR (2020) Green synthesis of quaternized chitosan/silver nanocomposites for targeting mycobacterium tuberculosis and lung carcinoma cells (A-549). Int J Biol Macromol 142:244–253
Elella MHA, Goda ES, Abdallah HM, Shalan AE, Gamal H, Yoon KR (2021) Innovative bactericidal adsorbents containing modified xanthan gum/montmorillonite nanocomposites for wastewater treatment. Int J Biol Macromol 167:1113–1125
Elella MA, Abdel-Aziz MM, Abd El-Ghany NA (2021) Synthesis of a high-performance antimicrobial o-quaternized alginate—a promising potential antimicrobial agent. Cellul Chem Technol Synth 55:75–86
Elgamal AM, Elella MHA, Saad GR, Abd El-Ghany NA (2022) Synthesis, characterization and swelling behavior of high-performance antimicrobial biocompatible copolymer based on carboxymethyl xanthan. Mater Today Commun 33:104209
Abu Elella MH, Abdallah HM, Gamal H, Moustafa EB, Goda ES (2022) Rational design of biocompatible IPNs hydrogels containing carboxymethyl starch and trimethyl chitosan chloride with high antibacterial activity. Cellulose 29(13):7317–7330
Elella MHA, Shalan AE, Sabaa MW, Mohamed RR (2022) One-pot green synthesis of antimicrobial chitosan derivative nanocomposites to control foodborne pathogens. RSC Adv 12(2):1095–1104
Shoda M, Sugano Y (2005) Recent advances in bacterial cellulose production. Biotechnol Bioprocess Eng 10:1–8
Schlufter K, Schmauder HP, Dorn S, Heinze T (2006) Efficient homogeneous chemical modification of bacterial cellulose in the ionic liquid 1-N-butyl-3-methylimidazolium chloride. Macromol Rapid Commun 27(19):1670–1676
Shi Z, Zhang Y, Phillips GO, Yang G (2014) Utilization of bacterial cellulose in food. Food Hydrocoll 35:539–545
Barud H, Ribeiro C, Crespi M, Martines M, Dexpert-Ghys J, Marques R, Ribeiro S (2007) Thermal characterization of bacterial cellulose–phosphate composite membranes. J Therm Anal Calorim 87(3):815–818
Lin N, Dufresne A (2014) Nanocellulose in biomedicine: current status and future prospect. Eur Polym J 59:302–325
Stumpf TR, Yang X, Zhang J, Cao X (2018) In situ and ex situ modifications of bacterial cellulose for applications in tissue engineering. Mater Sci Eng C 82:372–383
Hestrin S, Schramm MJBJ (1954) Synthesis of cellulose by Acetobacter xylinum. 2. Preparation of freeze-dried cells capable of polymerizing glucose to cellulose. Biochem J 58(2):345–352
Mikkelsen D, Flanagan BM, Dykes GA, Gidley MJ (2009) Influence of different carbon sources on bacterial cellulose production by Gluconacetobacter xylinus strain ATCC 53524. J Appl Microbiol 107(2):576–583
Du R, Wang Y, Zhao F, Qiao X, Song Q, Li S, Zhou Z (2020) Production, optimization and partial characterization of bacterial cellulose from Gluconacetobacter xylinus TJU-D2. Waste Biomass Valor 11(5):1681–1690
Keshk SMAS, Sameshima K (2005) Evaluation of different carbon sources for bacterial cellulose production. Afr J Biotechnol 4(6):478–482
Revin V, Liyaskina E, Nazarkina M, Bogatyreva A, Shchankin M (2018) Cost-effective production of bacterial cellulose using acidic food industry by-products. Braz J Microbiol 49:151–159
Liu M, Li S, **e Y, Jia S, Hou Y, Zou Y, Zhong C (2018) Enhanced bacterial cellulose production by Gluconacetobacter xylinus via expression of Vitreoscilla hemoglobin and oxygen tension regulation. Appl Microbiol Biotechnol 102(3):1155–1165
Gullo M, La China S, Falcone PM, Giudici P (2018) Biotechnological production of cellulose by acetic acid bacteria: current state and perspectives. Appl Microbiol Biotechnol 102(16):6885–6898
Vandamme EJ, De Baets S, Vanbaelen A, Joris K, De Wulf P (1998) Improved production of bacterial cellulose and its application potential. Polym Degrad Stab 59(1–3):93–99
Hungund BS, Gupta SG (2010) Factors affecting production of cellulose from Gluconacetobacter xylinus. Asian J Microbiol Biotechnol Environ Sci 12(3):517–522
Embuscado ME, Marks JS, Bemiller JN (1994) Bacterial cellulose. I. Factors affecting the production of cellulose by Acetobacter xylinum. Food Hydrocoll 8(5):407–418
Jagannath A, Kalaiselvan A, Manjunatha SS, Raju PS, Bawa AS (2008) The effect of pH, sucrose and ammonium sulphate concentrations on the production of bacterial cellulose (Nata-de-coco) by Acetobacter xylinum. World J Microbiol Biotechnol 24(11):2593
