Abstract
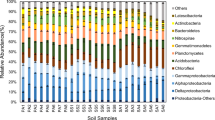
Soil denitrification is an important process in the emission of N2O, an atmospheric greenhouse gas. Environmental factors of different vegetation types are largely heterogeneous, which may directly or indirectly affect N2O fluxes. Through high-throughput sequencing of the nitrite reductase gene nirS, this study investigated the influence of vegetation type on the structure and diversity of denitrifying microbial communities in Qinghai Lake wetlands, China. The results showed that among the four vegetation types in the Qinghai Lake wetlands, Carex rigescens (CR) had the highest species richness index, and Leymus secalinus (LS) had the lowest species richness index. Species evenness followed the opposite trend. Proteobacteria were the main denitrifying bacterial phylum in the wetland soil of Qinghai Lake. There were 40 differential bacterial flora at different levels in the four vegetation types, most of which belonged to Proteobacteria. Magnetospirillum is a bacterium that differed significantly across the four vegetation types, and it was one of the main denitrifying taxa based on relative abundance in the LS vegetation type. Soil pH was the most important regulating factor of nirS-type denitrifying microbial community in Qinghai Lake wetland. In summary, the succession of vegetation types in the Qinghai Lake Wetlands changes the soil microenvironment and significantly affects the community structure and diversity of the denitrifying microbial communities. The large-area growth of CR might even increase the emission of nitrous oxide. This study can serve as a reference for further exploration of the N2O emission mechanism in the unique habitats of alpine wetlands.





Similar content being viewed by others
Data Availability
The raw data have been uploaded to NCBI, and Its BioProject is PRJNA807501.
Code Availability
The code and table data for this article are available from the web page(https:// github.com /Zhangni111 /nirSDMCoystersAnalysis) and are available.
References
IPCC (2013) Climate change 2013: the physical science basis. Contribution of working group I to the fifth assessment report of the intergovernmental panel on climate change. Cambridge University Press, Cambridge
Rodhe H (1990) A comparison of the contribution of various gases to the greenhouse effect. Science 248:1217–1219. https://doi.org/10.1126/science.248.4960.1217
Forster P, Ramaswamy V, Artaxo P et al (2007) Changes in atmospheric constituents and in radiative forcing. Cambridge University Press, Cambridge
Metz B, Davidson OR, Bosch PR, Dave R, Meyer LA (2007) Contribution of Working Group III to the fourth assessment report of the intergovernmental panel on climate change. Cambridge University Press, Cambridge
van den Elzen E, van den Berg LJL, van der Weijden B, Fritz C, Sheppard LJ, Lamers LPM (2018) Effects of airborne ammonium and nitrate pollution strongly differ in peat bogs, but symbiotic nitrogen fixation remains unaffected. Sci Total Environ 610–611:732–740. https://doi.org/10.1016/j.scitotenv.2017.08.102
Wu H, Zhang J, Ngo HH, Guo W, Liang S (2017) Evaluating the sustainability of free water surface flow constructed wetlands: methane and nitrous oxide emissions. J Clean Prod 147:152–156. https://doi.org/10.1016/j.jclepro.2017.01.091
Song C (2003) Research progress on carbon cycle in wetland ecosystems. Sci Geog Sin 23:622–628. https://doi.org/10.1097/00006982-200310000-00003
