Abstract
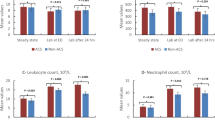
The aims of this study were to determine the possible relationships between the levels of hemin, hemopexin, acid sphingomyelinase, nitrite/nitrate (NOx), and other parameters in patients with SCD and to assess whether they were associated with vaso-occlusive crises (VOCs) or acute chest syndrome (ACS). Patients with SCD (homozygous or sickle beta-thalassemia) who were confirmed to have VOC or ACS were included. Blood samples were obtained at admission, on the third day of hospitalization, and at steady state. Demographic characteristics, pain (visual analog scale), complication history, complete blood count, lactate dehydrogenase, and C-reactive protein levels were recorded. Hemin, hemopexin, acid sphingomyelinase, and NOx were measured via ELISA. A total of 31 patients (22 VOC, 9 ACS) were included. Mean age was 16.4 ± 4.7 years. Admission white blood cell count and C-reactive protein levels were significantly higher in the ACS group. Patients with ACS also demonstrated a significant decreasing trend of LDH and an increasing trend of NOx values from admission to steady state. Notably, hemopexin levels were significantly lower on the third day of hospitalization compared to steady-state levels. Despite limited patient count in the ACS group, these patients appear to have strikingly greater inflammatory activation at admission, and the progression of ACS may be associated with LDH and NOx levels. Lower hemopexin levels during hospitalization versus steady state appear to support a role for the administration of hemopexin therapy during crises.


Similar content being viewed by others
References
Cappelli B, Gluckman E, Ghanem K, Abboud MR (2019) Hemoglobinopathies (sickle cell disease and thalassemia). In: Carreras E, Dufour C, Mohty M, Kröger N (eds) The EBMT handbook: hematopoietic stem cell transplantation and cellular therapies. Springer International Publishing, Cham, pp 595–601
Cançado RD (2012) Sickle cell disease: looking back but towards the future. Rev Bras Hematol Hemoter 34(3):175–177. https://doi.org/10.5581/1516-8484.20120041
Darbari DS, Sheehan VA, Ballas SK (2020) The vaso-occlusive pain crisis in sickle cell disease: definition, pathophysiology, and management. Eur J Haematol 105(3):237–246. https://doi.org/10.1111/ejh.13430
Veluswamy S, Shah P, Denton CC, Chalacheva P, Khoo MCK, Coates TD (2019) Vaso-occlusion in sickle cell disease: is autonomic dysregulation of the microvasculature the trigger? J Clin Med 8(10):1690. https://doi.org/10.3390/jcm8101690
Itoh T, Yamada T, Kodera Y, Matsushima A, Hiroto M, Sakurai K, Nishimura H, Inada Y (2001) Hemin (Fe(3+))– and heme (Fe(2+))–smectite conjugates as a model of hemoprotein based on spectrophotometry. Bioconjug Chem 12(1):3–6. https://doi.org/10.1021/bc000055q
Ansari J, Gavins FNE (2019) Ischemia-reperfusion injury in sickle cell disease: from basics to therapeutics. Am J Pathol 189(4):706–718. https://doi.org/10.1016/j.ajpath.2018.12.012
Jain S, Bakshi N, Krishnamurti L (2017) Acute chest syndrome in children with sickle cell disease. Pediatr Allergy Immunol Pulmonol 30(4):191–201. https://doi.org/10.1089/ped.2017.0814
Kato GJ, Steinberg MH, Gladwin MT (2017) Intravascular hemolysis and the pathophysiology of sickle cell disease. J Clin Invest 127(3):750–760. https://doi.org/10.1172/jci89741
Antwi-Boasiako C, Campbell AD (2018) Low nitric oxide level is implicated in sickle cell disease and its complications in Ghana. Vasc Health Risk Manag 14:199–204. https://doi.org/10.2147/vhrm.S163228
Jenkins RW, Canals D, Hannun YA (2009) Roles and regulation of secretory and lysosomal acid sphingomyelinase. Cell Signal 21(6):836–846. https://doi.org/10.1016/j.cellsig.2009.01.026
Awojoodu AO, Keegan PM, Lane AR, Zhang Y, Lynch KR, Platt MO, Botchwey EA (2014) Acid sphingomyelinase is activated in sickle cell erythrocytes and contributes to inflammatory microparticle generation in SCD. Blood 124(12):1941–1950. https://doi.org/10.1182/blood-2014-01-543652
Milton JN, Rooks H, Drasar E, Mccabe EL, Baldwin CT, Melista E, Gordeuk VR, Nouraie M, Kato GR, Minniti C, Taylor J, Campbell A, Luchtman-Jones L, Rana S, Castro O, Zhang Y, Thein SL, Sebastiani P, Gladwin MT, Steinberg MH (2013) Genetic determinants of haemolysis in sickle cell anaemia. Br J Haematol 161(2):270–278. https://doi.org/10.1111/bjh.12245