Fernandes IDAA, Pedro AC, Ribeiro VR, Bortolini DG, Ozaki MSC, Maciel GM, Haminiuk CWI (2020) Bacterial cellulose: from production optimization to new applications. Int J Biol Macromol 164:2598–2611
Lotfiman S, Awang Biak DR, Ti TB, Kamarudin S, Nikbin S (2018) Influence of date syrup as a carbon source on bacterial cellulose production by Acetobacter xylinum 0416. Adv Polym Technol 37(4):1085–1091
Raiszadeh-Jahromi Y, Rezazadeh-Bari M, Almasi H, Amiri S (2020) Optimization of bacterial cellulose production by Komagataeibacter xylinus PTCC 1734 in a low-cost medium using optimal combined design. J Food Sci Technol 57(7):2524–2533
Tunga R, Banerjee R, Bhattacharyya BC (1998) Optimizing some factors affecting protease production under solid state fermentation. Bioproc Eng 19(3):187–190
Son HJ, Heo MS, Kim YG, Lee SJ (2001) Optimization of fermentation conditions for the production of bacterial cellulose by a newly isolated Acetobacter sp.A9 in shaking cultures. Biotechnol Appl Biochem 33(1):1–5
Qi GX, Luo MT, Huang C, Guo HJ, Chen XF, **ong L, Chen XD (2017) Comparison of bacterial cellulose production by Gluconacetobacter xylinus on bagasse acid and enzymatic hydrolysates. J Appl Polym Sci 134(28):45066
Urbina L, Hernández-Arriaga AM, Eceiza A, Gabilondo N, Corcuera MA, Prieto MA, Retegi A (2017) By-products of the cider production: an alternative source of nutrients to produce bacterial cellulose. Cellul 24:2071–2082
Machado RT, Meneguin AB, Sábio RM, Franco DF, Antonio SG, Gutierrez J, Barud HS (2018) Komagataeibacter rhaeticus grown in sugarcane molasses-supplemented culture medium as a strategy for enhancing bacterial cellulose production. Ind Crops Prod 122:637–646
Molina-Ramírez C, Castro C, Zuluaga R, Gañán P (2018) Physical characterization of bacterial cellulose produced by Komagataeibacter medellinensis using food supply chain waste and agricultural by-products as alternative low-cost feedstocks. J Polym Environ 26:830–837
Abdelraof M, Hasanin MS, El-Saied H (2019) Ecofriendly green conversion of potato peel wastes to high productivity bacterial cellulose. Carbohydr Polym 211:75–83
Ye J, Zheng S, Zhang Z, Yang F, Ma K, Feng Y, Yang X (2019) Bacterial cellulose production by Acetobacter xylinum ATCC 23767 using tobacco waste extract as culture medium. Bioresour Technol 274:518–524
Skiba EA, Budaeva VV, Ovchinnikova EV, Gladysheva EK, Kashcheyeva EI, Pavlov IN, Sakovich GV (2020) A technology for pilot production of bacterial cellulose from oat hulls. J Chem Eng 383:123128
Souza EF, Furtado MR, Carvalho CW, Freitas-Silva O, Gottschalk LM (2020) Production and characterization of Gluconacetobacter xylinus bacterial cellulose using cashew apple juice and soybean molasses. Int J Biol Macromol 146:285–289
Li ZY, Azi F, Ge ZW, Liu YF, Yin XT, Dong MS (2021) Bio-conversion of kitchen waste into bacterial cellulose using a new multiple carbon utilizing Komagataeibacter rhaeticus: fermentation profiles and genome-wide analysis. Int J Biol Macromol 191:211–221
Mohamad S, Abdullah LC, Jamari SS, Osman Al Edrus SS, Aung MM, Sy Mohamad SF (2022) Production and characterization of bacterial cellulose nanofiber by Acetobacter xylinum 0416 using only oil palm frond juice as fermentation medium. J Nat Fibers 19(17):16005–16016
Li W, Huang X, Liu H, Lian H, Xu B, Zhang W, Zhong C (2023) Improvement in bacterial cellulose production by co-culturing Bacillus cereus and Komagataeibacter xylinus. Carbohydr Polym 313:120892
Bae S, Shoda M (2005) Statistical optimization of culture conditions for bacterial cellulose production using Box–Behnken design. Biotechnol Bioeng 90(1):20–28
Panesar PS, Chavan Y, Chopra HK, Kennedy JF (2012) Production of microbial cellulose: response surface methodology approach. Carbohydr Polym 87(1):930–934
Montgomery DC (2017) Design and analysis of experiments. Wiley, Hoboken
Lee KY, Buldum G, Mantalaris A, Bismarck A (2014) More than meets the eye in bacterial cellulose: biosynthesis, bioprocessing, and applications in advanced fiber composites. Macromol Biosci 14(1):10–32
Cannon RE, Anderson SM (1991) Biogenesis of bacterial cellulose. Crit Rev Microbiol 17(6):435–447
Mohite BV, Salunke BK, Patil SV (2013) Enhanced production of bacterial cellulose by using Gluconacetobacter hansenii NCIM 2529 strain under shaking conditions. Appl Biochem Biotechnol 169:1497–1511
Ross P, Mayer R, Benziman M (1991) Cellulose biosynthesis and function in bacteria. Microbiol Rev 55(1):35–58