Zhao J, Luo T, Li R, Wei H, Li X, Du M, Tang Y (2018) Precipitation alters temperature effects on ecosystem respiration in Tibetan alpine meadows. Agric For Meteorol 252:121–129. https://doi.org/10.1016/j.agrformet.2018.01.014
**ng Y, Jiang Q, Li W, Bai L (2009) Landscape spatial patterns changes of the wetland in Qinghai-Tibet Plateau. Ecol Environ Sci 18:1010–1015. https://doi.org/10.16258/j.cnki.1674-5906.2009.03.034
Bai J, Ouyang H, Xu H, Zhou C, Gao J (2004) Recent changes in wetlands on the Tibetan Plateau: a review. Prog Geog 23:1–9. https://doi.org/10.11820/dlkxjz.2004.04.001
Chen H, Du M, Wang D, Zhou Y, Zeng L, Yang X (2020) Influence of chlortetracycline as an antibiotic residue on nitrous oxide emissions from wastewater treatment. Bioresour Technol 313:123696. https://doi.org/10.1016/j.biortech.2020.123696
Wang Y, Xue M, Huang Y, Liu G, Wang M, Gi B (2003) Greenhouse gases emission or uptake in Inner Mongolia natural and free-grazing grasslands. J Appl Ecol 14:372–376. https://doi.org/10.1287/j.1001-9332.2003.0084
Bouwman AF (1990) Soils and the greenhouse effect. Wiley, New York
Yin C, Fan F, Song A, Fan X, Ding H, Ran W, Qiu H, Liang Y (2017) The response patterns of community traits of N2O emission-related functional guilds to temperature across different arable soils under inorganic fertilization. Soil Biol Biochem 108:65–77. https://doi.org/10.1016/j.soilbio.2017.01.022
Zumft WG (1997) Cell biology and molecular basis of denitrification. Microbiol Mol Biol Rev 61:533–616. https://doi.org/10.1128/mmbr.61.4.533-616.1997
Mosier AC, Francis CA (2010) Denitrifier abundance and activity across the San Francisco Bay estuary. Environ Microbiol Rep 2:667–676. https://doi.org/10.1111/j.1758-2229.2010.00156.x
Zhao S, Zhou J, Yuan D, Wang W, Zhou L, Pi Y, Zhu G (2020) NirS-type N2O-producers and nosZ II-type N2O-reducers determine the N2O emission potential in farmland rhizosphere soils. J Soils Sediments 20:461–471. https://doi.org/10.1007/s11368-019-02395-3
Wu X, Liu G, Butterbach-Bahl K, Fu B, Zheng X, Brüggemann N (2013) Effects of land cover and soil properties on denitrification potential in soils of two semi-arid grasslands in Inner Mongolia, China. J Arid Environ 92:98–101. https://doi.org/10.1016/j.jaridenv.2013.02.003
Groh TA, Davis MP, Isenhart TM, Jaynes DB, Parkin TB (2019) Denitrification potential in three saturated riparian buffers. Agric Ecosyst Environ 286:106656. https://doi.org/10.1016/j.agee.2019.106656
Bai J, Wang X, Jia J, Zhang G, Wang Y, Zhang S (2017) Denitrification of soil nitrogen in coastal and inland salt marshes with different flooding frequencies. Phys Chem Earth Parts A/B/C 97:31–36. https://doi.org/10.1016/j.pce.2017.01.015
Bowen H, Maul JE, Cavigelli MA, Yarwood S (2020) Denitrifier abundance and community composition linked to denitrification activity in an agricultural and wetland soil. Appl Soil Ecol 151:103521. https://doi.org/10.1016/j.apsoil.2020.103521
Šimek M, Jı́šová L, Hopkins DW (2002) What is the so-called optimum pH for denitrification in soil? Soil Biol Biochem 34(1227):1234. https://doi.org/10.1016/s0038-0717(02)00059-7
Du Y, Ke X, Li J, Wang Y, Cao G, Guo X, Chen K (2021) Nitrogen deposition increases global grassland N2O emission rates steeply: a meta-analysis. CATENA 199:105105. https://doi.org/10.1016/j.catena.2020.105105
Zhang L, Song L, Zhang L, Shao H, Chen X, Yan K (2013) Seasonal dynamics in nitrous oxide emissions under different types of vegetation in saline-alkaline soils of the Yellow River Delta, China and implications for eco-restoring coastal wetland. Ecol Eng 61:82–89. https://doi.org/10.1016/j.ecoleng.2013.09.065