Kato GJ, Mcgowan V, Machado RF, Little JA, Taylor JT, Morris CR, Nichols JS, Wang X, Poljakovic M, Morris SM Jr, Gladwin MT (2006) Lactate dehydrogenase as a biomarker of hemolysis-associated nitric oxide resistance, priapism, leg ulceration, pulmonary hypertension, and death in patients with sickle cell disease. Blood 107(6):2279–2285. https://doi.org/10.1182/blood-2005-06-2373
Gardner K, Thein SL (2015) Super-elevated LDH and thrombocytopenia are markers of a severe subtype of vaso-occlusive crisis in sickle cell disease. Am J Hematol 90(10):E206–E207. https://doi.org/10.1002/ajh.24126
Damanhouri GA, Jarullah J, Marouf S, Hindawi SI, Mushtaq G, Kamal MA (2015) Clinical biomarkers in sickle cell disease. Saudi J Biol Sci 22(1):24–31. https://doi.org/10.1016/j.sjbs.2014.09.005
StankovicStojanovic K, Lionnet F (2016) Lactate dehydrogenase in sickle cell disease. Clin Chim Acta 458:99–102. https://doi.org/10.1016/j.cca.2016.04.035
Ballas SK (2015) Clinical utility of lactate dehydrogenase in determining the severity of hemolysis in sickle cell anemia. Am J Clin Pathol 144(1):173–174. https://doi.org/10.1309/AJCP3CCVKYYUZLFD
Bartolucci P, Habibi A, Khellaf M, Roudot-Thoraval F, Melica G, Lascaux AS, Moutereau S, Loric S, Wagner-Ballon O, Berkenou J, Santin A, Michel M, Renaud B, Lévy Y, Galactéros F, Godeau B (2016) Score predicting acute chest syndrome during vaso-occlusive crises in adult sickle-cell disease patients. EBioMedicine 10:305–311. https://doi.org/10.1016/j.ebiom.2016.06.038
Yalcinkaya A, Unal S, Oztas Y (2019) Altered HDL particle in sickle cell disease: decreased cholesterol content is associated with hemolysis, whereas decreased Apolipoprotein A1 is linked to inflammation. Lipids Health Dis 18(1):225. https://doi.org/10.1186/s12944-019-1174-5
Muller-Eberhard U, Javid J, Liem HH, Hanstein A, Hanna M (1968) Plasma concentrations of hemopexin, haptoglobin and heme in patients with various hemolytic diseases. Blood 32(5):811–815
Naumann HN, Diggs LW, Barreras L, Williams BJ (1971) Plasma hemoglobin and hemoglobin fractions in sickle cell crisis. Am J Clin Pathol 56(2):137–147. https://doi.org/10.1093/ajcp/56.2.137
Lundh B, Gardner FH (1971) The effect of testosterone in pharmacological doses on plasma volume and on some serum proteins in patients with sickle cell anemia and in sexually impotent men. Scand J Clin Lab Invest 28(1):72–78
Santiago RP, Guarda CC, Figueiredo CVB, Fiuza LM, Aleluia MM, Adanho CSA, Carvalho MOS, Pitanga TN, Zanette DL, Lyra IM, Nascimento VML, Vercellotti GM, Belcher JD, Goncalves MS (2018) Serum haptoglobin and hemopexin levels are depleted in pediatric sickle cell disease patients. Blood Cells Mol Dis 72:34–36. https://doi.org/10.1016/j.bcmd.2018.07.002
Carvalho MOS, Rocha LC, Reis JHO, Santos TDA, Do Nascimento VML, Carvalho MB, Luz NF, Borges VDM, Goncalves MS (2015) Heme concentration as a biomarker of sickle cell disease severity: its role in steady-state and in crisis patients. Blood 126(23):975–975. https://doi.org/10.1182/blood.V126.23.975.975
Oninla VO, Breiden B, Babalola JO, Sandhoff K (2014) Acid sphingomyelinase activity is regulated by membrane lipids and facilitates cholesterol transfer by NPC2. J Lipid Res 55(12):2606–2619. https://doi.org/10.1194/jlr.M054528
**ong X, Lee C-F, Li W, Yu J, Zhu L, Kim Y, Zhang H, Sun H (2019) Acid sphingomyelinase regulates the localization and trafficking of palmitoylated proteins. Biology Open 8(10):bio040311. https://doi.org/10.1242/bio.040311
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008 (5). Ethical approval for the study was obtained from the Clinical Research Ethics Committee of Mersin University Faculty of Medicine (Decision number: 78017789/050.01.04/529310).
Informed consent
Informed consent was obtained from all patients for being included in the study. All patients or their caretakers signed written informed consent forms when they were admitted to the ward.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yildirim, N., Unal, S., Yalcinkaya, A. et al. Evaluation of the relationship between intravascular hemolysis and clinical manifestations in sickle cell disease: decreased hemopexin during vaso-occlusive crises and increased inflammation in acute chest syndrome. Ann Hematol 101, 35–41 (2022). https://doi.org/10.1007/s00277-021-04667-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-021-04667-w