Riaz T, Zeeshan R, Zarif F, Ilyas K, Muhammad N, Safi SZ, Rahim A, Rizvi SA, Rehman IU (2018) FTIR analysis of natural and synthetic collagen. Appl Spectroscop Rev 53(9):703–746
Nandiyanto ABD, Oktiani R, Ragadhita R (2019) How to read and interpret FTIR spectroscope of organic material. Ind J Sci Technol 4(1):97–118
Gong J, Li J, Xu J, **ang Z, Mo L (2017) Research on cellulose nanocrystals produced from cellulose sources with various polymorphs. RSC Adv 7(53):33486–33493
French AD (2014) Idealized powder diffraction patterns for cellulose polymorphs. Cellulose. https://doi.org/10.1007/s10570-013-0030-4
Chiriac AI, Pastor FI, Popa VI, Aflori M, Ciolacu D (2014) Changes of supramolecular cellulose structure and accessibility induced by the processive endoglucanase Cel9B from Paenibacillus barcinonensis. Cellulose 21:203–219
Moharram MA, Mahmoud OM (2007) X-ray diffraction methods in the study of the effect of microwave heating on the transformation of cellulose I into cellulose II during mercerization. J Appl Polym Sci 105(5):2978–2983
French AD, Santiago Cintrón M (2013) Cellulose polymorphy, crystallite size, and the Segal Crystallinity Index. Cellulose 20:583–588
Nam S, French AD, Condon BD, Concha M (2016) Segal crystallinity index revisited by the simulation of X-ray diffraction patterns of cotton cellulose Iβ and cellulose II. Carbohydr Polym 135:1–9
George J, Sajeevkumar VA, Kumar R, Ramana KV, Sabapathy SN, Bawa AS (2008) Enhancement of thermal stability associated with the chemical treatment of bacterial (Gluconacetobacter xylinus) cellulose. J Appl Polym Sci 108(3):1845–1851
Ul-Islam M, Khan T, Park JK (2012) Water holding and release properties of bacterial cellulose obtained by in situ and ex situ modification. Carbohydr Polym 88(2):596–603
Mohite BV, Patil SV (2014) Physical, structural, mechanical and thermal characterization of bacterial cellulose by G. hansenii NCIM 2529. Carbohydr Polym 106:132–141
Yan H, Huang D, Chen X, Liu H, Feng Y, Zhao Z, Lin Q (2018) A novel and homogeneous scaffold material: preparation and evaluation of alginate/bacterial cellulose nanocrystals/collagen composite hydrogel for tissue engineering. Polym Bullet 75:985–1000
Acknowledgements
Authors acknowledge the support of Technical Educational Quality Improvement Program-III (TEQIP-III), Ministry of Education, and Govt. of India.
Funding
There is no funding agency directly funded or supported this research work. The research work is conducted for the partial fulfilment of doctor of philosophy of Mr. Ashutosh Pandey.
Author information
Authors and Affiliations
Contributions
Conceptualization: [AP, AS, MKS], Methodology: [AS, AP], formal analysis and investigation: [MKS, AP], writing—original draft preparation: [AP, MKS, AS], writing—review and editing: [AS, AP], resources: [MKS, AP], supervision: [MKS, AS].
Corresponding author
Ethics declarations
Conflict of interest
Mr. Ashutosh Pandey has received the scholarship under Dr. Homi Bhabha scheme by Dr. APJ Kalam Technical University, Lucknow, India and partial support under Technical Educational Quality Improvement Program-III (TEQIP-III), Ministry of Education, and Govt. of India. Mukesh Kumar Singh and Annika Singh did not receive any research fund from any funding agency/company/department.
Ethical approval
Author ensures that all the authors mentioned in the article have agreed for authorship, read and approved the manuscript, and given consent for the submission and subsequent publication of the manuscript (the authorship should be based on the ICMJE guidelines).
Consent for participate
Informing participants about the research work and its nature, purpose and seeking their consent for participation. Author state that written or oral informed consent have been obtained from the all authors mentioned in the article and relevant documents must be provide when requested by journal.
Consent for publication
All three authors have extended their consent to communicate this research work for publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pandey, A., Singh, A. & Singh, M.K. Novel low-cost green method for production bacterial cellulose. Polym. Bull. 81, 6721–6741 (2024). https://doi.org/10.1007/s00289-023-05023-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-023-05023-w