Kachenchart B, Jones DL, Gajaseni N, Edwards-Jones G, Limsakul A (2012) Seasonal nitrous oxide emissions from different land uses and their controlling factors in a tropical riparian ecosystem. Agric Ecosyst Environ 158:15–30. https://doi.org/10.1016/j.agee.2012.05.008
Masella AP, Bartram AK, Truszkowski JM, Brown DG, Neufeld JD (2012) PANDAseq: paired-end assembler for illumina sequences. BMC Bioinform 13:31. https://doi.org/10.1186/1471-2105-13-31
Schmieder R, Edwards R (2011) Quality control and preprocessing of metagenomic datasets. Bioinformatics 27:863–864. https://doi.org/10.1093/bioinformatics/btr026
Edgar R (2018) Taxonomy annotation and guide tree errors in 16S rRNA databases. PeerJ 6:e5030. https://doi.org/10.7717/peerj.5030
Wang Y, Sheng HF, He Y, Wu JY, Jiang YX, Tam NFY, Zhou HW (2012) Comparison of the levels of bacterial diversity in freshwater, intertidal wetland, and marine sediments by using millions of illumina tags. Appl Environ Microbiol 78:8264–8271. https://doi.org/10.1128/AEM.01821-12
Caporaso JG, Kuczynski J, Stombaugh J et al (2010) QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7:335–336. https://doi.org/10.1038/nmeth.f.303
Liang S, Deng J, Jiang Y, Wu S, Zhou Y, Zhu W (2019) Functional distribution of bacterial community under different land use patterns based on FAPROTAX function prediction. Pol J Environ Stud 29:1245–1261. https://doi.org/10.15244/pjoes/108510
Braak CJFT, Smilauer P (2002) CANOCO reference manual and canodraw for windows user’s guide: software for canonical community ordination (version 4.5). CANOCO, Ithaca
Morales SE, Jha N, Saggar S (2015) Biogeography and biophysicochemical traits link N2O emissions, N2O emission potential and microbial communities across New Zealand pasture soils. Soil Biol Biochem 82:87–98. https://doi.org/10.1016/j.soilbio.2014.12.01834
Elmendorf SC, Henry GH, Hollister RD, Bjork RG, Bjorkman AD, Callaghan TV, Collier LS, Cooper EJ, Cornelissen JH, Day TA (2012) Global assessment of experimental climate warming on tundra vegetation: heterogeneity over space and time. Ecol Lett 15:164–175. https://doi.org/10.1111/j.1461-0248.2011.01716.x
Yang H, Wu M, Liu W, Zhang Z, Zhang N, Wan S (2011) Community structure and composition in response to climate change in a temperate steppe. Glob Chang Biol 17:452–465. https://doi.org/10.1111/j.1365-2486.2010.02253.x
Sun J, Cheng G, Li W (2013) Meta-analysis of relationships between environmental factors and aboveground biomass in the alpine grassland on the Tibetan Plateau. Biogeosciences 10:1707–1715. https://doi.org/10.5194/bg-10-1707-2013
Nemergut DR, Anderson SP, Cleveland CC, Martin AP, Miller AE, Seimon A, Schmidt SK (2006) Microbial community succession in an unvegetated, recently deglaciated soil. Microb Ecol 53:110–122. https://doi.org/10.1007/s00248-006-9144-7
He M, He Y, Wu C, Ou J, Lin Y, Fang Z, Han X, Xu X (2019) Effects of rocky desertification intensity on soil fungal composition and diversity during karst vegetation succession. Mycosystema 38:471–484. https://doi.org/10.13346/j.mycosystema.180277
Tan B, Wang L, Pei H, **a X, Ding J, Chang Y, Hao Z (2020) The seasonal structural and functional characteristics of bacterial community in Apostichopus japonicus culture pond water. Prog Fish Sci 42:77–88. https://doi.org/10.19663/j.issn2095-9869.20200716002
Li Y, Ma J, Gao C, Huo S, **a X (2021) Denitrification rates and nirS-type denitrifying bacteria community structure characteristics of bulk and rhizosphere soil in spring and summer in alpine wetlands of Qinghai-Tibet Plateau. Environ Sci. https://doi.org/10.13227/j.hjkx.202101258
Bazylinski DA, Frankel RB, Heywood BR, Mann S, King JW, Donaghay PL, Hanson AK (1995) Controlled biomineralization of magnetite (Fe(inf3)O(inf4)) and Greigite (Fe(inf3)S(inf4)) in a magnetotactic bacterium. Appl Environ Microbiol 61:3232–3239. https://doi.org/10.1128/aem.61.9.3232-3239.1995
Bo T (2012) Study on the denitrification pathway and DNA damage repair system in magnetospirilum magneticum AMB-1 and their influences on magnetsome formation as well as its genetic stability. Shandong University, **an
Waid JS (1999) Does soil biodiversity depend upon metabiotic activity and influences? Appl Soil Ecol 13:151–158. https://doi.org/10.1016/s0929-1393(99)00030-x
Liu J, Dang P, Gao Y, Zhu H, Zhu H, Zhao F, Zhao Z (2018) Effects of tree species and soil properties on the composition and diversity of the soil bacterial community following afforestation. For Ecol Manag 427:342–349. https://doi.org/10.1016/j.foreco.2018.06.017
Azziz G, Monza J, Etchebehere C, Irisarri P (2017) nirS- and nirK-type denitrifier communities are differentially affected by soil type, rice cultivar and water management. Eur J Soil Biol 78:20–28. https://doi.org/10.1016/j.ejsobi.2016.11.003
Tripathi BM, Stegen JC, Kim M, Dong K, Adams JM, Lee YK (2018) Soil pH mediates the balance between stochastic and deterministic assembly of bacteria. ISME J 12:1072–1083. https://doi.org/10.1038/s41396-018-0082-4
Qin J, Gao Z, Yue A, Zhang Y, Gao C, Zhao J, Wang M, Du W (2020) Isolation, identification and stress resistance analysis of a mesorhizobium isolated from soybean variety **da 53. Soyb Sci 39:898–905. https://doi.org/10.11861/j.issn.1000-9841.2020.06.0898
He XY, Su YR, Liang YM, Chen XB, Zhu HH, Wang KL (2012) Land reclamation and short-term cultivation change soil microbial communities and bacterial metabolic profiles. J Sci Food Agric 92:1103–1111. https://doi.org/10.1002/jsfa.5547
Acknowledgements
We declare that we have no financial and personal relationships with other people or organizations that can inappropriately influence our work. The authors would like to thank the editor and anonymous reviewers for their valuable comments and suggestions to improve the quality of this paper.
Funding
This work was supported by the National Natural Science Foundation of China [Grant Number 41661023], the Second Comprehensive Scientific Expedition to the Qinghai-Tibet Plateau [Grant Number 2019QZKK0405], and Qinghai Provincial Key Laboratory of Physical Geography and Environmental Process, Special Project of Qinghai Innovation Platform Construction [Grant Number 2020-ZJ-Y06].
Author information
Authors and Affiliations
Contributions
NZ, KC and YD designed the experiment, NZ, YY, HB, DZ, WQ and BC jointly completed the experiment, ZN, YY and JY jointly analyzed the data, NZ completed the first draft, KC and YD revised the manuscript; All authors have read and approved the manuscript for publication.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, N., Chen, K., Du, Y. et al. The Influence Mechanism of Vegetation Type on the Characteristics of nirS-Type Denitrifying Microbial Communities in Qinghai Lake Wetlands. Curr Microbiol 79, 242 (2022). https://doi.org/10.1007/s00284-022-02912-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00284-022-02912-7